Abstract
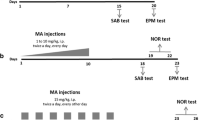
Methamphetamine (MA) alters dopamine markers and cognitive function in heavy users. In rodents, there are MA dosing regimens that induce concordant effects using repeated administration at spaced intervals. These regimens are effective but complicate experiments designed to disentangle the effects of the drug on different brain regions in relation to their cognitive effects because of treatment spacing. In an effort to simplify the model, we tested whether a single dose of MA could induce the same monoamine and cognitive effects as multiple, spaced dosing without affecting survival. Adult male Sprague-Dawley rats were treated with 40 mg/kg MA subcutaneously once and tested starting 2 weeks later. MA-treated rats showed deficits in egocentric navigation in Cincinnati water maze, in spatial navigation in the Morris water maze, and in choosing a consistent problem-solving strategy in the Star water maze when given the option to show a preference. MA-treated rats had persistent dopamine and serotonin reductions in the neostriatum and nucleus accumbens, and serotonin reductions in the hippocampus of the same magnitude as in repetitive treatment models. The data demonstrate that a single dose recapitulates the neurocognitive and monoamine effects of multiple-dose regimens, thereby simplifying the model of MA-induced neurotoxicity.







Similar content being viewed by others
References
Bortolato M, Frau R, Piras AP, Luesu W, Bini V, Diaz G, Gessa G, Ennas MG, Castelli MP (2009) Methamphetamine induces long-term alterations in reactivity to environmental stimuli: correlation with dopaminergic and serotonergic toxicity. Neurotox Res 15(3):232–245. https://doi.org/10.1007/s12640-009-9024-2
Braun AA, Amos-Kroohs RM, Gutierrez A, Lundgren KH, Seroogy KB, Skelton MR, Vorhees CV, Williams MT (2015) Dopamine depletion in either the dorsomedial or dorsolateral striatum impairs egocentric Cincinnati water maze performance while sparing allocentric Morris water maze learning. Neurobiol Learn Mem 118:55–63. https://doi.org/10.1016/j.nlm.2014.10.009
Braun AA, Amos-Kroohs RM, Gutierrez A, Lundgren KH, Seroogy KB, Vorhees CV, Williams MT (2016) 6-Hydroxydopamine-induced dopamine reductions in the nucleus accumbens, but not the medial prefrontal cortex, impair Cincinnati water maze egocentric and Morris water maze allocentric navigation in male Sprague–Dawley rats. Neurotox Res 30(2):199–212. https://doi.org/10.1007/s12640-016-9616-6
Braun AA, Graham DL, Schaefer TL, Vorhees CV, Williams MT (2012) Dorsal striatal dopamine depletion impairs both allocentric and egocentric navigation in rats. Neurobiol Learn Mem 97(4):402–408. https://doi.org/10.1016/j.nlm.2012.03.004
Buzsaki G, Moser EI (2013) Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci 16(2):130–138. https://doi.org/10.1038/nn.3304
Cappon GD, Pu C, Vorhees CV (2000) Time-course of methamphetamine-induced neurotoxicity in rat caudate-putamen after single-dose treatment. Brain Res 863(1-2):106–111. https://doi.org/10.1016/S0006-8993(00)02107-7
Chang L, Alicata D, Ernst T, Volkow N (2007) Structural and metabolic brain changes in the striatum associated with methamphetamine abuse. Addiction 102:16–32. https://doi.org/10.1111/j.1360-0443.2006.01782.x
Cheng RK, Etchegaray M, Meck WH (2007) Impairments in timing, temporal memory, and reversal learning linked to neurotoxic regimens of methamphetamine intoxication. Brain Res 1186:255–266. https://doi.org/10.1016/j.brainres.2007.10.002
Cook D, Kesner RP (1988) Caudate nucleus and memory for egocentric localization. Behav Neural Biol 49(3):332–343. https://doi.org/10.1016/S0163-1047(88)90338-X
Daberkow DP, Kesner RP, Keefe KA (2005) Relation between methamphetamine-induced monoamine depletions in the striatum and sequential motor learning. Pharmacol Biochem Behav 81(1):198–204. https://doi.org/10.1016/j.pbb.2005.03.010
Fouquet C, Babayan BM, Watilliaux A, Bontempi B, Tobin C, Rondi-Reig L (2013) Complementary roles of the hippocampus and the dorsomedial striatum during spatial and sequence-based navigation behavior. PLoS One 8(6):e67232. https://doi.org/10.1371/journal.pone.0067232
Friedman SD, Castañeda E, Hodge GK (1998) Long-term monoamine depletion, differential recovery, and subtle behavioral impairment following methamphetamine-induced neurotoxicity. Pharmacol Biochem Behav 61(1):35–44. https://doi.org/10.1016/S0091-3057(98)00066-5
Friend DM, Keefe K a (2013) A role for D1 dopamine receptors in striatal methamphetamine-induced neurotoxicity. Neurosci Lett 555:243–247. https://doi.org/10.1016/j.neulet.2013.08.039
Fukumura M, Cappon GD, Pu C, Broening HW, Vorhees CV (1998) A single dose model of methamphetamine-induced neurotoxicity in rats: effects on neostriatal monoamines and glial fibrillary acidic protein. Brain Res 806(1):1–7. https://doi.org/10.1016/S0006-8993(98)00656-8
Gasbarri A, Pompili A, Packard MG, Tomaz C (2014) Habit learning and memory in mammals: behavioral and neural characteristics. Neurobiol Learn Mem 114:198–208. https://doi.org/10.1016/j.nlm.2014.06.010
Ghahremani DG, Tabibnia G, Monterosso J, Hellemann G, Poldrack RA, London ED (2011) Effect of modafinil on learning and task-related brain activity in methamphetamine-dependent and healthy individuals. Neuropsychopharmacology 36(5):950–959. https://doi.org/10.1038/npp.2010.233
Gross NB, Duncker PC, Marshall JF (2011) Striatal dopamine D1 and D2 receptors: widespread influences on methamphetamine-induced dopamine and serotonin neurotoxicity. Synapse 65(11):1144–1155. https://doi.org/10.1002/syn.20952
Gutierrez, A., Jablonski, S.A., Amos-kroohs, R.M., Barnes, A.C., Williams, M.T., Vorhees, C. V, 2017. Effects of housing on methamphetamine-induced neurotoxicity and spatial learning and memory. doi:https://doi.org/10.1021/acschemneuro.6b00419
Hafting T, Fyhn M, Molden S, Moser M-B, Moser EI (2005) Microstructure of a spatial map in the entorhinal cortex. Nature 436(7052):801–806. https://doi.org/10.1038/nature03721
Herring, N.R., Gudelsky, G. a., Vorhees, C. V., Williams, M.T., 2010. (+)-Methamphetamine-induced monoamine reductions and impaired egocentric learning in adrenalectomized rats is independent of hyperthermia. Synapse 64, 773–785. doi:https://doi.org/10.1002/syn.20784
Herring NR, Schaefer TL, Gudelsky GA, Vorhees CV, Williams MT (2008a) Effect of (+)-methamphetamine on path integration learning, novel object recognition, and neurotoxicity in rats. Psychopharmacology 199(4):637–650. https://doi.org/10.1007/s00213-008-1183-y
Herring NR, Schaefer TL, Tang PH, Skelton MR, Lucot JP, Gudelsky GA, Vorhees CV, Williams MT (2008b) Comparison of time-dependent effects of (+)-methamphetamine or forced swim on monoamines, corticosterone, glucose, creatine, and creatinine in rats. BMC Neurosci 9(1):49. https://doi.org/10.1186/1471-2202-9-49
Heysieattalab S, Naghdi N, Zarrindast MR, Haghparast A, Mehr SE, Khoshbouei H (2016) The effects of GABAA and NMDA receptors in the shell-accumbens on spatial memory of METH-treated rats. Pharmacol Biochem Behav 142:23–35. https://doi.org/10.1016/j.pbb.2015.12.008
Howland JG, Harrison RA, Hannesson DK, Phillips AG (2008) Ventral hippocampal involvement in temporal order, but not recognition, memory for spatial information. Hippocampus 18(3):251–257. https://doi.org/10.1002/hipo.20396
Kelley AE, Domesick VB (1982) The distribution of the projection from the hippocampal formation to the nucleus accumbens in the rat: an anterograde and retrograde-horseradish peroxidase study. Neuroscience 7(10):2321–2335. https://doi.org/10.1016/0306-4522(82)90198-1
Kesner RP, Farnsworth G, DiMattia BV (1989) Double dissociation of egocentric and allocentric space following medial prefrontal and parietal cortex lesions in the rat. Behav Neurosci 103(5):956–961. https://doi.org/10.1037/0735-7044.103.5.956
Kish SJ, Fitzmaurice PS, Boileau I, Schmunk GA, Ang LC, Furukawa Y, Chang LJ, Wickham DJ, Sherwin A, Tong J (2009) Brain serotonin transporter in human methamphetamine users. Psychopharmacology 202(4):649–661. https://doi.org/10.1007/s00213-008-1346-x
Simon L, Domier C, Jennife S, Domier C, Carnell J, Brethen P, Rawson R, Ling W (2000) Cognitive impairment in individuals currently using methamphetamine. Am J Addict 9(3):222–231. https://doi.org/10.1080/10550490050148053
Marshall, J.F., Belcher, A.M., Feinstein, E.M., O’Dell, S.J., 2007. Methamphetamine-induced neural and cognitive changes in rodents. Addiction 102 Suppl, 61–9. doi:https://doi.org/10.1111/j.1360-0443.2006.01780.x, 69
McCann UD, Wong DF, Yokoi F, Villemagne V, Dannals RF, Ricaurte GA (1998) Reduced striatal dopamine transporter density in abstinent methamphetamine and methcathinone users: evidence from positron emission tomography studies with [11C]WIN-35,428. J Neurosci 18(20):8417–8422
McGeorge AJ, Faull RLM (1989) The organization of the projection from the cerebral cortex to the striatum in the rat. Neuroscience 29(3):503–537. https://doi.org/10.1016/0306-4522(89)90128-0
McNamara RK, Skelton RW (1993) The neuropharmacological and neurochemical basis of place leaning in the Morris water maze. Brain Res Brain Res Rev Jan-Apr 18(1):33–49. https://doi.org/10.1016/0165-0173(93)90006-L
Moon M, Do KS, Park J, Kim D (2007) Memory impairment in methamphetamine dependent patients. Int J Neurosci 117(1):1–9. https://doi.org/10.1080/00207450500535503
Morris RGM, Garrud P, Rawlins JNP, O’Keefe J (1982) Place navigation impaired in rats with hippocampal lesions. Nature 297(5868):681–683. https://doi.org/10.1038/297681a0
Morris RGM, Hagan JJ, Rawlins JNP (1986) Allocentric spatial learning by Hippocampectomised rats: a further test of the “spatial mapping” and “working memory” theories of hippocampal function. Q J Exp Psychol Sect B 38:365–395. https://doi.org/10.1080/14640748608402242
O’Callaghan JP, Miller DB (1994) Neurotoxicity profiles of substituted amphetamines in the C57BL/6J mouse. J Pharmacol Exp Ther 270(2):741–751
O’Keefe J, Nadel L (1979) Précis of O’Keefe & Nadel’s The hippocampus as a cognitive map. Behav Brain Sci 2:487–494. https://doi.org/10.1017/S0140525X00063949
Packard MG (2009) Exhumed from thought: basal ganglia and response learning in the plus-maze. Behav Brain Res 199(1):24–31. https://doi.org/10.1016/j.bbr.2008.12.013
Reichel CM, Ramsey LA, Schwendt M, McGinty JF, See RE (2012) Methamphetamine-induced changes in the object recognition memory circuit. Neuropharmacology 62(2):1119–1126. https://doi.org/10.1016/j.neuropharm.2011.11.003
Rondi-Reig L (2006) Impaired sequential egocentric and allocentric memories in forebrain-specific-NMDA receptor knock-out mice during a new task dissociating strategies of navigation. J Neurosci 26(15):4071–4081. https://doi.org/10.1523/JNEUROSCI.3408-05.2006
Salo R, Nordahl TE, Possin K, Leamon M, Gibson DR, Galloway GP, Flynn NM, Henik A, Pfefferbaum A, Sullivan EV (2002) Preliminary evidence of reduced cognitive inhibition in methamphetamine-dependent individuals. Psychiatry Res 111(1):65–74. https://doi.org/10.1016/S0165-1781(02)00111-7
Schroder N, O’Dell SJ, Marshall JF (2003) Neurotoxic methamphetamine regimen severely impairs recognition memory in rats. Synapse 49(2):89–96. https://doi.org/10.1002/syn.10210
Sekine Y, Ouchi Y, Takei N, Yoshikawa E, Nakamura K, Futatsubashi M, Okada H, Minabe Y, Suzuki K, Iwata Y, Tsuchiya KJ, Tsukada H, Iyo M, Mori N (2006) Brain serotonin transporter density and aggression in abstinent methamphetamine abusers. Arch Gen Psychiatry 63(1):90–100. https://doi.org/10.1001/archpsyc.63.1.90
Son J-H, Latimer C, Keefe KA (2011) Impaired formation of stimulus–response, but not action–outcome, associations in rats with methamphetamine-induced neurotoxicity. Neuropsychopharmacology 36(12):2441–2451. https://doi.org/10.1038/npp.2011.131
Sonsalla PK, Gibb JW, Hanson GR (1986) Roles of D1 and D2 dopamine receptor subtypes in mediating the methamphetamine-induced changes in monoamine systems. J Pharmacol Exp Ther 238(3):932–937
Thierry AM, Gioanni Y, Dégénétais E, Glowinski J (2000) Hippocampo-prefrontal cortex pathway: anatomical and electrophysiological characteristics. Hippocampus 10(4):411–419. https://doi.org/10.1002/1098-1063(2000)10:4<411::AID-HIPO7>3.0.CO;2-A
Volkow ND, Chang L, Wang GJ, Fowler JS, Leonido-Yee M, Franceschi D, Sedler MJ, Gatley SJ, Hitzemann R, Ding YS, Logan J, Wong C, Miller EN (2001) Association of dopamine transporter reduction with psychomotor impairment in methamphetamine abusers. Am J Psychiatry 158(3):377–382. https://doi.org/10.1176/appi.ajp.158.3.377
Vorhees CV, He E, Skelton MR, Graham DL, Schaefer TL, Grace CE, Braun A a, Amos-Kroohs R, Williams MT (2011) Comparison of (+)-methamphetamine, ±-methylenedioxymethamphetamine, (+)-amphetamine and ±-fenfluramine in rats on egocentric learning in the Cincinnati water maze. Synapse 65(5):368–378. https://doi.org/10.1002/syn.20854
Vorhees CV, Herring NR, Schaefer TL, Grace CE, Skelton MR, Johnson HL, Williams MT (2008) Effects of neonatal (+)-methamphetamine on path integration and spatial learning in rats: effects of dose and rearing conditions. Int J Dev Neurosci 26(6):599–610. https://doi.org/10.1016/j.ijdevneu.2008.04.002
Vorhees CV, Skelton MR, Grace CE, Schaefer TL, Graham DL, Braun AA, Williams MT (2009) Effects of (+)-methamphetamine on path integration and spatial learning, but not locomotor activity or acoustic startle, align with the stress hyporesponsive period in rats. Int J Dev Neurosci 27(3):289–298. https://doi.org/10.1016/j.ijdevneu.2008.12.003
Vorhees CV, Williams MT (2016) Cincinnati water maze: a review of the development, methods, and evidence as a test of egocentric learning and memory. Neurotoxicol Teratol 57:1–19. https://doi.org/10.1016/j.ntt.2016.08.002
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1(2):848–858. https://doi.org/10.1038/nprot.2006.116.Morris
Wilson JM, Kalasinsky KS, Levey AI, Bergeron C, Reiber G, Anthony RM, Schmunk GA, Shannak K, Haycock JW, Kish SJ (1996) Striatal dopamine nerve terminal markers in human, chronic methamphetamine users. Nat Med 2(6):699–703. https://doi.org/10.1038/nm0696-699
Yin HH, Knowlton BJ, Balleine BW (2004) Lesions of dorsolateral striatum preserve outcome expectancy but disrupt habit formation in instrumental learning. Eur J Neurosci 19(1):181–189. https://doi.org/10.1111/j.1460-9568.2004.03095.x
Zhong N, Jiang H, Du J, Zhao Y, Sun H, Xu D, Li C, Zhuang W, Li X, Hashimoto K, Zhao M (2016) The cognitive impairments and psychological wellbeing of methamphetamine dependent patients compared with health controls. Prog Neuro-Psychopharmacology Biol Psychiatry 69:31–37. https://doi.org/10.1016/j.pnpbp.2016.04.005
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gutierrez, A., Williams, M.T. & Vorhees, C.V. A Single High Dose of Methamphetamine Reduces Monoamines and Impairs Egocentric and Allocentric Learning and Memory in Adult Male Rats. Neurotox Res 33, 671–680 (2018). https://doi.org/10.1007/s12640-018-9871-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-018-9871-9