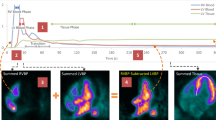
Abstract
Background
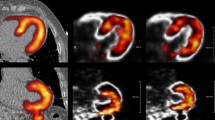
Patient motion can lead to misalignment of left ventricular volumes of interest and subsequently inaccurate quantification of myocardial blood flow (MBF) and flow reserve (MFR) from dynamic PET myocardial perfusion images. We aimed to identify the prevalence of patient motion in both blood and tissue phases and analyze the effects of this motion on MBF and MFR estimates.
Methods
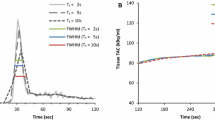
We selected 225 consecutive patients that underwent dynamic stress/rest rubidium-82 chloride (82Rb) PET imaging. Dynamic image series were iteratively reconstructed with 5- to 10-second frame durations over the first 2 minutes for the blood phase and 10 to 80 seconds for the tissue phase. Motion shifts were assessed by 3 physician readers from the dynamic series and analyzed for frequency, magnitude, time, and direction of motion. The effects of this motion isolated in time, direction, and magnitude on global and regional MBF and MFR estimates were evaluated. Flow estimates derived from the motion corrected images were used as the error references.
Results
Mild to moderate motion (5-15 mm) was most prominent in the blood phase in 63% and 44% of the stress and rest studies, respectively. This motion was observed with frequencies of 75% in the septal and inferior directions for stress and 44% in the septal direction for rest. Images with blood phase isolated motion had mean global MBF and MFR errors of 2%-5%. Isolating blood phase motion in the inferior direction resulted in mean MBF and MFR errors of 29%-44% in the RCA territory. Flow errors due to tissue phase isolated motion were within 1%.
Conclusions
Patient motion was most prevalent in the blood phase and MBF and MFR errors increased most substantially with motion in the inferior direction. Motion correction focused on these motions is needed to reduce MBF and MFR errors.





Similar content being viewed by others
Abbreviations
- LV:
-
Left ventricular
- MBF:
-
Myocardial blood flow
- MFR:
-
Myocardial flow reserve
- PET:
-
Positron emission tomography
- VOI:
-
Volume of interest
- TAC:
-
Time-activity curve
- LAD:
-
Left anterior descending
- LCX:
-
Left circumflex
- RCA:
-
Right coronary artery
References
Naya M, Murthy VL, Taqueti VR, Foster CR, Klein J, Garber M, et al. Preserved coronary flow reserve effectively excludes high-risk coronary artery disease on angiography. J Nucl Med 2014;55:248–55. https://doi.org/10.2967/jnumed.113.121442.
Ziadi MC, deKemp RA, Williams K, Guo A, Renaud JM, Chow BJW, et al. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multivessel coronary artery disease? J Nucl Cardiol 2012;19:670–80. https://doi.org/10.1007/s12350-011-9506-5.
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, Di Carli G, et al. Improved cardiac risk assessment with noninvasive measures of coronary flow reserve. Circulation 2011;124:2215–24. https://doi.org/10.1161/CIRCULATIONAHA.111.050427.
Ziadi MC, deKemp RA, Williams KA, Guo A, Chow BJW, Renaud JM, et al. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol 2011;58:740–8. https://doi.org/10.1016/j.jacc.2011.01.065.
Chun SY, Reese TG, Ouyang J, Guerin B, Catana C, Zhu X, et al. MRI-based nonrigid motion correction in simultaneous PET/MRI. J Nucl Med 2012;53:1284–91. https://doi.org/10.2967/jnumed.111.092353.
Küstner T, Schwartz M, Martirosian P, Gatidis S, Seith F, Gilliam C, et al. MR-based respiratory and cardiac motion correction for PET imaging. Med Image Anal 2017;42:129–44. https://doi.org/10.1016/j.media.2017.08.002.
Lamare F, Le Maitre A, Dawood M, Schäfers KP, Fernandez P, Rimoldi OE, et al. Evaluation of respiratory and cardiac motion correction schemes in dual gated PET/CT cardiac imaging: Motion correction strategies in dual gated cardiac. Med Phys 2014;41:072504. https://doi.org/10.1118/1.4881099.
Naum A, Laaksonen MS, Tuunanen H, Oikonen V, Teräs M, Kemppainen J, et al. Motion detection and correction for dynamic 15O-water myocardial perfusion PET studies. Eur J Nucl Med Mol Imaging 2005;32:1378–83. https://doi.org/10.1007/s00259-005-1846-4.
Koshino K, Watabe H, Enmi J, Hirano Y, Zeniya T, Hasegawa S, et al. Effects of patient movement on measurements of myocardial blood flow and viability in resting 15O-water PET studies. J Nucl Cardiol 2012;19:524–33. https://doi.org/10.1007/s12350-012-9522-0.
Yu Y, Chan C, Ma T, Liu Y, Gallezot J-D, Naganawa M, et al. Event-by-event continuous respiratory motion correction for dynamic PET imaging. J Nucl Med 2016;57:1084–90. https://doi.org/10.2967/jnumed.115.167676.
Kesner AL, Schleyer PJ, Büther F, Walter MA, Schäfers KP, Koo PJ. On transcending the impasse of respiratory motion correction applications in routine clinical imaging: A consideration of a fully automated data driven motion control framework. EJNMMI Phys 2014;1:8. https://doi.org/10.1186/2197-7364-1-8.
Woo J, Tamarappoo B, Dey D, Nakazato R, Le Meunier L, Ramesh A, et al. Automatic 3D registration of dynamic stress and rest 82 Rb and flurpiridaz F 18 myocardial perfusion PET data for patient motion detection and correction: Automated PET motion correction. Med Phys 2011;38:6313–26. https://doi.org/10.1118/1.3656951.
Hunter CRRN, Klein R, Beanlands RS, deKemp RA. Patient motion effects on the quantification of regional myocardial blood flow with dynamic PET imaging: Patient motion effects on regional myocardial blood flow. Med Phys 2016;43:1829–40. https://doi.org/10.1118/1.4943565.
Memmott MJ, Tonge CM, Saint KJ, Arumugam P. Impact of pharmacological stress agent on patient motion during rubidium-82 myocardial perfusion PET/CT. J Nucl Cardiol 2017. https://doi.org/10.1007/s12350-016-0767-x.
Turkington TG, DeGrado TR, Hanson MW, Coleman RE. Alignment of dynamic cardiac PET images for correction of motion. IEEE Trans Nucl Sci 1997;44:235–42. https://doi.org/10.1109/23.568814.
Efseaff M, Klein R, Ziadi MC, Beanlands RS, deKemp RA. Short-term repeatability of resting myocardial blood flow measurements using rubidium-82 PET imaging. J Nucl Cardiol 2012;19:997–1006.
Lee BC, Moody JB, Weinberg RL, Corbett JR, Ficaro EP, Murthy VL. Optimization of temporal sampling for 82rubidium PET myocardial blood flow quantification. J Nucl Cardiol 2017. https://doi.org/10.1007/s12350-017-0899-7.
Ficaro E, Lee B, Kritzman J, Corbett J. Corridor4DM: The Michigan method for quantitative nuclear cardiology. J Nucl Cardiol 2007;14:455–65. https://doi.org/10.1016/j.nuclcard.2007.06.006.
Lortie M, Beanlands RSB, Yoshinaga K, Klein R, Dasilva JN, DeKemp RA. Quantification of myocardial blood flow with 82Rb dynamic PET imaging. Eur J Nucl Med Mol Imaging 2007;34:1765–74.
Friedman J, Van Train K, Maddahi J, Rozanski A, Prigent F, Bietendorf J, et al. “Upward creep” of the heart: A frequent source of false-positive reversible defects during thallium-201 stress-redistribution SPECT. J Nucl Med 1989;30:1718–22.
Murthy VL, Bateman TM, Beanlands RS, Berman DS, Borges-Neto S, Chareonthaitawee P, et al. Clinical quantification of myocardial blood flow using PET: joint position paper of the SNMMI cardiovascular council and the ASNC. J Nucl Med 2017. https://doi.org/10.2967/jnumed.117.201368.
Disclosures
B.C. Lee, J.B. Moody, and A. Poitrasson-Rivière are employees of INVIA Medical Imaging Solutions. A.C. Melvin and R.L. Weinberg have no disclosures. J.R. Corbett and E.P. Ficaro are owners of INVIA Medical Imaging Solutions. V.L. Murthy has received consulting fees from Ionetix, Inc, and owns stock in General Electric and Cardinal Health and stock options in Ionetix, Inc. V.L. Murthy is supported by 1R01HL136685 from the National, Heart, Lung, Blood Institute, and research grants from INVIA Medical Imaging Solutions and Siemens Medical Imaging.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, B.C., Moody, J.B., Poitrasson-Rivière, A. et al. Blood pool and tissue phase patient motion effects on 82rubidium PET myocardial blood flow quantification. J. Nucl. Cardiol. 26, 1918–1929 (2019). https://doi.org/10.1007/s12350-018-1256-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-018-1256-1