Abstract
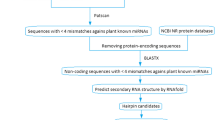
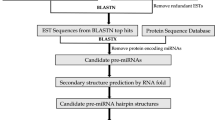
MicroRNAs (miRNAs) are a large family of 19–25 nucleotides, regulatory, non-coding RNA molecules that control gene expression by cleaving or inhibiting the translation of target gene transcripts in animals and plants. Despite the important functions of miRNAs related to regulation of plant growth and development processes, metabolism, and abiotic and biotic stresses, little is known about the disease-related miRNA. Here, we present a new pipeline for miRNA analysis using expressed sequence tags (ESTs)-based bioinformatics approach in Kinnow mandarin, a commercially important citrus fruit crop. For this, 56,041 raw EST sequences of Citrus reticulata Blanco were retrieved from EST database in NCBI through step-by-step filtering and processing methods and 130 miRNAs were predicted. Upon blast with Citrus sinensis transcriptome data, these produced potential targets related to disease resistance proteins, pectin lyase-like superfamily proteins, lateral organ boundaries (LOB) domain-containing proteins 11, and protein phosphatase 2C family proteins, protein kinases, dehydrogenases, and methyltransferases. Majority of the predicted miRNAs were of 22, 23, and 24 nucleotides in length. To validate these computationally predicted miRNA, poly(A)-tailed Reverse Transcription-PCR was applied to detect the expression of seven miRNA which showed disease-related potential targets, in citrus greening diseased leaf tissues in comparison to the healthy tissues of Kinnow mandarin. Our study provides information on regulatory roles of these potential miRNAs for the citrus greening disease development, miRNA targets, and would be helpful for future research of miRNA function in citrus.




Similar content being viewed by others
References
Panda D, Dehury B, Sahu J, Barooah M, Sen P, Modi MK (2014) Computational identification and characterization of conserved miRNAs and their target genes in garlic (Allium sativum L.) expressed sequence tags. Gene 537:333–342
Mohanpuria P, Yadav SK (2012) Characterization of novel small RNAs from tea (Camellia sinensis L.). Mol Biol Rep 39:3977–3986
Navarro L, Dunoyer P, Jay F, Arnold B, Dharmasiri N, Estelle M, Voinnet O, Jones JD (2006) A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 312:436–439
Quintero A, Pérez-Quintero AL, López C (2013) Identification of ta-siRNAs and cis-nat-siRNAs in cassava and their roles in response to cassava bacterial blight. Gen Proteom Bioinform 11:172–181
Niu QW, Lin SS, Reyes JL, Chen KC, Wu HW, Yeh SD, Chua NH (2006) Expression of artificial microRNAs in transgenic Arabidopsis thaliana confers virus resistance. Nat Biotechnol 24:1420–1428
Zuo J, Wang Y, Liu H, Ma Y, Ju Z, Zhai B, Fu D, Zhu Y, Luo Y, Zhu B (2011) MicroRNAs in tomato plants. Sci China Life Sci 54:599–605
Pinweha N, Asvarak T, Viboonjun U, Narangajavana J (2015) Involvement of miR160/miR393 and their targets in cassava responses to anthracnose disease. J Plant Physiol 174:26–35
Sharma S, Singh B, Rani G, Virk GS, Zaidi AA, Nagpal A (2007) In vitro response of encapsulated and non-encapsulated somatic embryos of Kinnow mandarin (Citrus nobilis Lour × C. deliciosa Tenora). Plant Biotechnol Rep 1:101–107
Moxon S, Jing R, Szittya G, Schwach F, Pilcher RLR, Moulton V, Dalmay T (2008) Deep sequencing of tomato short RNAs identifies microRNAs targeting genes involved in fruit ripening. Genome Res 18:1602–1609
Falgueras J, Lara AJ, Fernández-Pozo N, Cantón FR, Pérez-Trabado G, Claros MG (2010) SeqTrim: a high-throughput pipeline for pre-processing any type of sequence read. BMC Bioinform 11:38
Smit AF (1996) The origin of interspersed repeats in the human genome. Curr Opin Genet Dev 6:743–748
Wang JF, Zhou H, Chen YQ, Luo QJ, Qu LH (2004) Identification of 20 microRNAs from Oryza sativa. Nucleic Acids Res 32:1688–1695
Jones-Rhoades MW, Bartel DP, Bartel B (2006) MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol 57:19–53
Zuker M (2003) Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acid Res 31:3406–3415
Omer A, Singh S, Duhan N (2012) Identification of miRNAs in C. roseus and their potential targets. Bioinformation 8:75–80
Fu H-J, Zhu J, Yang M, Zhang Z-Y, Tie Y, Jiang H, Sun Z-X, Zheng X-F (2006) A novel method to monitor the expression of microRNAs. Mol Biotechnol 32:197–204
Goto K, Kanazawa A, Kusaba M, Masuta C (2003) A simple and rapid method to detect plant siRNAs using nonradioactive probes. Plant Mol Biol Rep 21:51–58
Singh K, Raizada J, Bhardwaj P, Ghawana S, Rani A, Singh H, Kaul K, Kumar S (2004) 26S rRNA-based internal control gene primer pair for reverse transcription-polymerase chain reaction based quantitative expression studies in diverse plant species. Anal Biochem 335:330–333
Guddeti S, Zhang DC, Li AL, Leseberg CH, Kang H, Li XG, Zhai WX, Johns MA, Mao L (2005) Molecular evolution of the rice miR395 gene family. Cell Res 15:631–638
Xie FL, Huang SQ, Guo K, Xiang AL, Zhu YY, Nie L, Yang ZM (2007) Computational identification of novel microRNAs and targets in Brassica napus. FEBS Lett 581:1464–1474
Zhang BH, Pan XP, Cox SB, Cobb GP, Anderson TA (2006) Evidence that miRNAs are different from other RNAs. Cell Mol Life Sci 63:246–254
Fuchs S, Grill E, Meskiene I, Schweighofer A (2013) Type 2C protein phosphatases in plants. FEBS J 280:681–693
Walton JD (1994) Deconstructing the cell wall. Plant Physiol 104:1113–1118
Collmer A, Keen NT (1986) The role of pectic enzymes in plant pathogenesis. Ann Rev Phytopath 24:383–409
Linhardt RJ, Galliher PM, Cooney CL (1986) Polysaccharide lyases. Appl Biochem Biotechnol 12:135–176
Beaulieu C, Boccara M, Gijsegem FV (1993) Pathogenic behavior of pectinase-defective Erwinia Chrysanthemi mutants on different Plants. Mol Plant Microbe Interact 6:197–202
Crawford MS, Kolatlukudy PE (1987) Pectate lyase from Fusarium solani f. sp. pisi: purification, characterization, in vitro translation of the mRNA, and involvement in pathogenicity. Arch Biochem Biophys 258:196–205
Thatcher LF, Powell JJ, Aitken EA, Kazan K, Manners JM (2012) The lateral organ boundaries domain transcription factor LBD20 functions in Fusarium wilt Susceptibility and jasmonate signaling in Arabidopsis. Plant Physiol 160:407–418
Lorang JM, Sweat TA, Wolpert TJ (2007) Plant disease susceptibility conferred by a ‘‘resistance’’ gene. PNAS 104:14861–14866
Ooijen VG, Mayr G, Kasiem MM, Albrecht M, Cornelissen BJ, Takken FL (2008) Structure-function analysis of the NB-ARC domain of plant disease resistance proteins. J Exp Bot 59:1383–1397
Axtell MJ, Bartel DP (2005) Antiquity of microRNAs and their targets in land plants. Plant Cell 17:1658–1673
Jin H (2008) Endogenous small RNAs and antibacterial immunity in plants. FEBS Lett 582:2679–2684
Katiyar-Agarwal S, Jin HL (2010) Role of small RNAs in host–microbe interactions. Ann Rev Phytopathol 48:225–246
Zhang X, Zhao H, Gao S, Wang WC, Katiyar-Agarwal S, Huang HD, Raikhel N, Jin H (2011) Arabidopsis Argonaute 2 regulates innate immunity via miRNA393(*)-mediated silencing of a Golgi-localized SNARE gene, MEMB12. Mol Cell 42:356–366
Jones-Rhoades MW (2012) Conservation and divergence in plant microRNAs. Plant Mol Biol 80:3–16
Sun G (2012) MicroRNAs and their diverse functions in plants. Plant Mol Biol 80:17–36
Sunkar R (2010) MicroRNAs with macro-effects on plant stress responses. Semin Cell Dev Biol 21:805–811
Albrecht U, Bowman KD (2008) Gene expression in Citrus sinensis (L.) Osbeck following infection with the bacterial pathogen Candidatus Liberibacter asiaticus causing Huanglongbing in Florida. Plant Sci 175:291–306
Albrecht U, Bowman KD (2012) Transcriptional response of susceptible and tolerant citrus to infection with Candidatus Liberibacter asiaticus. Plant Sci 185–186:118–130
Zhao H, Sun R, Albrecht U, Padmanabhan C, Wang A, Coffey MD, Girke T, Wang Z, Close TJ, Roose M, Yokomi RK, Folimonova S, Vidalakis G, Rouse R, Bowmane KD, Jina H (2013) Small RNA profiling reveals phosphorus deficiency as a contributing factor in symptom expression for citrus Huanglongbing disease. Mol Plant 6:301–310
Kumar R (2014) Role of microRNAs in biotic and abiotic stress responses in crop plants. Appl Biochem Biotechnol 174:93–115
Acknowledgements
Authors are thankful to Dr. (Mrs.) Parveen Chunneja to provide bioinformatics facility funded by the Department of Biotechnology, Government of India to carry out this work. We would like to thank Dr. G.S. Sidhu for providing citrus greening samples for this study. The work grant was provided by the internal funding of School of Agricultural Biotechnology, PAU Ludhiana.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohanpuria, P., Duhan, N., Sarao, N.K. et al. In Silico Identification and Validation of Potential microRNAs in Kinnow Mandarin (Citrus reticulata Blanco). Interdiscip Sci Comput Life Sci 10, 762–770 (2018). https://doi.org/10.1007/s12539-017-0235-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-017-0235-6