Abstract
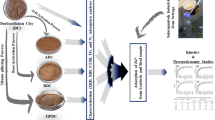
Homogenized samples of raw clays resulting from two (2) different lots of natural clays from Maghnia (Algeria) have been assessed for their potential use in the removal of Pb2+ and Zn2+ ions from industrial liquid wastes (LW). Raw and acid-activated samples have been characterized by powder X-ray diffraction, FT-IR spectroscopy, electron microscopy (SEM), and X-ray fluorescence (XRF) and used as adsorbents for the removal of Pb2+ and Zn2+ ions from aqueous system using adsorption method under different conditions. The effect of factors including contact time, pH, and dosage on the adsorption properties of Pb2+ and Zn2+ ions onto clays was investigated at 25 °C. The obtained results revealed that the removal percentages of Pb2+ and Zn2+ ions, from both aqueous solution (AS) and LW, were varying between 90 and 98% for 40 min and optimal pH values ranged from 5 to 6 for Pb2+ and Zn2+ ions, respectively. The kinetics of both Pb2+ and Zn2+ ion adsorption fitted well with the pseudo-second-order model. Langmuir, Freundlich, and Temkin adsorption isotherms were used, and their constants were evaluated. The values of thermodynamic parameters, ΔH°, ΔS°, and ΔG° indicated that the adsorption of Pb2+ and Zn2+ ions was spontaneous and exothermic process in nature. The adsorption and desorption isotherms indicated that Pb2+ and Zn2+ adsorption to raw clays was reversible. The experimental results obtained showed that the raw clays from Maghnia (Algeria) had a great potential for removing Pb2+ and Zn2+ ions from industrial liquid wastes using adsorption method.


















Similar content being viewed by others
References
Ait Sidhoum D, Socías-Viciana MM, Ureña-Amate MD, Derdour A, González-Pradas E, Debbagh-Boutarbouch N (2013) Removal of paraquat from water by an Algerian bentonite. Appl Clay Sci 83–84:441–448
Al-Degs YS, El-Barghouthi MI, Issa AA, Khraisheh MA, Walker GM (2006) Sorption of Zn(II), Pb(II), and Co(II) using natural sorbents: equilibrium and kinetic studies. Water Res 40(14):2645–2658. https://doi.org/10.1016/j.watres.2006.05.018
Assameur H, Boufatit M (2012) Contribution to the removal study of Co2+ ions by acid-activated clay from Maghnia (Algeria): equilibrium and kinetic studies. Desalin Water Treat 45(1-3):315–323. https://doi.org/10.1080/19443994.2012.692023
Ayari F, Srasra E, Trabelsi-Ayadi M (2007) Retention of lead from an aqueous solution by use of bentonite as adsorbent for reducing leaching from industrial effluents. Desalination 206(1-3):270–278. https://doi.org/10.1016/j.desal.2006.03.569
Barrett, E. P., Joyner, L. G., Halenda, P. F., 1951. The determination of pore volume and area distribution in porous substances. I. Computation of nitrogen isotherms. J Am Chem Soc. 73, 373–380
Bhattacharya AK, Mandal SN, Das SK (2006) Adsorption of Zn (II) from aqueous solution by using different adsorbents. Chem Eng J 123(1-2):43–51. https://doi.org/10.1016/j.cej.2006.06.012
Boufatit M, Ait-Amar H, McWhinnie WR (2007) Development of Algerian material montmorillonite clay. Adsorption of phenol, 2-dichlorophenol and 2, 4, 6-trichlorophenol from aqueous solutions onto montmorillonite exchanged with transition metal complexes. Desalination 206(1-3):394–406. https://doi.org/10.1016/j.desal.2006.04.060
Calvet R (1989) Adsorption of organic chemical in soils. Environ Health Perspect 83:145–177. https://doi.org/10.1289/ehp.8983145
Carrillo-Gonzalez R, Simunek J, Sauvé S, Domy A (2006) Mechanisms and pathways of trace element mobility in soils. Adv Agron 91:111–176. https://doi.org/10.1016/S0065-2113(06)91003-7
Chegrouche S, Mellah A, Barkat M (2009) Removal of strontium from aqueous solutions by adsorption onto activated carbon: kinetic and thermodynamic studies. Desalination 235(1-3):306–318. https://doi.org/10.1016/j.desal.2008.01.018
Dib S, Boufatit M (2009) Equilibrium study of adsorption of Pb2+ from aqueous solution onto Algerian bentonite clay. Desalin Water Treat 5(1-3):106–110. https://doi.org/10.5004/dwt.2009.570
Eloussaief M, Benzina M (2010) Efficiency of natural and acid-activated clays in the removal of Pb(II) from aqueous solutions. J Hazard Mater 178(1-3):753–757. https://doi.org/10.1016/j.jhazmat.2010.02.004
Eloussaief, M., Hamza, W., Kallel, N., Benzina, M. (2013). Wastewaters decontamination: mechanisms of Pb(II), Zn(II), and Cd(II) competitive adsorption on Tunisian smectite in single and multi-solute systems. Environ Prog Sustain Energy 32 (2), 229–238
Fan QH, Li Z, Zhao H, Jia Z, Xu J, Wu WS (2009) Adsorption of Pb (II) on palygorskite from aqueous solution: effects of pH, ionic strength and temperature. Appl Clay Sci 45(3):111–116. https://doi.org/10.1016/j.clay.2009.04.009
Fathi H, PariZangane A, Hamed F, Moradi H, Alidoost Saharkhiz Lahiji A (2014) Municipal solid waste characterization and its assessment for potential compost product: a case study in Zanjan city, Iran. Sky J Soil Sci Environ Manage 3(4):36–41
Giles CH, MacEwan TH, Nakhwa SN, Smith D (1960) Studies in adsorption: 11. A systematic classification of solution adsorption isotherms and its use in diagnosis of adsorption mechanisms and measurements of specific surface areas of solids. J Chem Soc 111:3973–3993
Giles CH, Smith D, Huitson A (1974a) A general treatment and classification of the solute. Adsorption isotherm. I—theoretical. J Colloid Interface Sci 47(3):755–765. https://doi.org/10.1016/0021-9797(74)90252-5
Giles CH, D’Silva AP, Easton IA (1974b) A general treatment and classification of the solute. Adsorption isotherm. Part II. Experimental interpretation. J Colloid Interface Sci 47(3):767–778
Hefne JA, Mekhemer WK, Alandis NM, Aldayel OA, Alajyan T (2008) Kinetic and thermodynamic study of the adsorption of Pb (II) from aqueous solution to the natural and treated bentonite. Int J Phys Sci 3(11):281–288
Ho YS (2005) Effect of pH on lead removal from water using tree fern as the sorbent. Bioresour Technol 96(11):1292–1296. https://doi.org/10.1016/j.biortech.2004.10.011
Ho YS, MacKay G (1999) Pseudo-second order model for sorption process. Process Biochem 34(5):451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Ho YS, MacKay G (1998) A comparison study of chemisorptions kinetic models applied to pollutant removal on various sorbents. TransIChemE 76(B):332–340
JO, RADP, Décret exécutif No. 06-141 (2006) 24 Rabie El Aouel 1427, correspondant au 23 avril 2006. Valeurs limites de rejets d’effluents liquides industriels
Larakeb M, Youcef L, Achour S (2017) Removal of zinc from water by adsorpion on bentonite and kaolin. Athens J Sci:47–57
Lasheras-Zubiate I, Navarro-Blasco M, Fernández JM, Álvarez JI (2012) Encapsulation, solid-phases identification and leaching of toxic metals in cement systems modified by natural biodegradable polymers. J Hazard Mater 233–234:7–17
Liu Y, WU P, Dang Z, YE D (2006) Heavy metal removal from water by adsorption using pillared montmorillonite. Acta Geol Sin 80(2):219–225
Malamis, S., Katsou, E., 2013. A review on zinc and nickel adsorption on natural and modified zeolite, bentonite and vermiculite: examination of process parameters, kinetics and isotherms. J Hazard Mater 252–253, 428–461, 252-253, DOI: https://doi.org/10.1016/j.jhazmat.2013.03.024
Mimanne G, Benhabib K, Benghalem A, Taleb S (2014) Etude de l’Adsorption des MétauxLourds (Pb et Cd) en solution Aqueuse sur Charbon Actif et montmorillonite Sodée de l’OuestAlgérien (study of the adsorption of heavy metals (Pb and Cd) in aqueous solution on activated carbon and sodium montmorillonite from western Algeria). J Mater Environ Sci 5(4):1298–1307
Mohammed-Azizi F, Dib S, Boufatit M (2013) Removal of heavy metals from aqueous solutions by Algerian bentonite. Desalin Water Treat 51(22–24):4447–4458. https://doi.org/10.1080/19443994.2013.770241
Nguyen TAH, Ngo HH, Guo WS, Zhang J, Liang S, Yue QY, Li Q, Nguyen TV (2013) Review. Applicability of agricultural waste and by-products for adsorptive removal of heavy metals from wastewater. Bioresour Technol 148:574–585. https://doi.org/10.1016/j.biortech.2013.08.124
Panda H, Mohanty CR (2014) Removal of heavy metals (Cr6+) from aqueous solution using industrial waste. Int J Emerg Technol Adv Eng 4(4):33–38
Poëy J, Philibert C (2000) Toxicité des métaux. Rev Fr Lab 323:35–43
Piri, S., Alikhani-Zanjani, Z., Piri, F., Zamani, A., Yaftian, M. and Davari, M., 2016. Potential of polyaniline modified clay nanocomposite as a selective decontamination adsorbent for Pb(II) ions from contaminated waters: kinetics and thermodynamic study. J Environ Health Sci Eng. 2-10
Quek SY, Wase DAJ, Forster CF (1998) The use of sago waste for the sorption of lead and copper. Water SA 24(3):251–256
Rai UN, Prasad D, Verma S, Upadhyay AK, Singh NK (2012) Biomonitoring of metals in Ganga water at different ghats of Haridwar: implications of constructed wetland for sewage detoxification. Bull Environ ContamToxicol 89(4):805–810. https://doi.org/10.1007/s00128-012-0766-9
Rodier, J., Legube, B., Merlet N., 2009. L’analyse de l’eau, 9ème Ed. Dunod, Paris, Partie D: Eaux résiduaires, 966-1007
Sari A, Tuzen M, Soylak M (2007) Adsorption of Pb(II) and Cr(III) from aqueous solution on Celtek clay. J Hazard Mater 114:41–46
Sdiri A, Higashi T, Jamoussi F, Bouaziz S (2012) Effects of impurities on the removal of heavy metals by natural limestones in aqueous systems. J Environ Manag 93(1):245–253. https://doi.org/10.1016/j.jenvman.2011.08.002
Sdiri A, Higashi T, Hattab T, Jamoussi F, Tasea N (2011) Evaluating the adsorptive capacity of montmorillonitic and calcareous clays on the removal of several heavy metals in aqueous systems. Chem Eng J 172(1):37–46. https://doi.org/10.1016/j.cej.2011.05.015
Sdiri A, Khairy M, Bouaziz, El-Safty S (2016) A natural clayey adsorbent for selective removal of lead from aqueous solutions. Appl Clay Sci 126:89–97. https://doi.org/10.1016/j.clay.2016.03.003
Xu D, Tan XL, Chen CL, Wang XK (2008) Adsorption of Pb(II) from aqueous solution to MX-80 bentonite: effect of pH, ionic strength, foreign ions and temperature. Appl Clay Sci 41(1-2):37–46. https://doi.org/10.1016/j.clay.2007.09.004
Acknowledgements
The samples of wastes examined in this study were kindly supplied by ENPEC and LRC-ONEDD. The authors are grateful to the ENPEC (Unité de Production des batteries à Oued Semmar, Alger) and LRC-ONEDD (Alger) for their helpful cooperation in the experimental tests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohammed-Azizi, F., Boufatit, M. Assessment of raw clays from Maghnia (Algeria) for their use in the removal of Pb2+ and Zn2+ ions from industrial liquid wastes: a case study of wastewater treatment. Arab J Geosci 11, 58 (2018). https://doi.org/10.1007/s12517-017-3360-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-017-3360-y