Abstract
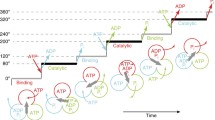
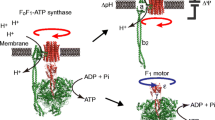
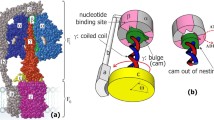
In this paper, based on the structure of the ATPase and the master equation, we put forward a stochastic hopping model which describes the rotary four-state motor’s dynamics action. Finally, we obtained the relationship of the angle velocity ω, diffusion coefficient and the ATP’s concentration, and got a result which accords with the rotary motor’s biological mechanism.
Similar content being viewed by others
References
Antes, I., Chandler, D., Wang, H., Oster, G. 2003. The unbinding of ATP from F1 ATPase. Biophys J 85, 695–706.
Gao, Y.Q., Yang, W., Karplus, M. 2005. A Structure-Based Model for Theory the Synthesis and Hydrolysis of ATP by F1-ATPase. Cell 123, 195–205.
Kagawa, R., Montgomery, M.G., Braig, K., Leslie, A.G., Walker, J.E. 2004. The structure of bovine F1-ATPase inhibited by ADP and beryllium fluoride. EMBO J 23, 2734–2744.
Kinosita K.J., Yasuda, R., Noji, H., Adachi, K. 2000. A rotary molecular motor that can work at near 100% efficiency. Phil Trans R Soc Lond B 55, 473–489.
Ma, J., Flynn, T., Cui, Q., Leslie, A., Walker, J., Karplus, M. 2002. A Dynamic Analysis of the Rotation Mechanism in F1-ATPase. Structure 10, 921–931.
Muneyuki, E., Noji, H., Amano, T. 2000. F0F1-ATP synthase: general structural features of ATP-engine and a problem on free energy transduction. Biochimica et Biophysica Acta 1458, 467–481.
Oster, G., Wang, H.Y. 2000. Reverse engineering a protein: the mechanochemistry of ATP synthase. Biochim Biophys Acta 1458, 482–510.
Oster, G., Wang, H.Y. 1999. ATP synthase: two motors, two fuels. Structure 7, R67–R72.
Oster, G., Wang, H.Y. 2000. Why is the mechanical efficiency of F1-ATPase so high? Journal of Bioenergetics and Biomembranes 32, 459–469.
Paul, D.B. 1998. ATP synthase — past and future. Biochimica et Biophysica Acta 1365, 3–9.
Rubinstein, J.L., Walker, J.E. 2002. ATP synthase from Saccharomyces cerevisiae: Location of the OSCP Subunit in the Peripheral Stalk Region. Journal of Molecular Biology 321, 613–619.
Wu, W.X., Zhan, Y., Zhao, T.J., Guan, R.H., Mei, J.P. 2003. Directed Motion of a molecular motor based on the four-state model with unequal substeps commun. Theor Phys 40, 9–14.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, Y., Yang, MJ., Wu, WX. et al. Research of the ATPase’s rotation catalysis stochastic transition dynamics. Interdiscip Sci Comput Life Sci 1, 229–234 (2009). https://doi.org/10.1007/s12539-009-0038-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-009-0038-5