Abstract
Objectives
Examine the effects of a 24-week exercise intervention against a social intervention on body weight, body mass index (BMI) and nutritional status in PWD living in nursing homes.
Design
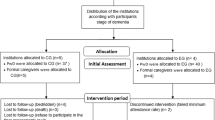
Randomized controlled trial.
Participants
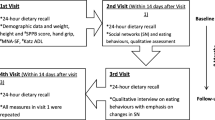
Ninety-one older people with dementia living in nursing homes. Interventions: Exercise (n=44) or social-based activities (n=47), taking place twice per week, for 60 minutes/session, during 24 weeks.
Measurements
Nutritional status was measured with the mini-nutritional assessment (MNA), weight and BMI.
Results
After the 24-week intervention, none of MNA (B-coeff. 1.28; 95% CI -2.55 to 0.02), weight (-0.06; -1.58 to 1.45) and BMI (-0.05; -0.85 to 0.74) differed significantly between groups after adjustment for multiplicity. In the social group, MNA significantly improved while it remained stable in the exercise group. The percentage of at-risk and malnourished patients reduced in both groups by more than 6%.
Conclusion
The results suggest that social activities have as good effects as exercise activities on nutritional status in PWD nursing home residents.



Similar content being viewed by others
References
Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. The American journal of clinical nutrition 2010;91 (4):1123S–1127S
Doherty TJ. Invited review: Aging and sarcopenia. J Appl Physiol (1985) 2003;95 (4):1717–1727. doi:10.1152/japplphysiol.00347.2003
Koopmans RT, van der Sterren KJ, Van der Steen JT. The ‘natural’endpoint of dementia: death from cachexia or dehydration following palliative care? International journal of geriatric psychiatry 2007;22 (4):350–355
Sergi G, De Rui M, Coin A, Inelmen EM, Manzato E. Weight loss and Alzheimer’s disease: temporal and aetiologic connections. Proceedings of the Nutrition Society 2013;72 (1):160–165
López AP, i Figuls MR, Cuchi GU, Berenstein EG, Pasies BA, Alegre MB, Herdman M. Systematic review of megestrol acetate in the treatment of anorexia-cachexia syndrome. Journal of pain and symptom management 2004;27 (4):360–369
Morley JE, Thomas DR. Cachexia: new advances in the management of wasting diseases. Journal of the American Medical Directors Association 2008;9 (4):205–210
Doucet E, Imbeault P, Alméras N, Tremblay A. Physical Activity and Low-Fat Diet: Is it Enough to Maintain Weight Stability in the Reduced-Obese Individual Following Weight Loss by Drug Therapy and Energy Restriction? Obesity 1999;7 (4):323–333
Genton L, Karsegard VL, Chevalley T, Kossovsky MP, Darmon P, Pichard C. Body composition changes over 9 years in healthy elderly subjects and impact of physical activity. Clinical Nutrition 2011;30 (4):436–442. doi:http://dx.doi.org/10.1016/j.clnu.2011.01.009
Crocker T, Young J, Forster A, Brown L, Ozer S, Greenwood DC. The effect of physical rehabilitation on activities of daily living in older residents of long-term care facilities: systematic review with meta-analysis. Age and ageing 2013;42 (6):682–688
Rolland Y, Pillard F, Klapouszczak A, Reynish E, Thomas D, Andrieu S, Rivière D, Vellas B. Exercise program for nursing home residents with Alzheimer’s disease: A 1-year randomized, controlled trial. Journal of the American Geriatrics Society 2007;55 (2):158–165
Vreugdenhil A, Cannell J, Davies A, Razay G. A community-based exercise programme to improve functional ability in people with Alzheimer’s disease: a randomized controlled trial. Scand J Caring Sci 2012;26 (1):12–19. doi:10.1111/j.1471-6712.2011.00895.x
Conradsson M, Littbrand H, Lindelof N, Gustafson Y, Rosendahl E. Effects of a high-intensity functional exercise programme on depressive symptoms and psychological well-being among older people living in residential care facilities: A cluster-randomized controlled trial. Aging Ment Health 2010;14 (5):565–576. doi:10.1080/13607860903483078
de Souto Barreto P, Denormandie P, Lepage B, Armaingaud D, Rapp T, Chauvin P, Vellas B, Rolland Y. Effects of a long-term exercise programme on functional ability in people with dementia living in nursing homes: Research protocol of the LEDEN study, a cluster randomised controlled trial. Contemporary clinical trials 2016;47:289–295
de Souto Barreto P, Cesari M, Denormandie P, Armaingaud D, Vellas B, Rolland Y. Exercise or Social Intervention for Nursing Home Residents with Dementia: A Pilot Randomized, Controlled Trial. J Am Geriatr Soc 2017. doi:10.1111/jgs.14947
Guigoz Y, Jensen G, Thomas D, Vellas B. The mini nutritional assessment (mna®) review of the literature-what does it tell us?/discussion. The journal of nutrition, health & aging 2006;10 (6):466
Vellas B, Guigoz Y, Garry PJ, Nourhashemi F, Bennahum D, Lauque S, Albarede J-L The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999;15 (2):116–122
Guigoz Y. The Mini Nutritional Assessment (MNA (R)) review of the literature -What does it tell us? J Nutr Health Aging 2006;10 (6):466–485
Organization WH. Obesity: preventing and managing the global epidemic. vol 894. World Health Organization, 2000
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the royal statistical society Series B (Methodological) 1995:289–300
Allen VJ, Methven L, Gosney MA. Use of nutritional complete supplements in older adults with dementia: systematic review and meta-analysis of clinical outcomes. Clinical nutrition 2013;32 (6):950–957
Ikeda M, Brown J, Holland AJ, Fukuhara R, Hodges J. Changes in appetite, food preference, and eating habits in frontotemporal dementia and Alzheimer’s disease. Journal of Neurology, Neurosurgery & Psychiatry 2002;73 (4):371–376
Pedersen BK, Saltin B. Exercise as medicine–evidence for prescribing exercise as therapy in 26 different chronic diseases. Scandinavian journal of medicine & science in sports 2015;25 (S3):1–72
Forbes D, Thiessen EJ, Blake CM, Forbes SC, Forbes S. Exercise programs for people with dementia. Cochrane Database Syst Rev 2013;12:0
Poehlman ET, Dvorak RV. Energy expenditure, energy intake, and weight loss in Alzheimer disease. The American journal of clinical nutrition 2000;71 (2):650s–655s
de Souto Barreto P, Morley JE, Chodzko-Zajko W, Pitkala KH, Weening-Djiksterhuis E, Rodriguez-Mañas L, Barbagallo M, Rosendahl E, Sinclair A, Landi F. Recommendations on physical activity and exercise for older adults living in longterm care facilities: A taskforce report. Journal of the American Medical Directors Association 2016;17 (5):381–392
Schnelle JF, MacRae PG, Giacobassi K, MacRae HS, Simmons SF, Ouslander JG. Exercise with physically restrained nursing home residents: maximizing benefits of restraint reduction. Journal of the American Geriatrics Society 1996;44 (5):507–512
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Maltais, M., Rolland, Y., Haÿ, PE. et al. The Effect of Exercise and Social Activity Interventions on Nutritional Status in Older Adults with Dementia Living in Nursing Homes: A Randomised Controlled Trial. J Nutr Health Aging 22, 824–828 (2018). https://doi.org/10.1007/s12603-018-1025-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-018-1025-5