Abstract
Purpose
Non-invasive cerebral oxygen saturation (ScO2) monitoring is an established tool in the intraoperative phase of pediatric congenital cardiac surgery (CCS). This study investigated the association between ScO2 and postoperative outcome by investigating both baseline ScO2 values and intraoperative desaturations from baseline.
Methods
All CCS procedures performed in the period 2010-2017 in our institution in which ScO2 was monitored were included in this historical cohort study. Baseline ScO2 was determined after tracheal intubation, before surgical incision. Subgroups were based on cardiac pathology and degree of intracardiac shunting. Poor outcome was defined based on length of stay (LOS) in the intensive care unit (ICU)/hospital, duration of mechanical ventilation (MV), and 30-day mortality. Intraoperatively, ScO2 total time below baseline (TBBL) and ScO2 time-weighted average (TWA) were calculated.
Results
Data from 565 patients were analyzed. Baseline ScO2 was significantly associated with LOS in ICU (odds ratio [OR] per percentage decrease in baseline ScO2, 0.95; 95% confidence interval [CI], 0.93 to 0.97; P < 0.001), with LOS in hospital (OR, 0.93; 95% CI, 0.91 to 0.96; P < 0.001), with MV duration (OR, 0.92; 95% CI, 0.90 to 0.95; P < 0.001) and with 30-day mortality (OR, 0.94; 95% CI, 0.91 to 0.98; P = 0.007). Cerebral oxygen saturation TWA had no associations, while ScO2 TBBL had only a small association with LOS in ICU (OR, 1.02; 95% CI, 1.01 to 1.03; P < 0.001), MV duration (OR,1.02; 95% CI, 1.01 to 1.03; P = 0.002), and LOS in hospital (OR, 1.02; 95% CI, 1.01 to 1.04; P < 0.001).
Conclusion
In pediatric patients undergoing cardiac surgery, low baseline ScO2 values measured after tracheal intubation were associated with several adverse postoperative outcomes. In contrast, the severity of actual intraoperative cerebral desaturation was not associated with postoperative outcomes. Baseline ScO2 measured after tracheal intubation may help identify patients at increased perioperative risk.
Résumé
Objectif
Le monitorage non invasif de la saturation cérébrale en oxygène (ScO2) est un outil bien établi en phase peropératoire de chirurgie cardiaque congénitale pédiatrique. Cette étude a examiné l’association entre la ScO2 et le pronostic postopératoire en étudiant les valeurs de ScO2 initiales et les désaturations peropératoires par rapport à ces valeurs.
Méthode
Toutes les interventions en chirurgie cardiaque congénitale réalisées entre 2010 et 2017 dans notre établissement et au cours desquelles la ScO2 a été monitorée ont été incluses dans cette étude de cohorte historique. La ScO2 de base était déterminée après l’intubation trachéale, avant l’incision chirurgicale. Les sous-groupes ont été catégorisés en fonction de la pathologie cardiaque et des shunts intracardiaques. Un mauvais pronostic était défini en fonction de la durée de séjour à l’unité de soins intensifs (USI)/ l’hôpital, de la durée de ventilation mécanique et de la mortalité à 30 jours. Pendant l’intervention, le temps total pendant lequel la ScO2 était au-dessous des valeurs de base et la moyenne pondérée dans le temps ont été calculés.
Résultats
Les données de 565 patients ont été analysées. Une association significative a été observée entre la ScO2 de base et la durée de séjour à l’USI (diminution du rapport de cotes [RC] par pourcentage de la ScO2 de base, 0,95; intervalle de confiance [IC] 95 %, 0,93 à 0,97; P < 0,001), la durée de séjour à l’hôpital (RC, 0,93; IC 95 %, 0,91 à 0,96; P < 0,001), la durée de ventilation mécanique (RC, 0,92; IC 95 %, 0,90 à 0,95; P < 0,001) et la mortalité à 30 jours (RC, 0,94; IC 95 %, 0,91 à 0,98; P = 0,007). La moyenne pondérée dans le temps de la saturation cérébrale en oxygène n’a pas révélé d’association, alors que le temps total au-dessous des valeurs de base de ScO2 n’a révélé qu’une petite association avec la durée de séjour à l’USI (RC, 1,02; IC 95 %, 1,01 à 1,03; P < 0,001), la durée de ventilation mécanique (RC, 1,02; IC 95 %, 1,01 à 1,03; P = 0,002), et la durée de séjour à l’hôpital (RC, 1,02; IC 95 %, 1,01 à 1,04; P < 0,001).
Conclusion
Chez les patients pédiatriques subissant une chirurgie cardiaque, des valeurs de ScO2 basses lorsque mesurées après l’intubation trachéale étaient associées à plusieurs complications postopératoires. En revanche, la gravité de la désaturation cérébrale peropératoire n’était pas associée aux devenirs postopératoires. La ScO2 de base mesurée après l’intubation trachéale pourrait nous aider à identifier les patients courant un risque périopératoire accru.
Similar content being viewed by others
The survival of pediatric patients with congenital heart disease (CHD) that undergo congenital cardiac surgery (CCS) has improved substantially in the last decades.1 Still, in the (early) postoperative phase following CCS, mortality remains increased.1,2 Especially in the perioperative phase, CCS procedures can be complicated by organ injury3 such as acute kidney injury,4 and adverse neurologic events such as seizures and strokes.5 The development of such complications can prolong treatment in the intensive care unit (ICU), including protracted duration of mechanical ventilation (MV), and can lead to longer hospitalization, which is associated with increased morbidity and mortality,1,2,3,6 along with higher costs.1,6,7
The use of cerebral near-infrared spectroscopy (NIRS) for measuring cerebral oxygen saturation (ScO2) has gained an established role in the perioperative monitoring of cerebral perfusion and oxygenation in pediatric CCS procedures.8,9 Yet, its effect on influencing outcome by improving perioperative hemodynamic management in CCS remains elusive; in particular conditions of CCS, e.g., in surgical correction of the hypoplastic left heart syndrome,10 ScO2 was shown to predict postoperative outcome and improve postoperative outcome only in small studies.11
The spectrum of CHD is, however, broad with substantial differences in cardiopulmonary anatomy and pathology—associated differences in perioperative NIRS readings precludes generalization of perioperative ScO2 readings.12,13,14
Moreover, the influence of intraoperative cerebral desaturation on postoperative outcome in this patient population is unclear. There is also no evidence that prevention of intraoperative cerebral desaturation will reduce the likelihood of an adverse (neurologic) event.
Therefore, the primary goal of this study was to investigate whether there is an association between postoperative outcome and ScO2 values determined either at baseline (after induction of general anesthesia and tracheal intubation, pre-incision) or during the intraoperative phase in a broad population of pediatric CHD patients undergoing CCS.
Methods
Design and selection criteria
This was a historical cohort study of pediatric patients undergoing CCS in our institution from January 2010 until December 2017. The study has been approved by the local ethics committee (University Medical Center Groningen, Netherlands, Registration number: 2016/036; 22 February 2016) and the requirement for written informed consent was waived by the institutional review board given its retrospective design. This manuscript adheres to the applicable Strengthening the Reporting of Observational Studies in Epidemiology guideline.
All elective cases of pediatric CCS were included for analysis when a) the surgical procedure involved surgical correction of a congenital cardiac problem, and b) ScO2 was monitored continuously in the intraoperative period.
To account for differences in congenital cardiac conditions, patients were divided into four subgroups as described previously14: “no cyanosis, no shunting”, “no cyanosis, but left-to-right (L–R) shunting”, “cyanosis without L–R shunting” and “cyanosis with L–R shunting”.
Anesthetic management
Given the historical character of the current study, induction and maintenance of anesthesia, and intraoperative hemodynamic management in response to ScO2 reductions were at the discretion of the attending anesthesiologist. General anesthesia was induced either by inhalation using sevoflurane or intravenously using propofol or etomidate. Maintenance of anesthesia was achieved with continuous infusion of propofol or midazolam and sufentanil; muscle relaxation was achieved using rocuronium or pancuronium. Ventilation was adjusted to the patient’s individual weight, physiology, and desired intrathoracic pressure using either pressure- or volume-controlled mode. Arterial oxygen saturation, five-leads continuous electrocardiogram, and end-tidal carbon dioxide (etCO2) were monitored continuously through the operation. Invasive blood pressure was also monitored through cannulation of the radial or femoral artery. The central vein was accessed through the internal jugular, subclavian, or femoral vein, depending on patient and surgical characteristics. Arterial and venous blood gas samples were taken during the operation at appropriate time points. Cerebral oxygen saturation was measured by NIRS (INVOS® 5100C cerebral oximetry monitor; Medtronic, MN, USA). One or two pediatric sensors were placed on either side of the forehead, depending on the preference of the attending anesthesiologist, and the size of the child. In the study period, there was no hemodynamic optimization algorithm on which interventions were based. Also, transesophageal echocardiography, continuous cardiac output monitoring, and somatic oxygen saturation monitoring were not used routinely and were not further considered in this retrospective study. Anesthesiologists were not blinded to the ScO2 measurements.
Data collection and handling
In our institution, all cardiothoracic surgery operating rooms are equipped with medical grade computers for collecting the electronic medical records, including the continuous registration of data from the ventilator and vital signs monitor (IntelliVue MX800, Philips, Eindhoven, The Netherlands). For the purpose of this study, relevant data were extracted to a separate database (Excel, Microsoft, Redmond, USA) for further analysis. Along with all intraoperative data, patient characteristics were collected (diagnosis, sex, age, weight, and height at the time of surgical intervention). Also, the duration of surgery, cardiopulmonary bypass (CPB) time, and aorta clamping time were collected.
An automated algorithm was used to eliminate ScO2 artefacts: ScO2 values were omitted when 1) values were implausible, i.e., below 15% and above 95% (according to the manufacturer specifications),15 and 2) obvious artefacts were observed, which were defined as abrupt changes in three consecutive values by more than 50% and an abrupt return to at least 80% of the first value. Additionally, all data were reviewed manually by two researchers independently to account for obvious artefacts not covered by the algorithm. When a mutual decision on artefactual data could not be reached, the case was excluded.
Per patient, baseline ScO2 was defined at two minutes before surgical incision, as ScO2 was assumed to have reached steady-state conditions with an inspired fraction of oxygen (FiO2) between 0.3 and 0.4, and etCO2 between 5 and 6% at that time point. Data were reviewed visually to assure that baseline ScO2 was defined at an appropriate, hemodynamically stable time point.
In case of two simultaneous ScO2 values (bi-frontal measurement), the mean of both values was taken and established as the baseline value per patient.
Additionally, the Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery (STS-EACTS) mortality score, which primarily depends on the complexity of the surgical intervention, was calculated per patient.16,17
Outcomes
The distribution of outcome variables was calculated per subgroup (as based on the congenital cardiac pathology described above) to provide an assessment of “poor outcome” per subgroup, since it was assumed that the absolute values of the outcome variables would substantially differ per subgroup. Here, “poor outcome”—which indicates a protracted postoperative duration of MV or LOS on the ICU or in the hospital—was defined by dichotomization, for which values above the third quartile were considered a “poor outcome”. Secondary, in-hospital mortality, 30-day mortality, and one-year mortality after surgery were investigated. All outcome variables were defined before the start of data collection.
Statistical analysis
Per patient, the individual ScO2 baseline values (defined as outlined above) served as basis for subsequent statistical analyses. Intraoperative declines in ScO2 relative to this baseline were calculated as the individual time below ScO2 baseline (TBBL). After calculating the individual area under the curve (AUC = product of absolute ScO2 deviation below baseline and the corresponding duration in minutes), the time-weighted average (TWA) of intraoperative desaturations could be calculated, which is equivalent to dividing the AUC by the individual duration of surgery.18
The relationship between outcome and ScO2 was assessed using a logistic regression model, which included the variables baseline ScO2, TBBL, TWA, and the congenital cardiac subgroup (the latter accounted for the different diagnoses). These predictors were entered in a stepwise backward model using the likelihood ratio statistic for each of the three outcome variables LOS in ICU, LOS in hospital, and length of MV. For the relationship with 30-day mortality, the STS-EACTS score was added to the model. Goodness of fit was assessed using the Hosmer-Lemeshow test and associated odds ratios (OR) are given to assess the odds after a unit change in the investigated predictors.
Receiver operating characteristics (ROC) analysis was performed to assess the predictive ability of baseline ScO2 with regard to mortality in terms of sensitivity and specificity. Optimal cut-off values were calculated using the Youden index. The STS-EACTS score was included in this analysis as a reference. The ROC-derived optimal baseline ScO2 cut-off value was included in a Kaplan-Meier survival analysis for comparison of mortality.
For all performed tests, statistical significance was set at a P value of < 0.05. Tests were adjusted for multiple testing to control for the family-wise inflation of type I error rate, when deemed relevant. All tests were performed two-sided. All data were collected and synchronized in Microsoft Excel 2010 (Redmond, USA). Statistical analysis was performed using IBM SPSS Statistics 23 (IBM Inc., Chicago, IL, USA) and MedCalc (MedCalc Software, Ostend, Belgium).
Results
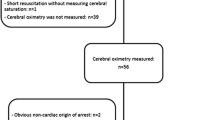
A total of 1,060 congenital cardiac surgical procedures were performed from January 2010 to December 2017 in our institution (Fig. 1). Patients in whom ScO2 was not available (n = 317) or in whom non-cardiac surgical procedures were performed (n = 40) were not included for further analysis. Eventually, 565 patients were included in the final analysis. The distribution of patients between the CHD subgroups, as well as their individual diagnoses, is given in Table 1. Patient characteristics and general surgical characteristics are presented in Table 2. The postoperative outcome characteristics of all patients are summarized in Table 3.
Baseline ScO2
In general, baseline values of ScO2 (i.e., after tracheal intubation, before surgical incision) were lower in cyanotic patients (subgroup 3 and 4) than in non-cyanotic patients (subgroup 1 and 2): 60% vs 67%, respectively, P < 0.001 (Table 4). There were no further differences in baseline ScO2 in patients with and without L-R shunting in either non-cyanotic (group 2 vs group 1, respectively) and cyanotic patients (group 4 vs group 3, respectively).
Baseline ScO2 was significantly associated with all defined postoperative outcome variables (Table 5), meaning that a lower baseline ScO2 was associated with an increased odds for the respective outcome variables (LOS in ICU: OR, 0.95; 95% confidence interval [CI], 0.93 to 0.97; duration of MV: OR, 0.92; 95% CI, 0.90 to 0.95; LOS in hospital: OR, 0.93; 95% CI, 0.91 to 0.96; 30-day mortality: OR, 0.94; 95% CI. 0.91 to 0.98). Of note, the cardiac pathology subgroup itself was either not related to outcome (LOS in ICU: OR, 0.85; 95% CI, 0.71 to 1.03; P = 0.1; duration of MV: OR, 0.85; 95% CI, 0.69 to 1.05; P = 0.13) or was removed from the model (LOS in hospital and 30-day mortality) as it did not improve the model.
Baseline ScO2 showed a stronger association with 30-day mortality than the STS-EACTS score (OR per percentage decrease in baseline ScO2, 0.94; 95% CI, 0.91 to 0.98; vs OR per unit increase in STS-EACTS score, 1.56; 95% CI, 0.95 to 2.53, respectively).
Intraoperative ScO2
The data on intraoperative ScO2 values are presented in Table 4. Overall intraoperative ScO2 values were lower in cyanotic patients (subgroups 3 and 4) than in non-cyanotic patients (subgroups 1 and 2) (61% vs 66%, respectively; P < 0.001).
In 483 patients (86%), ScO2 decreased below individual baseline values at some time point during surgery, irrespective of the length and degree of desaturation. There were no differences in the incidence of intraoperative ScO2 desaturations between the subgroups (n = 11, 47, 3, and 21 for subgroups 1-4, respectively).
Cerebral oxygen saturation TWA showed no association with any of the outcome variables (Table 5). Cerebral oxygen saturation TBBL was only weakly associated with the other outcome variables, except 30-day mortality (LOS in ICU: OR, 1.02; 95% CI, 1.01 to 1.03; P < 0.001; duration of MV: OR, 1.02; 95% CI, 1.01 to 1.03; P = 0.002; LOS in hospital: OR, 1.02; 95% CI, 1.01 to 1.04; P < 0.001).
Prediction of postoperative outcome by baseline ScO2 and STS-EACTS score
The areas under the ROC curve for the prediction of 30-day mortality by baseline ScO2 and STS-EACTS was comparable (0.66 [95% CI, 0.55 to 0.76] vs 0.65 [95% CI, 0.54 to 0.77], respectively) and both were significantly different from the reference line (P = 0.006 and P = 0.007, respectively). The optimal cut-off baseline ScO2 value of 60%—as derived by the Youden index—for predicting 30-day mortality was subsequently used as factor in a Kaplan-Meier survival analysis for analyzing one-year mortality. This analysis revealed a substantial difference in mortality: patients with a baseline ScO2 > 60% survived significantly longer than patients with a baseline ScO2 below 60% (Mantel-Cox [X2 = 10]; P = 0.001; Fig. 2).
Discussion
We investigated the association between intraoperative NIRS-derived ScO2 and surgical outcome after pediatric CCS performed in our hospital over an eight-year period. In this large historical analysis, lower baseline ScO2 values—determined after tracheal intubation but before surgical incision—were associated with a longer ICU and hospital stay, as well as with a longer duration of MV, irrespective of the congenital cardiac pathology. Moreover, baseline ScO2 showed a stronger association with 30-day mortality than the STS-EACTS score did. In contrast, although intraoperative cerebral desaturation occurred frequently in a substantial number of patients, there was no clear association between both the extent and severity of cerebral desaturation and any of the investigated outcome variables.
Baseline ScO2
Cerebral oxygen saturation monitoring has gained an established role during CCS procedures; however, little is known about its impact on postoperative outcome. Here, we performed a large historical analysis of the association between intraoperative ScO2 and postoperative outcome in patients that underwent CCS. Also, we included all forms of both cyanotic and non-cyanotic cardiac pathologies while we analyzed outcomes separately per group, accounting for intrinsic between-group differences. For baseline ScO2, there was a clear association with all of the four investigated outcome variables, i.e., 30-day mortality, LOS in hospital, LOS in the ICU, and length of MV. This finding is only in partial agreement with a recent study,19 as we found an association of baseline ScO2 with outcome only in cyanotic CHD patients. Of note, in that small retrospective study (n = 59), outcome was defined differently, and was a composite of death, need for renal replacement therapy, use of extracorporeal membrane oxygenation, or increased length of MV or ICU stay. Also, ScO2 values were lower in cyanotic patients with poor outcome compared with cyanotic patients without poor outcome, while in non-cyanotic patients, no clear association could be found using univariate analysis. In our multivariable logistic regression model, we incorporated the different congenital cardiac conditions as defined previously.14 Additionally, we used the distribution of primary outcome variables per subgroup (Table 1) to define “poor outcome” (i.e., > third quartile of that variable), and hence we were able to provide a more nuanced association between postoperative outcome and CCS, for specific types of CHD, including non-cyanotic (groups 1 and 2) and cyanotic (groups 3 and 4) patients. The latter discrimination is highly relevant, since age and complexity of the surgical procedure are acknowledged predictors of outcome following CCS,1 but are intrinsically linked in the definition(s) of the subgroups, which improves the robustness of our analysis.
It is important to consider that, although the observed association between baseline ScO2 and outcome was clear, the magnitude of its impact is relatively limited, reflected by ORs between 0.92 and 0.95. It should, however, be reckoned that in a complex setting like CCS, factors influencing outcome are multifactorial.20 The finding that even an established mortality prediction score like the STS-EACTS score only moderately predicts mortality reflects the complex and multifactorial context of CCS, in which the value of only one variable should not be overemphasized. Nevertheless, baseline ScO2 values measured after tracheal intubation may at least have some role in identifying those patients at increased risk for adverse postoperative outcome13,21—with a stronger association with 30-day mortality than the STS-EACTS score—and can offer a fast and feasible method to serve as an “early warning tool”, either intraoperatively or in the early postoperative phase.22,23,24,25,26 Hence, baseline ScO2—determined after tracheal intubation—may guide in clinical routing and pre-emptive cardiopulmonary optimization and risk-stratification. Future studies should evaluate this issue further.
Importantly, as shown previously in adult patients undergoing cardiac surgery,21 low ScO2 values (i.e., < 50%) were associated not only with neurologic adverse outcome but also with increased mortality rates. Therefore, as stated in an accompanying editorial,27 the brain might be an “index organ”, reflecting the severity of cardiopulmonary compromise in the individual patient. The observations on baseline ScO2 in our study confirm that this statement might apply to pediatric patients undergoing CCS, even when corrected for the baseline differences in ScO2 per cardiac pathology subgroup.
The definition of a baseline ScO2 value for the population of pediatric CHD patients is debatable, in the context of evaluating its association with intra- and postoperative hemodynamic management and outcome. The observed differences in baseline ScO2 values between cyanotic and non-cyanotic patients (60% vs 67%, respectively) were typical and as expected for these populations.14 In this context, it is important to consider that we determined baseline values under general anesthesia after tracheal intubation, before surgical incision. This time period was chosen for determining baseline values because hemodynamics and FiO2 were both stable. In addition, the previous stress around induction of anesthesia has had time to subside. Also, we speculate that setting a baseline condition (i.e., ScO2) under general anesthesia is more likely to resemble physiologic conditions in the perioperative phase (including the postoperative ICU phase); e.g., cerebral oxygen delivery and consumption of an awake pediatric patient in the ward a day before surgery might be substantially different than under conditions in the surgical or ICU setting. As such, directing ScO2 monitoring and treatment upon ScO2 obtained during stable conditions in an equivalent setting might yield more realistic treatment goals for postoperative hemodynamic management. Nevertheless, to our knowledge, the association between pre- and post-induction ScO2 has not been documented previously, neither has the association of both of these values with postoperative outcome. This issue on “personalized” goal-directed hemodynamic management requires further elucidation in future trials.
Intraoperative ScO2 desaturations
In contrast to our observation on the importance of baseline ScO2 on outcome, we found only a minimal association between intraoperative ScO2 decrease and outcome—only for the length of cerebral desaturation periods intraoperatively, and not for its severity (low values). This observation is surprising, as in a substantial number of patients (> 80%), ScO2 values did decrease below their individual baseline at least some point during surgery, and it was shown that intraoperative cerebral desaturations are associated with reduced postoperative neurologic functioning.28,29 Another study in infants showed abnormalities in psychomotor development and brain magnetic resonance imaging, one year after surgery, in those patients with perioperative periods of diminished cerebral oxygen delivery.30 A more recent multicentre international study measuring the incidence of low regional cerebral oxygenation using NIRS in infants during anesthesia for non-cardiac surgery found that mild and moderate cerebral and arterial desaturations occur frequently, but they were unlikely associated with learning and behavioral abnormalities.31
An important consideration is that the attending anesthesiologist was not blinded to ScO2 readings. In fact, ScO2 monitoring was intentionally applied and was actually used, together with measurement of other variables such as mean arterial pressure and etCO2, for guiding hemodynamic management. Hence, an assumed association between intraoperative ScO2 desaturation and postoperative outcome is likely to be blunted to some extent, as intraoperative management may have prevented more serious ScO2 desaturations than those observed, also by additional interventions such as the transfusion of blood and the monitoring of other vital aspects, e.g., processed electroencephalographic depth-of-anesthesia monitoring, cardiac ultrasound, etc. Hence, the true association between (temporal) decreases in ScO2 and outcome might be underemphasized in our study given that it is likely that in case of ScO2 decreases, prompt action(s) were taken for correction, maybe even before actual desaturation occurred; e.g., in another setting in low birth weight patients (n = 59) who were randomized to receive either dopamine or epinephrine, distinct drug-related changes were seen in cerebral hemodynamics,32 while in another study33 intraoperative ScO2 desaturations were linked with concomitant drops in mean arterial blood pressure. Like in adult cardiac surgery,34 a prospective validation of the use of continuous ScO2 monitoring—and the effects of vasopressors and/or inotropes in optimizing ScO2 and improving outcome in CCS—is still required.
Study limitations
First, this study is a historical cohort study with all the inherent disadvantages; e.g., in most cases ScO2 was measured unilaterally, while bilateral readings were obtained in all cases in the ideal setting. The choice whether ScO2 was measured uni- or bilaterally was, however, made by the attending anesthesiologist, most likely depending on patient characteristics and personal preference, as there is no uniform agreement when to assess ScO2 uni- or bilaterally.35,36,37 Also, while the analysis was based on historical data and outcome variables that were defined a priori or before study initiation, we only assessed lengths of stay, duration of MV, and mortality—not direct measures of neurologic functioning according to uniform definitions.38
Second, our regression model was constructed to identify the strength of the association between NIRS-derived ScO2 values and outcome variables using a stepwise logistic regression model. In addition, the use of stepwise regression models like the one used in this study may suffer from substantial limitations described elsewhere,39,40 which may result in an “overestimation” of the observed associations and their robustness. Given that in this exploratory study was derived from a relatively large data set, for which only five or fewer predictors were defined, the influence of these limitations on the outcome of our analysis is substantially reduced, although it is unknown to what extent. The additional influence of residual confounding, i.e., other variables that are not taken into account but simultaneously affect outcome variables, cannot be determined based on the analysis that we performed. For example, age is intrinsically involved in the underlying cardiac pathology, the indication for CCS, and supposedly, in the risk of morbidity and mortality itself, but was not included in the model itself.14
Third, the ScO2 AUC relative to baseline was calculated using an automated algorithm after elimination of artefacts. Here, the accuracy of the AUC and its derived variable (TWA) is as “good” as the data on which it is based, and is primarily dependent on the availability of ScO2 in a high frequency. For instance, a gap of ScO2 data availability for 30 sec indicated that ScO2 was stable for that time period, and AUC calculations were based on these interpolated values.
Fourth, we have not investigated postoperative values of ScO2. Various studies have looked at these values and their relation with postoperative outcome, at least during the first 24-48 hr after CCS during stay on the ICU stay.11,41,42,43 It was shown in these studies that children with low ScO2 values were hospitalized longer and suffered more complications, including neurologic impairment than children with higher values did. In one study,11 the mean cerebral NIRS of less than 56% over the first 48 hr after surgery yielded a sensitivity of 75% and a specificity of 79% to predict those at risk for subsequent adverse events. In adults,29 the incidence of prolonged cerebral desaturation is significantly higher in patients with cognitive decline. It was suggested to implement NIRS as a standardized tool for monitoring ScO2 in the postoperative period, since time points such as right after weaning from CPB or a few hours postoperatively helped in predicting complications in the early postoperative phase.41,42,44,45
Finally, a study in healthy adults showed that ScO2 readings can be affected by extracranial “contamination”, suggesting that ScO2 does not solely reflect cerebral oxygenation.46 This issue has, however, not yet been studied in pediatric patients, and it is unknown to which extent this might have impacted the observed associations in our study.
Conclusion
A low baseline cerebral oxygen saturation measured after tracheal intubation and before surgical incision was associated with adverse postoperative outcomes in pediatric patients undergoing CCS, while the length of intraoperative cerebral desaturation was only minimally associated with postoperative outcome. Surprisingly, the severity of intraoperative cerebral desaturation was not associated with postoperative outcome. Furthermore, baseline ScO2 values predicted mortality to some extent, equivalent to the already established STS-EACTA score. Therefore, measuring baseline ScO2 after tracheal intubation, might help identify patients at increased perioperative risk.
References
Kempny A, Dimopoulos K, Uebing A, et al. Outcome of cardiac surgery in patients with congenital heart disease in England between 1997 and 2015. PLoS One 2017; . https://doi.org/10.1371/journal.pone.0178963.
Spector LG, Menk JS, Knight JH, et al. Trends in long-term mortality after congenital heart surgery. J Am Coll Cardiol 2018; 71: 2434-46.
DeSena HC, Nelson DP, Cooper DS. Cardiac intensive care for the neonate and child after cardiac surgery. Curr Opin Cardiol 2015; 30: 81-8.
Joffe R, Al Aklabi M, Bhattacharya S, et al. Cardiac surgery-associated kidney injury in children and renal oximetry. Pediatr Crit Care Med 2018; 19: 839-45.
Jafri SK, Ehsan L, Abbas Q, et al. Frequency and outcome of acute neurologic complications after congenital heart disease surgery. J Pediatr Neurosci 2017; 12: 328-31.
Agarwal HS, Wolfram KB, Saville BR, Donahue BS, Bichell DP. Postoperative complications and association with outcomes in pediatric cardiac surgery. J Thorac Cardiovasc Surg 2014; 148: 609-16.e1.
Pasquali SK, He X, Jacobs ML, et al. Excess costs associated with complications and prolonged length of stay after congenital heart surgery. Ann Thorac Surg 2014; 98: 1660-6.
Wong JJ, Chen CK, Moorakonda RB, et al. Changes in near-infrared spectroscopy after congenital cyanotic heart surgery. Front Pediatr 2018; DOI: https://doi.org/10.3389/fped.2018.00097.
Sood BG, McLaughlin K, Cortez J. Near-infrared spectroscopy: applications in neonates. Semin Fetal Neonatal Med 2015; 20: 164-72.
Uebing A, Furck AK, Hansen JH, et al. Perioperative cerebral and somatic oxygenation in neonates with hypoplastic left heart syndrome or transposition of the great arteries. J Thorac Cardiovasc Surg 2011; 142: 523-30.
Phelps HM, Mahle WT, Kim D, et al. Postoperative cerebral oxygenation in hypoplastic left heart syndrome after the Norwood procedure. Ann Thorac Surg 2009; 87: 1490-4.
Jacobs JP, Mayer JE Jr, Pasquali SK, et al. The Society of Thoracic Surgeons Congenital Heart Surgery Database: 2018 update on outcomes and quality. Ann Thorac Surg 2018; 105: 680-9.
Kurth CD, Steven JL, Montenegro LM, et al. Cerebral oxygen saturation before congenital heart surgery. Ann Thorac Surg 2001; 72: 187-92.
Fenton KN, Freeman K, Glogowski K, Fogg S, Duncan KF. The significance of baseline cerebral oxygen saturation in children undergoing congenital heart surgery. Am J Surg 2005; 190: 260-3.
Covidien llc. Operations manual INVOS® system, model 5100C. Mansfield, Massachusetts, USA, 2013. Available at URL: http://www.wemed1.com/downloads/dl/file/id/7947/product/10495/manual_for_mo_s_5100c.pdf (accessed April 2020).
O’Brien SM, Clarke DR, Jacobs JP, et al. An empirically based tool for analyzing mortality associated with congenital heart surgery. J Thorac Cardiovasc Surg 2009; 138: 1139-53.
Jacobs JP, Jacobs ML, Maruszewski B, et al. Initial application in the EACTS and STS congenital heart surgery databases of an empirically derived methodology of complexity adjustment to evaluate surgical case mix and results. Eur J Cardiothorac Surg 2012; 42: 775-80.
Maheshwari K, Khanna S, Bajracharya GR, et al. A randomized trial of continuous noninvasive blood pressure monitoring during noncardiac surgery. Anesth Analg 2018; 127: 424-31.
Saito J, Takekawa D, Kawaguchi J, et al. Preoperative cerebral and renal oxygen saturation and clinical outcomes in pediatric patients with congenital heart disease. J Clin Monit Comput 2019; 33: 1015-22.
Jacobs JP, O’Brien SM, Pasquali SK, et al. The importance of patient-specific preoperative factors: an analysis of the Society of Thoracic Surgeons Congenital Heart Surgery Database. Ann Thorac Surg 2014; 98: 1653-9.
Heringlake M, Garbers C, Kabler JH, et al. Preoperative cerebral oxygen saturation and clinical outcomes in cardiac surgery. Anesthesiology 2011; 114: 58-69.
Hoffman GM, Ghanayem NS, Scott JP, et al. Postoperative cerebral and somatic near-infrared spectroscopy saturations and outcome in hypoplastic left heart syndrome. Ann Thorac Surg 2017; 103: 1527-35.
Murkin JM, Adams SJ, Novick RJ, et al. Monitoring brain oxygen saturation during coronary bypass surgery: a randomized, prospective study. Anesth Analg 2007; 104: 51-8.
Murkin JM, Arango M. Near-infrared spectroscopy as an index of brain and tissue oxygenation. Br J Anaesth 2009; 103(Suppl 1): i3-13.
Ghosal S, Trivedi J, Chen J, et al. Regional cerebral oxygen saturation level predicts 30-day mortality rate after left ventricular assist device surgery. J Cardiothorac Vasc Anesth 2018; 32: 1185-90.
Lee JH, Jang YE, Song IK, et al. Near-infrared spectroscopy and vascular occlusion test for predicting clinical outcome in pediatric cardiac patients: a prospective observational study. Pediatr Crit Care Med 2018; 19: 32-9.
Murkin JM. Cerebral oximetry: monitoring the brain as the index organ. Anesthesiology 2011; 114: 12-3.
Sanchez-de-Toledo J, Chrysostomou C, Munoz R, et al. Cerebral regional oxygen saturation and serum neuromarkers for the prediction of adverse neurologic outcome in pediatric cardiac surgery. Neurocrit Care 2014; 21: 133-9.
Slater JP, Guarino T, Stack J, et al. Cerebral oxygen desaturation predicts cognitive decline and longer hospital stay after cardiac surgery. Ann Thorac Surg 2009; 87: 36-45.
Kussman BD, Wypij D, Laussen PC, et al. Relationship of intraoperative cerebral oxygen saturation to neurodevelopmental outcome and brain magnetic resonance imaging at 1 year of age in infants undergoing biventricular repair. Circulation 2010; 122: 245-54.
Olbrecht VA, Skowno J, Marchesini V, et al. An international, multicenter, observational study of cerebral oxygenation during infant and neonatal anesthesia. Anesthesiology 2018; 128: 85-96.
Pellicer A, Valverde E, Elorza MD, et al. Cardiovascular support for low birth weight infants and cerebral hemodynamics: a randomized, blinded, clinical trial. Pediatrics 2005; 115: 1501-12.
Razlevice I, Rugyte DC, Strumylaite L, Macas A. Assessment of risk factors for cerebral oxygen desaturation during neonatal and infant general anesthesia: an observational, prospective study. BMC Anesthesiol 2016; DOI: https://doi.org/10.1186/s12871-016-0274-2.
Grocott HP. Commentary: Optimizing cerebral oxygenation in cardiac surgery: neurocognitive and perioperative outcomes. J Thorac Cardiovasc Surg 2020; 159: 956-7.
de la Matta M, Dominguez A. Prediction of bilateral cerebral oxygen desaturations from a single sensor in adult cardiac surgery: a prospective observational study. Eur J Anaesthesiol 2018; 35: 365-71.
Grocott HP. Cerebral oximetry monitoring. To guide physiology, avert catastrophe or both? Eur J Anaesthesiol 2019; 36: 82-3.
de la Matta M, Dominguez A. Reply to: Cerebral oximetry monitoring. To guide physiology, avert catastrophe or both? Eur J Anaesthesiol 2019; DOI: https://doi.org/10.1097/eja.0000000000000896.
Evered L, Silbert B, Knopman DS, et al. Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery-2018. Can J Anesth 2018; 65: 1248-57.
Lewis ME. Stepwise versus hierarchical regression: pros and cons. Southwest Educational Research Association 2007 Annual Meeting, San Antonio, USA. Available from URL: https://www.researchgate.net/publication/235464734_Stepwise_versus_hierarchical_regression_Pros_and_cons (accessed April 2020).
Flom P. Stopping stepwise: why stepwise selection is bad and what you should use instead. Towards Data Science 2018. Available from URL: https://towardsdatascience.com/stopping-stepwise-why-stepwise-selection-is-bad-and-what-you-should-use-instead-90818b3f52df. (accessed April 2020).
Flechet M, Guiza F, Vlasselaers D, et al. Near-infrared cerebral oximetry to predict outcome after pediatric cardiac surgery: a prospective observational study. Pediatr Crit Care Med 2018; 19: 433-41.
Hansen JH, Schlangen J, Armbrust S, et al. Monitoring of regional tissue oxygenation with near-infrared spectroscopy during the early postoperative course after superior cavopulmonary anastomosis. Eur J Cardiothorac Surg 2013; 43: 37-43.
Spaeder MC, Klugman D, Skurow-Todd K, et al. Perioperative near-infrared spectroscopy monitoring in neonates with congenital heart disease: relationship of cerebral tissue oxygenation index variability with neurodevelopmental outcome. Pediatr Crit Care Med 2017; 18: 213-8.
Ghanayem NS, Hoffman GM. Near infrared spectroscopy as a hemodynamic monitor in critical illness. Pediatr Crit Care Med 2016; 17: 201-6.
Vida VL, Tessari C, Cristante A, et al. The role of regional oxygen saturation using near-infrared spectroscopy and blood lactate levels as early predictors of outcome after pediatric cardiac surgery. Can J Cardiol 2016; 32: 970-7.
Davie SN, Grocott HP. Impact of extracranial contamination on regional cerebral oxygen saturation: a comparison of three cerebral oximetry technologies. Anesthesiology 2012; 116: 834-40.
Disclosures
Thomas W.L. Scheeren received research grants and honoraria from Edwards Lifesciences (Irvine, CA, USA) and Masimo Inc. (Irvine, CA, USA) for consulting and lecturing, and from Pulsion Medical Systems SE (Feldkirchen, Germany) for lecturing in the past. Thomas W.L. Scheeren is an editor of the Journal of Clinical Monitoring and Computing and is an associate editor for Anesthesia & Analgesia. All other authors report no conflicts of interest.
Funding statement
Departmental funds only.
Editorial responsibility
This submission was handled by Dr. Philip M. Jones, Associate Editor, Canadian Journal of Anesthesia.
Author information
Authors and Affiliations
Contributions
Marco Modestini helped in designing the study, analyzing and interpreting the data, and drafting and revising the manuscript. Lisa Hoffmann helped in collecting, analyzing, and interpreting the data and drafting and revising the manuscript. Caren Niezen helped in drafting and revising the manuscript. Benedetta Armocida helped in collecting data, and drafting and revising the manuscript. Jaap Jan Vos helped in analyzing and interpreting the data, and drafting and revising the manuscript. Thomas W.L. Scheeren helped in designing the study, analyzing and interpreting the data, and drafting and revising the manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Modestini, M., Hoffmann, L., Niezen, C. et al. Cerebral oxygenation during pediatric congenital cardiac surgery and its association with outcome: a retrospective observational study. Can J Anesth/J Can Anesth 67, 1170–1181 (2020). https://doi.org/10.1007/s12630-020-01733-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-020-01733-1