Abstract
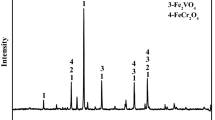
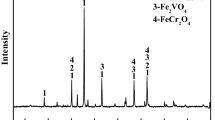
As part of a research project to develop a novel clean smelting process for the comprehensive utilization of Hongge vanadium titanomagnetite (HVTM), in this study, the effect of Cr2O3 addition on the oxidation induration mechanism of HVTM pellets (HVTMPs) was investigated in detail. The results showed that the compressive strength of the HVTMPs was greatly weakened by the Cr2O3 addition, mainly because of a substantial increase in the porosity of the HVTMPs. The Cr2O3 addition marginally affected the phase composition but greatly affected the microstructural changes of the HVTMPs. Increased amounts of Cr2O3 resulted in a decrease in the uniform distribution of the hematite grains and in an increase in the Fe–Cr solid solutions (Fe1.2Cr0.8O3 and Fe0.7Cr1.3O3) embedded in the hematite grains. Moreover, the compact hematite was destroyed by forming a dispersed structure and the hematite recrystallization was hindered during the oxidation induration, which adversely affected the compressive strength. On the basis of these results, a schematic was formulated to describe the oxidation induration mechanism with different amounts of added Cr2O3. This study provides theoretical and technical foundations for the effective production of HVTMPs and a reference for chromium-bearing minerals.
Similar content being viewed by others
References
X.W. Lv, Z.G. Lun, J.Q. Yin, and C.G. Bai, Carbothermic reduction of vanadium titanomagnetite by microwave irradiation and smelting behavior, ISIJ Int., 53(2013), No. 7, p. 1115.
Z.H. Wang, G.F. Li, Y.S. Sun, and M.Z. He, Reduction behavior of hematite in the presence of coke, Int. J. Miner. Metall. Mater., 23(2016), No. 11, p. 1244.
S. Samal, B.K. Mohapatra, P.S. Mukherjee, and S.K. Chatterjee, Integrated XRD, EPMA and XRF study of ilmenite and titania slag used in pigment production, J. Alloys Compd., 474(2009), No. 1-2, p. 484.
J.H. Luo, Z.Y. Wu, E.H. Wu, J.H. Li, X.J. Liao, R. Tang, and S.L. Yang, Studies on mineralogical characteristics of iron concentrates in Hongge vanadium titano-magnetite, Iron Steel Vanadium Titanium, 36(2015), No. 2, p. 73.
S.T. Yang, M. Zhou, T. Jiang, S.F. Guan, W.J. Zhang, and X.X. Xue, Application of a water cooling treatment and its effect on coal-based reduction of high-chromium vanadium and titanium iron ore, Int. J. Miner. Metall. Mater., 23(2016), No. 12, p. 1353.
M. Zhou, T. Jiang, S.T. Yang, and X.X. Xue, Sintering behaviors and consolidation mechanism of high-chromium vanadium and titanium magnetite fines, Int. J. Miner. Metall. Mater., 22(2015), No. 9, p. 917.
T. Hu, X.W. Lv, and C.G. Bai, Enhanced reduction of coal-containing titanomagnetite concentrates briquette with multiple layers in rotary hearth furnace, Steel Res. Int., 87(2016), No. 4, p. 494.
E.H. Wu, R. Zhu, S. L. Yang, L. Ma, J. Li, and J. Hou, Influences of technological parameters on smelting-separation process for metallized pellets of vanadium-bearing titanomagnetite concentrates, J. Iron Steel Res. Int., 23(2016), No. 7, p. 655.
S. Samanta, M.C. Goswami, T.K. Baidya, S. Mukherjee, and R. Dey, Mineralogy and carbothermal reduction behaviour of vanadium-bearing titaniferous magnetite ore in Eastern India, Int. J. Miner. Metall. Mater., 20(2013), No. 10, p. 917.
M.Y. Wang, S.F. Zhou, X.W. Wang, B.F. Chen, H.X. Yang, S.K. Wang, and P.F. Luo, Recovery of iron from chromium vanadium-bearing titanomagnetite concentrate by direct reduction, JOM, 68(2016), No. 10, p. 2698.
C. Geng, T.C. Sun, Y.W. Ma, C.Y. Xu, and H.F. Yang, Effects of embedding direct reduction followed by magnetic separation on recovering titanium and iron of beach titanomagnetite concentrate, J. Iron Steel Res. Int., 24(2017), No. 2, p. 156.
T. Hu, X.W. Lv, C.G. Bai, Z.G. Lun, and G.B. Qiu, Reduction behavior of Panzhihua titanomagnetite concentrates with coal, Metall. Mater. Trans. B, 44(2013), No. 2, p. 252.
L.S. Zhao, L.N. Wang, D.S. Chen, H.X. Zhao, Y.H. Liu, and T. Qi, Behaviors of vanadium and chromium in coal-based direct reduction of high-chromium vanadium-bearing titanomagnetite concentrates followed by magnetic separation, Trans. Nonferrous Met. Soc. China, 25(2015), No. 4, p. 1325.
Y.Y. Zhu, L.Y. Yi, W. Zhao, D.S. Chen, H.X. Zhao, and T. Qi, Leaching of vanadium, sodium, and silicon from molten V-Ti-bearing slag obtained from low-grade vanadium-bearing titanomagnetite, Int. J. Miner. Metall. Mater., 23(2016), No. 8, p. 898.
Y.R. Liu, J.L. Zhang, Z.J. Liu, and X.D. Xing, Phase transformation behavior of titanium during carbothermic reduction of titanomagnetite ironsand, Int. J. Miner. Metall. Mater., 23(2016), No. 7, p. 760.
H.Y. Sun, J.S. Wang, Y.H. Han, X.F. She, and Q.G. Xue, Reduction mechanism of titanomagnetite concentrate by hydrogen, Int. J. Miner. Process., 125 (2013), p. 122.
W. Li, G.Q. Fu, M.S. Chu, and M.Y. Zhu, Gas-based direct reduction of Hongge vanadium titanomagnetite-oxidized pellet and melting separation of the reduced pellet, Steel Res. Int., 88(2017), No. 1, art No. 1600120.
Z.C. Huang, L.Y. Yi, and T. Jiang, Mechanisms of strength decrease in the initial reduction of iron ore oxide pellets, Powder Technol., 221(2012), p. 284.
K.V.S. Sastry, P. Dontula, and C. Hosten, Investigation of the layering mechanism of agglomerate growth during drum pelletization, Powder Technol., 130(2003), No. 1-3, p. 231.
F. Pan, Q.S. Zhu, Z. Du, and H.Y. Sun, Oxidation kinetics, structural changes and element migration during oxidation process of vanadium-titanium magnetite ore, J. Iron Steel Res. Int., 23 (2016), No. 11, p. 1160.
W. Li, G.Q. Fu, M.S. Chu, and M.Y. Zhu, Oxidation induration process and kinetics of Hongge vanadium titanium-bearing magnetite pellets, Ironmaking Steelmaking, 44(2017), No. 4, p. 294.
J.B. Zhang, G.Y. Zhang, Q.S. Zhu, C. Lei, Z.H. Xie, and H.Z. Li, Morphological changes and reduction mechanism for the weak reduction of the preoxidized Panzhihua ilmenite, Metall. Mater. Trans. B, 45(2014), No. 3, p. 914.
W. Li, N. Wang, G.Q. Fu, M.S. Chu, and M.Y. Zhu, Influence of roasting characteristics on gas-based direct reduction behavior of Hongge vanadium titanomagnetite pellet with simulated shaft furnace gases, Powder Technol., 310(2017), p. 343.
D.Q. Zhu, C.C. Yang, J. Pan, and X.B. Li, Comparison of the oxidation behaviors of high FeO chromite and magnetite concentrates relevant to the induration of ferrous pellets, Metall. Mater. Trans. B, 47(2016), No. 5, p. 2919.
J.L. Zhang, Z.Y. Wang, X.D. Xing, and Z.J. Liu, Effect of aluminum oxide on the compressive strength of pellet, Int. J. Miner. Metall. Mater., 21(2014), No. 4, p. 339.
S. Suresh, Fatigue of Materials, Cambridge University Press, New York, 1998.
L.J. Gibson and M.F. Ashby, Cellular Solids Structure and Properties, Cambridge University Press, Cambridge, 1997.
Y. Bao and Z. Jin, Size effects and a mean-strength criterion for ceramics, Fatigue Fract. Eng. Mater. Struct., 16(1993), No. 8, p. 829.
F.M. Shen, Q.J. Gao, X. Jiang, G. Wei, and H.Y. Zheng, Effect of magnesia on the compressive strength of pellets, Int. J. Miner. Metall. Mater., 21(2014), No. 5, p. 431.
Acknowledgements
This work is financially supported by National Natural Science Foundation of China (No. 51574067).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W., Wang, N., Fu, Gq. et al. Effect of Cr2O3 addition on the oxidation induration mechanism of Hongge vanadium titanomagnetite pellets. Int J Miner Metall Mater 25, 391–398 (2018). https://doi.org/10.1007/s12613-018-1583-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-018-1583-x