Abstract
Purpose
Application of adhesive tape to the eyelids during general anesthesia decreases the risk of corneal abrasion but may increase the risk of eyelid injury. The aim of this study was to determine if there is a difference in eyelid erythema when covering the eyelid with either Tegaderm™ or an EyeGard®.
Methods
We conducted a prospective, randomized, double-blind, split-face study of patients undergoing general anesthesia at an urban tertiary care academic medical centre. Each patient was randomized to having one eyelid covered with Tegaderm and the other with EyeGard. Photographs were taken prior to extubation and evaluated by three dermatologists. The primary outcome was the incidence of postoperative eyelid erythema. Secondary outcomes included the incidence of corneal abrasion and patient satisfaction.
Results
A total of 151 patients were included in our final analysis. Erythema was present on 117 (77%) eyelids covered with Tegaderm and 105 (70%) eyelids covered with EyeGard (% difference, 8; 95% confidence interval, 2 to 14; P = 0.03). No corneal abrasions were reported. The median [interquartile range] patient satisfaction score with eyelid condition was similar with Tegaderm vs EyeGard (5 [5–5] vs 5 [5–5], respectively; P = 0.84).
Conclusion
We found a small increase in postoperative eyelid erythema when using Tegaderm compared with EyeGard. While EyeGard could decrease the risk of eyelid erythema, this should be balanced against other potential benefits of Tegaderm such as protection from fluids leaking onto the cornea.
Trial registration
www.ClinicalTrials.gov (NCT03549429); registered 8 June, 2018.
Résumé
Objectif
L’application de ruban adhésif sur les paupières pendant l’anesthésie générale réduit le risque d’abrasion cornéenne mais pourrait augmenter le risque de lésion aux paupières. L’objectif de cette étude était de déterminer s’il existe une différence dans l’incidence d’érythème de la paupière lorsque l’on couvre la paupière à l’aide d’un Tegaderm™ ou d’un EyeGard®.
Méthode
Nous avons réalisé une étude prospective, randomisée, à double insu et à visage divisé auprès de patients subissant une anesthésie générale dans un centre médical universitaire urbain de soins tertiaires. Chaque patient a été randomisé à avoir une paupière couverte de Tegaderm et l’autre d’un EyeGard. Des photos ont été prises avant l’extubation et évaluées par trois dermatologistes. Le critère d’évaluation principal était l’incidence d’érythème postopératoire de la paupière. Les critères secondaires comprenaient l’incidence d’abrasion cornéenne et la satisfaction des patients.
Résultats
Au total, 151 patients ont été inclus dans notre analyse finale. Il y avait présence d’érythème sur 117 (77 %) paupières couvertes de Tegaderm et 105 (70 %) paupières couvertes d’un EyeGard (% de différence, 8; intervalle de confiance 95 %, 2 à 14; P = 0,03). Aucune abrasion cornéenne n’a été rapportée. Le score médian [écart interquartile] de satisfaction des patients en ce qui touchait à l’état de leurs paupières était semblable pour le Tegaderm et le EyeGard (5 [5–5] vs 5 [5–5], respectivement; P = 0,84).
Conclusion
Nous avons observé une légère augmentation du nombre d’érythèmes postopératoires de la paupière lors de l’utilisation du Tegaderm comparativement au EyeGard. Alors que le EyeGard pourrait réduire le risque d’érythème de la paupière, il convient de sous-peser ses avantages par rapport aux autres avantages potentiels du Tegaderm tels que la protection contre les liquides coulant sur la cornée.
Enregistrement de l’étude
www.ClinicalTrials.gov (NCT03549429); enregistrée le 8 juin 2018.
Similar content being viewed by others
Covering closed eyelids with adhesive tape is one of the most common approaches to keeping the eyelids shut during general anesthesia.1 While this practice decreases the risk of corneal abrasions and lagophthalmos (incomplete closure of the eyelids),2 recent reports have revealed that various types of eyelid injury can occur when the adhesive tape is removed.3,4,5 One case report describes an erythematous and edematous reaction upon removal of Tegaderm™ (3M healthcare, Maplewood, MN, USA) after a general anesthetic, which the authors attributed to a reaction with benzoyl peroxide that had been used as a topical acne wash.6 Eyelid injuries after anesthesia may require treatment and could contribute to a poor patient experience. It is likely that the incidence of eyelid injury after general anesthesia is underreported, but it is important to understand how the use of adhesive tape affects the risk of dermatological injury.
Currently, there is no standard practice or guidelines for choosing which adhesive tape to cover the eyelids with. In our institution, Durapore, Micropore, Tegaderm (all 3M healthcare, Maplewood, MN, USA), and the EyeGard® (Sharn Anesthesia, Tampa, FL, USA) are all used based primarily on provider preference and prior experience. While these products decrease the risk of corneal abrasion, it is possible that the risk of eyelid injury may differ among them. While no studies have addressed adhesive tape-related eyelid injuries among these four adhesive tapes, a randomized trial has evaluated eyelid injury of Kind Removal Silicone Tape (3M healthcare, Maplewood, MN, USA) vs Micropore.7 Those authors reported less eyelid injury with the silicone-based adhesive tape compared with the acrylic-based adhesive tape, but the results may be biased because the anesthesiologist who performed the evaluations was not blinded to group assignment.
As Tegaderm and EyeGard are the two tapes most commonly used in our institution, our objective was to compare the risk of perioperative eyelid injury between them. Although epidermal erosion of the eyelid would be the most direct form of injury to measure, it is relatively uncommon, and as such, we chose to study eyelid erythema, which represents a more common precursor to epidural erosion.8 As secondary outcomes, we measured patient satisfaction and the incidence of corneal abrasion. Given local anecdotal experience, our hypothesis was that there would be a difference in postoperative eyelid erythema between the two tapes. In addition, we performed hypothesis-generating exploratory analyses to identify potential risk factors for erythema.
Methods
We conducted a prospective, randomized, double-blind, split-face (where the face is divided into two experimental halves and randomly assigned different treatments) study of patients undergoing general anesthesia at an urban tertiary care academic medical centre. This study was approved by the Tufts Health Sciences Institutional Review Board (#12884; May 1, 2018) and was registered at clinicaltrials.gov (NCT03549429; May 25, 2018). The study was also listed on the medical centre’s “Clinical Research Recruitment Website” freely accessible to all patients (https://www.tuftsmedicalcenter.org/ClinicalTrials/Quality-Improvement-12884). Written informed consent was obtained from all study participants. This manuscript adheres to the Equator guidelines by the Consolidated Standards of Reporting Trials 2010.9
All patients 18 yr or older undergoing general anesthesia were eligible for enrolment. Exclusion criteria included any pre-existing eyelid abnormalities (including erythema, trauma, or piercings), surgery in the prone position or on the head or neck, and tape sensitivity or allergy. On the day of surgery, the research assistant approached potential subjects, explained the risks and benefits of participating in the study, and enrolled those who consented.
An online randomization tool (research randomizer version 4.0; www.randomizer.org) was used to randomly assign the eyelids of each study subject. Based on the randomization schema, one eyelid was covered with Tegaderm, which is composed of a sterile transparent urethane polymer membrane and an adhesive acrylic polymer, while the other eyelid was covered with EyeGard, which is composed of a non-sterile perforated transparent propylene polymer membrane and an adhesive acrylic polymer. Study subjects were blinded to the treatment allocation, which was determined by opening sequentially numbered, sealed, opaque envelopes containing the randomization assignment.
Upon entry into the operating room, a baseline photograph of the eyelids was taken. After induction of anesthesia (but prior to intubation), the Tegaderm and EyeGard tapes were applied to the eyelids by one research assistant in a standardized fashion. Both Tegaderm and EyeGard were applied to the eyelids immediately after removal from their respective backing, (i.e., they were never applied first to any other surface, such as a blanket to decrease adhesiveness). At the end of surgery but prior to extubation, the tapes were removed by the same research assistant in a standardized fashion and a photograph of the eyelids was taken, such that the outcomes assessed by the dermatologist represented an immediate postoperative measurement. All photographs were taken using an iPad mini 4 (Apple Inc., Cupertino, CA, USA) at a distance of 6 inches from the lens of the iSight camera to the eyelids.
The primary outcome was the number of patients who had postoperative eyelid erythema upon removal of the adhesive tapes. Secondary outcomes included the number of patients who had corneal abrasion and the patient satisfaction scores.
Three dermatologists, blinded to the randomization assignment, independently evaluated the eyelids for erythema by examining cropped postoperative photographs showing the periorbital region of each study subject. Baseline photographs (taken immediately before tape application) were used as a reference to differentiate baseline abnormalities from new ones, such that only if an abnormality was new would it be considered present in the final evaluation of the postoperative photograph. Erythema was evaluated on a scale of 0–3 (0 indicating no erythema, 1 indicating mild erythema, 2 indicating moderate erythema, and 3 indicating severe erythema), which has previously shown internal consistency, reliability, and sensitivity to changes in severity of the disease process when assessed by a trained dermatologist.10,11 After completing the evaluations, the erythema scale was converted into a dichotomous variable such that a score of 0 was considered absent erythema and a score of 1, 2, or 3 was considered present erythema. The results were combined after completing the evaluations. Evaluations that were concordant (i.e., the responses of the three dermatologists were the same for a particular outcome) were counted as such. For evaluations that were discordant among the three dermatologists, the majority (two out of three dermatologists) was counted.
In the recovery room, corneal abrasion was assessed if the patient complained of foreign body sensation, and patient satisfaction was assessed using a survey evaluating three questions on a five-point Likert scale developed based on a prior study12 (see eAppendix, available as Electronic Supplementary Material).
Statistical analysis
A previous study7 found that the proportion of eyelids with erythema was 50% when using acrylic adhesive tape (Medipore) and 33% when using silicone adhesive tape (Kind Removal Silicone Tape). To detect a difference in eyelid erythema in our study with an alpha of 0.05 and a power of 80% using the two-tailed McNemar’s test for matched pairs of subjects, a total of 227 eyelids would be needed. To account for possible attrition, we enrolled 159 patients (318 eyelids). G*Power13 version 3.1.9.3 was used to perform the sample size calculation.
Demographic characteristics of the cohort were summarized using descriptive statistics. The McNemar test was used as a test of correlated proportions to assess dermatological evaluations of the eyelids as both treatments were given to each individual subject.14 Patient satisfaction was assessed using the Wilcoxon rank-sum test. For exploratory analyses (and thus un-adjusted for multiple comparisons), using results from prior studies15,16,17 the data were stratified based on sex (male vs female), age (≤70 vs ≥71 yr old),18 skin type (non-Caucasian vs Caucasian), and duration of tape application (≥ two hours vs < two hours). The McNemar test was used to calculate the % difference and 95% confidence intervals (CI) of the proportion of eyelids with erythema. A P < 0.05 was considered statistically significant. All statistical analyses were performed using MedCalc (MedCalc Software, Ostend, Belgium).
Results
A total of 159 study subjects consented to participate and were randomized, of which 151 completed the study and were included in the final statistical analysis. Eight patients were excluded from the final analysis for the following reasons: protocol violation (n = 5), surgery cancelled (n = 1), withdrew from study (n = 1), and research assistant unavailable (n = 1). An enrolment chart is presented in Fig. 1. The first patient was recruited on June 1, 2018 and the primary completion date for recruitment was July 23, 2018. Demographic characteristics of the cohort are presented in Table 1.
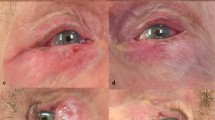
Table 2 shows the erythema grades over eyelids covered with Tegaderm and EyeGard. Erythema was present on 117 (77%) eyelids covered with Tegaderm and 105 (70%) eyelids covered with EyeGard (% difference, 8; 95% CI, 2 to 14; P = 0.03). Figure 2 shows characteristic erythema with Tegaderm but not with EyeGard, and Fig. 3 shows typical application of Tegaderm and EyeGard over the eyelids. No corneal abrasions were reported in either group. The median [interquartile range (IQR)] patient satisfaction score with eyelid condition was similar with Tegaderm vs EyeGard (5 [5–5] vs 5 [5–5], respectively; P = 0.84).
Exploratory analyses
Table 3 presents the number of patients who had postoperative eyelid erythema with Tegaderm compared with EyeGard stratified according to the exploratory outcomes. There was a statistically significant difference in erythema rates between Tegaderm and EyeGard in female patients (% difference, 10; 95% CI, 2 to 18; P = 0.04), non-Caucasian skin type (% difference, 25; 95% CI, 8 to 42; P = 0.03), duration of tape application equal to or longer than two hours (% difference, 13; 95% CI, 3 to 22; P = 0.02), and age ≤ 70 yr (% difference, 9; 95% CI, 1 to 16; P = 0.04).
Discussion
In this prospective, randomized, double-blind, split-face study, fewer patients had postoperative eyelid erythema with EyeGard compared with Tegaderm. Nevertheless, the majority of patients had erythema with both tapes and the % difference in erythema between the two eye tapes was relatively small. This study found no differences in corneal abrasions or patient satisfaction scores between EyeGard and Tegaderm tape use. When performing exploratory analyses by stratifying patients with postoperative eyelid erythema based on various patient characteristics, female sex, younger age, non-Caucasian skin type, and longer duration of tape application were associated with a greater difference in the percentage of eyelids with erythema.
There are several mechanisms that could explain the increased number of patients with postoperative eyelid erythema with Tegaderm compared with EyeGard. The application of an occlusive barrier such as Tegaderm or EyeGard over the epidermis decreases evaporation and increases the level of skin moisture.19 Greater skin moisture increases the skin pH, which affects the skin’s structural properties, including integrity and cohesion of the epidermis, leading to inflammation that may be manifested as erythema.20,21 The perforated membrane of the EyeGard likely allows for moisture to more freely evaporate compared with the continuous film of the Tegaderm, reducing moisture and erythema.
Our findings may also be explained by the acrylic in the adhesive. The adhesive in medical tape is composed of an acrylic polymer that is inert, but trace amounts of the acrylic monomer (which is responsible for most cutaneous reactions) may still be present.22,23,24 Therefore, the inflammatory response observed upon removal of adhesive tape could be secondary to an acrylic-mediated injury, possibly due to dysfunction of epidermal integrin.25 Importantly, integrin plays an essential role in maintaining the adhesion forces of the epidermis.26 While Tegaderm and EyeGard are both coated with acrylic adhesive, the perforations in the transparent membrane of the EyeGard likely reduce the surface area contact between the epidermis and the adhesive more than Tegaderm does, thereby reducing the inflammatory response.
The finding of an increased number of patients with postoperative eyelid erythema with Tegaderm compared with EyeGard should be interpreted cautiously. Even if one assumes that less erythema suggests a lower risk of eyelid injury with EyeGard, one cannot conclude that EyeGard should always be used preferentially over Tegaderm. While the perforated design of the EyeGard might decrease postoperative eyelid erythema, those perforations could also allow fluids to leak through the tape and into the cornea. Therefore, if fluids leaking through the perforations is a concern, a more occlusive membrane (such as Tegaderm) may be favourable. Still, while the continuous film design of Tegaderm may function as a better barrier to fluids, if the tape is not adequately applied, liquid can still leak through the edges.27,28,29 Therefore, the decision and application of adhesive tape for eye protection during general anesthesia requires careful thought and consideration.
While our study was not designed to examine patient-related risk factors for erythema, our exploratory analyses suggest that female sex, younger age, non-Caucasian skin type, and longer duration of tape application could be risk factors. These findings may be explained by moisture-related changes in pH that occur when adhesive tape is applied to the epidermis. Several studies have found subtle differences in skin pH based on sex and race, including lower pH of women and non-Caucasian skin types.15,16,17 Furthermore, longer duration of tape application likely increases moisture and changes skin pH. Nevertheless, the assessment of erythema in non-Caucasian skin types is challenging and can be misinterpreted, especially in those with highly pigmented skin.30,31 For this reason, our exploratory findings should be interpreted only as hypothesis-generating, and future studies should further investigate if these risk factors indeed increase the risk of erythema and epidermal injury.
Our study has important limitations. First, the scale we used to evaluate erythema was originally validated in patients with eczema, so its applicability in our study is not entirely known. Similarly, because we used photographs to perform the evaluations, the dermatologists could not make a physical assessment of the skin (e.g., assessment of blanchable vs non-blanchable erythema was not possible). This limits the extent of the evaluations. Nevertheless, using photographs was advantageous because it allowed for board-certified dermatologists to perform the assessments, greatly increasing reliability. Furthermore, photographic evaluation of erythema has been used in dermatology validation studies31 and dermatologists commonly use photographs to guide clinical decision-making.32 Second, it is possible that the dermatologists could have guessed which eyelid was covered with which tape despite the randomization because of the different sizes. Nevertheless, during those independently performed assessments, we did not give the dermatologists any clues, so it would be impossible for the dermatologists to know for sure if their guess was correct. Third, many patients had a significant amount of generalized flushing at the end of surgery presumably due to the vasodilatory effects of anesthetics and laying flat for an extended period of time. This flushing could have confounded the interpretation of erythema from the photographs. Nevertheless, given that in the split-face study design33 each patient was exposed to both adhesive tapes, the influence of facial flushing on the interpretation of erythema would be equal. Similarly, because providers used forced-air warming devices as needed, it is unknown if the heat had any effect on the adhesive properties of the tapes and resultant erythema. Nevertheless, any differences that might have been caused would be the same for each tape given the split-face study design. Fourth, while we did not find a difference in patient satisfaction, it is likely that our study design did not adequately address this question as the scale had not been previously validated. Furthermore, because we measured patient satisfaction in the recovery room, it is likely that eyelid irritation was not a priority for the patients at the time. Finally, while no corneal abrasions were reported, our study is underpowered to detect such a difference given the rarity of this outcome.
In summary, a small increase in the number of patients with postoperative eyelid erythema with Tegaderm compared with EyeGard was observed using a prospective, randomized, double-blind, split-face study design. Nevertheless, the clinical significance is not entirely clear because the difference was small and the association between erythema and more serious dermatological injury is not well understood. Adhesive tapes are often used throughout the healthcare industry as a dressing for wounds, for securing catheters, and in many other cases. The several options available may leave providers uncertain as to which tape to use in which circumstances, and very few studies have been performed to understand the risks and benefits of different types of tape. Our study suggests that EyeGard may be favourable when the goal is to decrease the risk of eyelid erythema with general anesthesia, but this conclusion should be approached with caution as the perforations in EyeGard may not protect from chemical splash-related injury. Future studies are needed to better understand the dermatological reactions of other tapes commonly used so that providers can make more informed decisions.
Change history
05 March 2020
In the article entitled: “The effect of Tegaderm™ versus EyeGard® on eyelid erythema during general anesthesia: a randomized-controlled trial” published online and in the May 2020 print issue of the Journal, Can J Anesth 2020; DOI: 10.1007/s12630-020-01588-6, the trial registration date in the abstract should read 25 May, 2018 (instead of 8 June, 2018).
References
Grixti A, Sadri M, Watts MT. Corneal protection during general anesthesia for nonocular surgery. Ocul Surf 2013; 11: 109-18.
Batra YK, Bali IM. Corneal abrasions during general anesthesia. Anesth Analg 1977; 56: 363-5.
Shamim R, Riaz R, Patro A, Priya V. Use of surgical tapes for eye protection: an eye opener. Anesth Essays Res 2016; 10: 687-9.
Drzymalski DM, Arkoff H, Azocar RJ. Eyelid injury after use of 3M Durapore™ tape during general anesthesia. Can J Anesth 2018; 65: 1273-4.
Tobias JD, Jagannathan N, Sawardekar A, Bhalla T. An unusual cause of post-operative orbital edema in a child after general anesthesia. Saudi J Anaesth 2011; 5: 105-7.
O’Connor JJ Jr, Vasudevan A. Tega-dermabrasion. Anesthesiology 2007. https://doi.org/10.1097/01.anes.0000278863.84694.34.
Zeng LA, Lie SA, Chong SY. Comparison of medical adhesive tapes in patients at risk of facial skin trauma under anesthesia. Anesthesiol Res Pract 2016. https://doi.org/10.1155/2016/4878246.
Smith SM, Zirwas MJ. Nonallergic reactions to medical tapes. Dermatitis 2015; 26: 38-43.
Schulz KF, Altman DG, Moher D; CONSORT Group. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMJ 2010; https://doi.org/10.1136/bmj.c332.
Barbier N, Paul C, Luger T, et al. Validation of the Eczema Area and Severity Index for atopic dermatitis in a cohort of 1550 patients from the pimecrolimus cream 1% randomized controlled clinical trials programme. Br J Dermatol 2004; 150: 96-102.
Hanifin JM, Thurston M, Omoto M, Cherill R, Tofte SJ, Graeber M. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol 2001; 10: 11-8.
Drzymalski DM, Lumbreras-Marquez MI, Tsen LC, Camann WR, Farber MK. The effect of patient-selected or preselected music on anxiety during cesarean delivery: a randomized controlled trial. J Matern Fetal Neonatal Med 2019. https://doi.org/10.1080/14767058.2019.1594766.
Faul F, Erdfelder E, Buchner A, Lang AG. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods 2009; 41: 1149-60.
Sainani K. The importance of accounting for correlated observations. PM R 2010; 2: 858-61.
Ehlers C, Ivens UI, Moller ML, Senderovitz T, Serup J. Females have lower skin surface pH than men. A study on the surface of gender, forearm site variation, right/left difference and time of the day on the skin surface pH. Skin Res Technol 2001; 7: 90-4.
Wesley NO, Maibach HI. Racial (ethnic) differences in skin properties: the objective data. Am J Clin Dermatol 2003; 4: 843-60.
Choi EH. Gender, age, and ethnicity as factors that can influence skin pH. Curr Probl Dermatol 2018; 54: 48-53.
Kaya G, Saurat JH. Dermatoporosis: a chronic cutaneous insufficiency/fragility syndrome. Clinicopathological features, mechanisms, prevention and potential treatments. Dermatology 2007; 215: 284-94.
Hartmann AA. Effect of occlusion on resident flora, skin-moisture and skin-pH. Arch Dermatol Res 1983; 275: 251-4.
Schmid-Wendtner MH, Korting HC. The pH of the skin surface and its impact on the barrier function. Skin Pharmacol Physiol 2006; 19: 296-302.
Plasencia I, Norlen L, Bagatolli LA. Direct visualization of lipid domains in human skin stratum corneum’s lipid membranes: effect of pH and temperature. Biophys J 2007; 93: 3142-55.
Austin AT, Basker RM. The level of residual monomer in acrylic denture base materials with particular reference to a modified method of analysis. Br Dent J 1980; 149: 281-6.
Pfeiffer P, Rosenbauer EU. Residual methyl methacrylate monomer, water sorption, and water solubility of hypoallergenic denture base materials. J Prosthet Dent 2004; 92: 72-8.
Sasseville D. Acrylates in contact dermatitis. Dermatitis 2012; 23: 6-16.
Hoshiba T, Yoshihiro A, Tanaka M. Evaluation of initial cell adhesion on poly (2-methoxyethyl acrylate) (PMEA) analogous polymers. J Biomater Sci Polym Ed 2017; 28: 986-99.
Watt FM. Role of integrins in regulating epidermal adhesion, growth and differentiation. EMBO J 2002; 21: 3919-26.
Reimer C. In reply: Eye taping and chlorhexidine exposure: caution when interpreting scarce evidence. Can J Anesth 2018; 65: 323-4.
Shive M. Eye taping and chlorhexidine exposure: caution when interpreting scarce evidence. Can J Anesth 2018; 65: 322.
Reimer C. Eye taping during anesthesia and chlorhexidine exposure. Can J Anesth 2017; 64: 1159-60.
Ben-Gashir MA, Hay RJ. Reliance on erythema scores may mask severe atopic dermatitis in black children compared with their white counterparts. Br J Dermatol 2002; 147: 920-5.
Zhao CY, Wijayanti A, Doria MC, et al. The reliability and validity of outcome measures for atopic dermatitis in patients with pigmented skin: a grey area. Int J Womens Dermatol 2015; 1: 150-4.
Buabbas AJ, Sharma P, Al-Abdulrazaq A, Shehab H. Smartphone use by government dermatology practitioners in Kuwait: a self-reported questionnaire based cross-sectional study. BMC Med Inform Decis Mak 2019; 19: DOI: https://doi.org/10.1186/s12911-019-0883-z.
Zhu H, Zhang S, Ahn C. Sample size considerations for split-mouth design. Stat Methods Med Res 2017; 26: 2543-51.
Author contributions
Dan M. Drzymalski designed the study, performed the randomization, interpreted the data, performed the statistical analysis, and wrote the final draft of the manuscript. Katelyn Ward designed the study, recruited the patients, and wrote the first draft of the manuscript. Josephine M. Hernandez helped design the study and revise the manuscript. Joyce Hoot designed the study and performed the dermatological evaluations of all photographs. Shiu-Chung Au performed the dermatological evaluations of all photographs. Fei-Shiuann Clarissa Yang designed the study and performed the dermatological evaluations of all photographs. Ruben J. Azocar supported the study and revised the manuscript. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Acknowledgements
We would like to thank Saul Weingart, M.D., Ph.D., Chief Medical Officer, Department of Medicine, Tufts Medical Center, Boston, Massachusetts, USA, and Dr. Sadeq Quraishi, M.D., Department of Anesthesiology and Perioperative Medicine, Tufts Medical Center, Boston, Massachusetts, USA, for offering advice on this manuscript.
Conflicts of interest
The authors disclose that there are no commercial or non-commercial affiliations that may be perceived to be a conflict of interest with the work, and there are no other associations in relation to this work.
Funding statement
The trial was funded by the 2018 Harold Williams, M.D. Medical Student Research Fellowship, awarded by the Tufts University School of Medicine Summer Research Committee.
Editorial responsibility
This submission was handled by Dr. Hilary P. Grocott, Editor-in-Chief, Canadian Journal of Anesthesia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Drzymalski, D.M., Ward, K., Hernandez, J.M. et al. The effect of Tegaderm™ versus EyeGard® on eyelid erythema during general anesthesia: a randomized-controlled trial. Can J Anesth/J Can Anesth 67, 560–567 (2020). https://doi.org/10.1007/s12630-020-01588-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-020-01588-6