Abstract
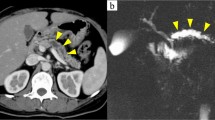
A 68-year-old woman was referred to our hospital for the treatment of bile duct stone, pancreatic tumor, and pancreatic cysts. First, bile duct stone was removed using endoscopic retrograde cholangiopancreatography. By abdominal contrast-enhanced computed tomography, a 12-mm diameter tumor was found in the pancreatic body. The tumor was isodense compared with the surrounding pancreatic parenchyma in the non-contrast phase and poorly enhanced in the arterial phase; it exhibited gradual enhancement from the portal vein phase to the late phase. Numerous pancreatic cysts were also observed by contrast-enhanced computed tomography. By magnetic resonance imaging, the tumor was hypointense in T1-weighted images, isointense in T2-weighted images, and hyperintense in diffusion-weighted images. By magnetic resonance cholangiopancreatography, the main pancreatic duct was not dilated, and pancreatic cysts communicated with the main pancreatic duct. The pancreatic cysts were diagnosed as branch-type intraductal papillary mucinous neoplasm. Histopathologic assessment of the specimens obtained by endoscopic ultrasound-guided fine-needle aspiration revealed the tumor as benign pancreatic granular cell tumor. The patient was followed up without surgical resection. On contrast-enhanced computed tomography at 6 months after admission, the tumor did not show any changes in diameter or characteristics.





Similar content being viewed by others
References
Cavaliere A, Sidoni A, Ferri I, et al. Granular cell tumor: an immunohistochemical study. Tumori. 1994;80:224–8.
Hatta J, Yanagihara M, Hasei M, et al. Case of multiple cutaneous granular cell tumors. J Dermatol. 2009;36:504–7. https://doi.org/10.1111/j.1346-8138.2009.00684.x.
Kawaida M, Fukuda H, Kohno N. Granular cell tumors arising nearly simultaneously in the larynx and subcutaneous cervical region. Ear Nose Throat J. 2000;79:162–6.
Nagaraj PB, Ongole R, Bhujanga-Rao BR. Granular cell tumor of the tongue in a 6-year-old girl—a case report. Med Oral Patol Oral Cir Bucal. 2006;11:E162–4.
El Aouni N, Laurent I, Terrier P, et al. Granular cell tumor of the breast. Diagn Cytopathol. 2007;35:725–7.
Markesbery WR, Duffy PE, Cowen D. Granular cell tumors of the central nervous system. J Neuropathol Exp Neurol. 1973;32:725–7.
Bean SM, Eloubeidi MA, Eltoum IA, et al. Preoperative diagnosis of a mediastinal granular cell tumor by EUS-FNA: a case report and review of the literature. Cytojournal. 2005;2:8.
Nakachi A, Miyazato H, Oshiro T, et al. Granular cell tumor of the rectum: a case report and review of the literature. J Gastroenterol. 2000;35:631–4.
Altavilla G, Brotto M, Busatto G, et al. Granular cell tumor of the intrapancreatic common bile duct: one case report and review of the literature. Ultrastruct Pathol. 2004;28:171–6.
Wellmann KF, Tsai CY, Reyes FB. Granular-cell myoblastoma in pancreas. N Y State J Med. 1975;75:1270.
Seidler A, Burstein S, Drweiga W, et al. Granular cell tumor of the pancreas. J Clin Gastroenterol. 1986;8:207–9.
Sekas G, Talamo TS, Julian TB. Obstruction of the pancreatic duct by a granular cell tumor. Dig Dis Sci. 1988;33:1334–7.
Nojiri T, Unemura Y, Hashimoto K, et al. Pancreatic granular cell tumor combined with carcinoma in situ. Pathol Int. 2001;51:879–82.
Bin-Sagheer ST, Brady PG, Brantley S, et al. Granular cell tumor of the pancreas: presentation with pancreatic duct obstruction. J Clin Gastroenterol. 2002;35:412–3.
Méklati EHM, Lévy P, O’Toole D, et al. Granular cell tumor of the pancreas. Pancreas. 1004;31:296–8.
Kanno A, Satoh K, Hirota M, et al. Granular cell tumor of the pancreas: a case report and review of literature. World J Gastrointest Oncol. 2010;2:121–4. https://doi.org/10.4251/wjgo.v2.i2.121.
Abrikossoff A. Über Myome ausgehend von der quergestreiften willkürlichen Muskulatur. Virchows Arch. 1926;260:215–33.
Rejas RA, Campos MS, Cortes AR, et al. The neural histogenetic origin of the oral granular cell tumor: an immunohistochemical evidence. Med Oral Patol Oral Cir Bucal. 2011;16:e6–10.
Le BH, Boyer PJ, Lewis JE, et al. Granular cell tumor: immunohistochemical assessment of inhibin-alpha, protein gene product 9.5, S100 protein, CD68, and Ki-67 proliferative index with clinical correlation. Arch Pathol Lab Med. 2004;128:771–5.
Vance SF 3rd, Hudson RP Jr. Granular cell myoblastoma. Clinicopathologic study of forty-two patients. Am J Clin Pathol. 1969;52:208–11.
Fanburg-Smith JC, Meis-Kindblom JM, Fante R, et al. Malignant granular cell tumor of soft tissue: diagnostic criteria and clinicopathologic correlation. Am J Surg Pathol. 1998;22:779–94.
Wani S, Muthusamy VR, Komanduri S. EUS-guided tissue acquisition: an evidence-based approach (with videos). Gastrointest Endosc. 2014;80(939–59):e7. https://doi.org/10.1016/j.gie.2014.07.066.
Ganc R, Colaiacovo R, Carbonari A, et al. Endoscopic ultrasonography-fine-needle aspiration of solid pancreatic lesions: a prospective, randomized, single-blinded, comparative study using the 22 Gauge EchoTip® ProCoreTM HD (A) and the 22 Gauge EchoTip® Ultra HD (B) endoscopic ultrasound needles. Endosc Ultrasound. 2014;3:S11.
Acknowledgements
The authors would like to thank all the staff involved in diagnosis and treatment of pancreatobiliary disease at Chiba University Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Koji Takahashi, Rintaro Mikata, Toshio Tsuyuguchi, Junichiro Kumagai, Masato Nakamura, Yotaro Iino, Ayako Shingyoji, Mutsumi Yamato, Hiroshi Ohyama, Yuko Kusakabe, Shin Yasui, Harutoshi Sugiyama, Takashi Kishimoto, Yukio Nakatani, and Naoya Kato declare that they have no conflict of interest.
Human rights
This study does not include any data about human specimen. All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
This is a case report and does not involve human subjects and does not apply to giving informed consent.
Rights and permissions
About this article
Cite this article
Takahashi, K., Mikata, R., Tsuyuguchi, T. et al. Granular cell tumor of the pancreas diagnosed by endoscopic ultrasound-guided fine-needle aspiration. Clin J Gastroenterol 11, 193–199 (2018). https://doi.org/10.1007/s12328-018-0821-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-018-0821-0