Abstract
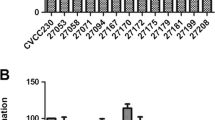
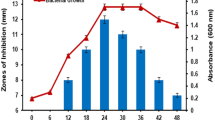
Multidrug resistance (MDR) is a serious health threat throughout the world resulting in reduced efficacy of antibacterial, antiparasitic, antiviral, and antifungal drugs. One of the most promising concepts that may represent a good alternative to antibiotics can be the use of bacteriocins obtained from lactic acid bacteria. The L. rhamnosus BTK 20-12 strain was isolated from traditional Armenian naturally fermented salted cheese. The probiotic potential of the strain was approved. It was shown that strain produced at less two bacteriocins (BCN 1 and BCN 2) with different molecular weight (1427 Da and 602.6 Da, respectively). Bacteriocins inhibited the growth of multidrug-resistant bacteria of different etiologies and belong to different taxonomic groups with diverse efficiency and it depends on properties of bacteriocins, as well as from isolation sources of pathogens. Thus, bacteriocins of L. rhamnosus BTK 20-12 have protein-like nature and a broad range of activity and are excellent candidates for the development of new prophylactic and therapeutic substances to complement or replace conventional antibiotics.




Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and available from the corresponding author on reasonable request.
References
Joshi VK (2015) Indigenous fermented foods of South Asia. Fermented foods and beverages series. CRC, Boca Raton ISBN 9781439887837 - CAT# K14059
Daliri EB-M, Lee BH (2015) New perspectives on probiotics in health and disease. Food Sci Hum Wellness 4:56–65
Hertzberger R, Arents J, Dekker HL, Pridmore RD, Gysler C, Kleerebezem M, de Mattos MJT (2014) H2O2 production in species of the Lactobacillus acidophilus group: a central role for a novel NADH-dependent flavin reductase. Appl Environ Microbiol 80(7):2229–2239. https://doi.org/10.1128/AEM.04272-13
Shenderov BA (2013) Metabiotics: novel idea or natural development of probiotic conception. Microb Ecol Health Dis 24(20399):1. https://doi.org/10.3402/mehd.v24i0.20399
Gillor O, Etzion A, Riley MA (2008) The dual role of bacteriocins as anti- and probiotics. Appl Microbiol Biotechnol 81(4):591–606
Dimitrijević R, Stojanović M, Zivković I, Petersen A, Jankov RM, Dimitrijević L, Gavrović-Jankulović M (2009) The identification of a low molecular mass bacteriocin rhamnosin A, produced by Lactobacillus rhamnosus strain 68. J Appl Microbiol 107(6):2108–2115
Falagas M, Betsi G, Athanasiou S (2006) Probiotics for prevention of recurrent vulvovaginal candidiasis: a review. J Antimicrob Chemother 58(2):266–272
Reid G, Charbonneau D, Erb J, Kochanowski B, Beuerman D, Poehner R, Bruce AW (2003) Oral use of Lactobacillus rhamnosus GR-1 and L.fermentum RC-14 significantly alters vaginal flora: randomized, placebo-controlled trial in 64 healthy women. FEMS Immunol Med Microbiol 35(2):131–134
Weese SJ, Anderson ME (2002) Preliminary evaluation of Lactobacillus rhamnosus strain GG, a potential probiotic in dogs. Can Vet J 43(10):771–774
Ammoscato F, Scirocco A, Altomare A, Matarrese P, Petitta C, Ascione B, Caronna R, Guarino M, Marignani M, Cicala M, Chirletti P, Malorni W, Severi C (2013) Lactobacillus rhamnosus protects human colonic muscle from pathogen lipopolysaccharide-induced damage. Neurogastroenterol Motil 25(12):984–e777
Aguilar-Uscanga BR, Solís-Pacheco JR, Plascencia L, Aguilar-Uscanga MG, García HS, Lacroix M (2013) Effect of culture medium on bacteriocin production by Lactobacillus rhamnosus HN001 and Lactobacillus reuteri ATCC 53608. J Microbiol Biotechnol Food Sci 2(6):2462–2468
Todorov SD, Dicks LMT (2005) Growth parameters influencing the production of Lactobacillus rhamnosus bacteriocins ST461BZ and ST462BZ. Ann Microbiol 55(4):283–289
World Health Organization (WHO) (2012) World health statistics. [Electronic Book] France: WHO Press; [cited 17 July 2012]; Available from: http://www.who.int/healthinfo/EN_WHS.Full.pdf
D’Andrea MM, Literacka E, Zioga A, Giani T, Baraniak A, Fiett J, Sadowy E, Tassios PT, Rossolini GM, Gniadkowski M, Miriagou V (2011) Evolution and spread of a multidrug-resistant Proteus mirabilis clone with chromosomal AmpC-type cephalosporinases in Europe. Antimicrob Agents Chemother 55(6):2735–2742
Wright MH, Adelskov J, Greene AC (2017) Bacterial DNA extraction using individual enzymes and phenol/chloroform separation. J Microbiol Biol Educ 18(2):1–3
Bürgmann H, Pesaro M, Widmer F, Zeyer J (2001) A strategy for optimizing quality and quantity of DNA extracted from soil. J Microbiol Methods 45(1):7–20
Pavlicek A et al (1999) Fre-tree-freeware program for construction of phylogenetic trees on the basis of distance data and bootstrap\jackknife analysis of tree robustness. Application in the RAPD analysis of genus Frenkelia. Folia Biol (Praha) 45(3):97–99
Aghajanyan AE, Tkhruni FN, Hovhannisyan GZ, Yeghiyan KI, Vardanyan AA, Saghyan AS (2015) Isolation and purification of bioinhibitory product from culture liquid of lactic acid bacteria Lactobacillus acidophilus 1991 VKPM 6257 and Lactobacillus rhamnosus INMIA 9614. J Biotechnol 3:32–40
Aslam M, Shahid M, Rehman FU, Naveed NH, Batool AI, Sharif S, Asia A (2011) Purification and characterization of bacteriocin, isolated from Streptococcus thermophilus. Afr J Microbiol Res 5(18):2642–2648
Parente E, Brienza C, Moles M, Ricciardi A (1995) A comparison of methods for the measurement of bacteriocin activity. J Microbiol Methods 22(1):95–108
Todorov SD, Leblanc JG, Franco BD (2012) Evaluation of the probiotic potential and effect of encapsulation on survival for Lactobacillus plantarum ST16Pa isolated from papaya. World J Microbiol Biotechnol 28(3):973–984
Linares DM, Gómez C, Renes E, Fresno JM, Tornadijo ME, Ross RP, Stanton C (2017) Lactic acid bacteria and bifidobacteria with potential to design natural biofunctional health-promoting dairy foods. Front Microbiol 8:846. https://doi.org/10.3389/fmicb.2017.00846
Henriksson A, Szewzyk R, Conway P (1991) Characteristics of the adhesive determinants of Lactobacillus fermentum 104. Appl Environ Microbiol 57(2):499–502
Sirota TV (2013) Use of nitro blue tetrazolium in the reaction of adrenaline autooxidation for the determination of superoxide dismutase activity. Biomed Khim 59(4):399–410 (Article in Russian)
Fernandes P, Lopez P, Corbi A, Pelaez C, Requena T (2008) Probiotic strains: survival under simulated gastrointestinal conditions, in vitro adhesion to Caco-2 cells and effect of cytokine secretion. Eur Food Res Technol 227(5):1475–1484
Gueimonde M, Sánchez B, de los Reyes-Gavilán CG, Margolles A (2013) Antibiotic resistance in probiotic bacteria. Front Microbiol 4:202. https://doi.org/10.3389/fmicb.2013.00202
Klaenhammer TR, Muriana PM (1991) Purification and partial characterization of lactacin F, a bacteriocin produced by Lactobacillus acidophilus 11088. Appl Environ Microbiol 57(1):114–121
Sarika AR, Lipton AP, Aishwarya MS (2010) Bacteriocin production by a new isolate of Lactobacillus rhamnosus GP1 under different culture conditions. Adv J Food Sci Technol 2(5):291–297
Tkhruni FN (2016) Efficiency of metabiotics from lactic acid bacteria against pathogens. Int J Curr Res Acad Rev 3:1–11
Israyelyan A, Karapetyan K, Tkhruni F, Arstamyan L, Balabekyan T (2015) Sensitivity of different pathogens to biological antimicrobial agents. Eur J Biol Life Sci 3:61–66
Karapetyan K, Tkhruni F, Israyelyan A, Yermolenko E, Verdyan A (2017) Comparative characterization of endemic lactic acid bacteria of Enterococcus Genus. Int J Sci Technol Res 6(7):357–361
Tkhruni FN, Saghyan AS, Balabekyan TsR, Karapetyan KJ, Khachatryan TV, Aghajanyan AE (2015) Lactobacillus rhamnosus BТК 20-12 bacteria strain, producing Rhamn 20-12 BCN1 and Rhamn 20-12 BCN2 bacteriocins RA Patent: № 2924 А. Available at https://armpat@aipa.am
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tkhruni, F.N., Aghajanyan, A.E., Balabekyan, T.R. et al. Characteristic of Bacteriocins of Lactobacillus rhamnosus BTK 20-12 Potential Probiotic Strain. Probiotics & Antimicro. Prot. 12, 716–724 (2020). https://doi.org/10.1007/s12602-019-09569-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-019-09569-y