Abstract
Introduction
Haptoglobin (Hp) genotypes were reported as an independent risk factor for metabolic diseases. This study aimed to investigate the association between Hp gene polymorphism and the severity of nonalcoholic fatty liver disease (NAFLD).
Methods
A total of 441 subjects (NAFLD group, n = 272; healthy control, n = 169) were recruited, and their clinical biochemical parameters were measured in all subjects. Haptoglobin genotyping was performed using genomic DNA extracted from peripheral blood leukocytes. Among the NAFLD group, 107 patients underwent liver biopsy, and histology was evaluated by a pathologist on the basis of the CRN scoring system.
Results
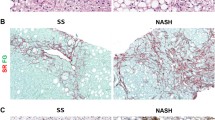
NAFLD patients had much lower frequency of Hp 1-1 genotype and higher frequency of Hp 2-2 than healthy controls (0.4% vs 9.5%, 55.8% vs 47.9%, P < 0.001). NAFLD patients with Hp 2-2 genotype had much higher levels of body mass index (BMI), total cholesterol (TC), liver enzymes, ferritin, and controlled attenuation parameter (CAP) values than non-Hp 2-2 genotype (P < 0.05). In histology, patients with nonalcoholic steatohepatitis (NASH) had higher frequency of Hp 2-2 genotype than non-NASH patients (71.3% vs 22.2%, P < 0.001); patients with significant fibrosis had higher frequency of Hp 2-2 genotype (78.3% vs 54.8%, P < 0.05) than no/mild fibrosis patients. NAFLD patients with Hp 2-2 genotype had higher proportion with higher steatosis scores, lobular inflammation scores, ballooning scores, NAFLD activity scores (NAS), and fibrosis stages (P < 0.05 for all) than Hp 2-2 groups. Furthermore, Hp 2-2 genotype was independently associated with NASH (OR = 5.985, P < 0.05) and significant fibrosis (OR = 6.584, P < 0.05).
Conclusions
Hp 2-2 genotype is closely associated with the severity of NAFLD.


Similar content being viewed by others
Abbreviations
- ALB:
-
Serum albumin
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- BMI:
-
Body mass index
- CAP:
-
Controlled attenuation parameter
- FER:
-
Ferritin
- FPG:
-
Fasting plasma glucose
- GGT:
-
Gamma-glutamyl transferase
- HA:
-
Hyaluronic acid
- HCC:
-
Hepatocellular carcinoma
- HDL-C:
-
High-density lipoprotein
- Hp:
-
Haptoglobin
- L:
-
Lymphocyte
- LDH:
-
Lactate dehydrogenase
- LDL-C:
-
Low-density lipoprotein
- LSM:
-
Liver stiffness measurement
- N:
-
Neutrophils
- NAFLD:
-
Nonalcoholic fatty liver disease
- NAS:
-
Nonalcoholic fatty liver disease activity score
- NASH:
-
Nonalcoholic steatohepatitis
- N/L ratio:
-
Neutrophil/lymphocyte ratio
- OR:
-
Odds ratio
- PIIINP:
-
Type III procollagen amino terminal peptide
- SI:
-
Serum iron
- T2DM:
-
Type 2 diabetes mellitus
- TC:
-
Total cholesterol
- TE:
-
Transient elastography
- TG:
-
Triglyceride
- TIBC:
-
Total ferritin binding
- TNF-α:
-
Tumor necrosis factor alpha
- UA:
-
Uric acid
- WBC:
-
White blood cell
References
Yki-Jarvinen H. Non-alcoholic fatty liver disease as a cause and aconsequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014;2:901–10.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67(1):328–357.
Fan JG, Kim SU, Wong VW. New trends on obesity and NAFLD in Asia. J Hepatol. 2017;67(4):862–73.
Wong RJ, Aguilar M, Cheung R, et al. Nonalcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States. Gastroenterology. 2015;148:547–55.
Younossi ZM, Otgonsuren M, Henry L, et al. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology. 2015;62(6):1723–30.
Xun YH, Fan JG, Zang GQ, et al. Suboptimal performance of simple noninvasive test for advanced fibrosis in Chinese patients with nonalcoholic fatty liver disease. J Dig Dis. 2012;13(11):588–95.
Feng S, Rui-Dan Z, Jun-Ping S, et al. Impact of skin capsular distance on the performance of controlled attenuation parameter in patients with chronic liver disease. Liver Int. 2015;35:2392–400.
Osterreicher CH, Brenner DA. The genetics of nonalcoholic fatty liver disease. Ann Hepatol. 2007;6(2):83–8.
Andersen CBF, Stødkilde K, Sæderup KL, et al. Haptoglobin. Antioxid Redox Signal. 2017;26(14):814–31.
Blum S, Vardi M, Brown JB, et al. Vitamin E reduces cardiovascular disease in individuals with diabetes mellitus and the haptoglobin 2–2 genotype. Pharmacogenomics. 2010;11(5):675–84.
Boettger LM, Salem RM, Handsaker RE, et al. Recurring exon deletions in the HP (haptoglobin) gene contribute to lower blood cholesterol levels. Nat Genet. 2016;48(4):359.
Suleiman M, Aronson D, Asleh R, et al. Haptoglobin polymorphism predicts 30-day mortality and heart failure in patients with diabetes and acute myocardial infarction. Diabetes. 2005;54(9):2802–6.
Asleh R, Blum S, Kalet-Litman S, et al. Correction of HDL dysfunction in individuals with diabetes and the haptoglobin 2–2 genotype. Diabetes. 2008;57(10):2794–800.
Levy AP, Hochberg I, Jablonski K, et al. Haptoglobin phenotype is an independent risk factor for cardiovascular disease in individuals with diabetes : the Strong Heart Study. J Am Coll Car. 2002;40(11):1984–90.
Roguin A, Koch W, Kastrati A, et al. Haptoglobin genotype is predictive of major adverse cardiac events in the one-year period after PTCA in individuals with diabetes. Diabetes Care. 2003;26:2628–31.
Milman U, Blum S, Shapira C, et al. Vitamin E supplementation reduces cardiovascular events in a subgroup of middle-aged individuals with both type 2 diabetes mellitus and the haptoglobin 2-2 genotype: a prospective, double-blinded clinical trial. Art Thromb Vasc Biol. 2008;28(2):341–7.
Costacou T, Ferrell RE, Orchard TJ. Haptoglobin genotype: a determinant of cardiovascular complication risk in type 1 diabetes. Diabetes. 2008;57:1702–6.
Nakagawa T, Muramoto Y, Hori M, et al. A preliminary investigation of the association between haptoglobin polymorphism, serum ferritin concentration and fatty liver disease. Clin Chim Acta. 2008;398(1):34–8.
Banini BA, Cazanave SC, Yates KP, et al. Haptoglobin 2 allele is associated with histologic response to vitamin E in subjects with nonalcoholic steatohepatitis. J Clin Gastroenterol. 2018. https://doi.org/10.1097/MCG.0000000000001142.
Fan JG, Jia JD, Li YM, et al. Guidelines for the diagnosis and management of nonalcoholic fatty liver disease: update 2010: (published in Chinese on Chinese Journal of Hepatology 2010; 18:163–166). J Dig Dis. 2011;12(1):45–50.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Chalasani NP, Sanyal AJ, Kowdley KV, et al. Pioglitazone versus vitamin E versus placebo for the treatment of non-diabetic patients with non-alcoholic steatohepatitis: PIVENS trial design. Contemp Clin Trials. 2009;30(1):88–96.
Koch W, Latz W, Eichinger M, et al. Genotyping of the common haptoglobin Hp 1/2polymorphism based on PCR. Clin Chem. 2002;48(9):1377–82.
Carter K, Worwood M. Haptoglobin: a review of the major allele frequencies worldwide and their association with diseases. Int J Lab Hematol. 2007;29(2):92–110.
Costacou T, Ferrell RE, Ellis D, et al. Haptoglobin genotype and renal function decline in type 1 diabetes. Diabetes. 2009;58(12):2904–9.
Shiyun W, Jie W, Rong Z, et al. Mendelian randomization analysis to assess a causal efect of haptoglobin on macroangiopathy in Chinese type 2 diabetes patients. Cardiovasc Diabetol. 2018;16(1):14.
Xiaohong S, Liang S, Li W, et al. Haptoglobin 2-2 genotype Is associated with increased risk of type 2 diabetes mellitus in Northern Chinese. Genet Test Mol Biomarkers. 2012;16(6):563–8.
Shufei Z, Jin C, Yu S, et al. Haptoglobin genotype and vitamin E versus placebo for the treatment of nondiabetic patients with nonalcoholic steatohepatitis in China: a multicenter, randomized, placebo-controlled trial design. Adv Ther. 2018;35:218–23.
Weisberg SP, McCann D, Desai M, et al. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Investig. 2003;112(12):1796–808.
Zethelius B, Berglund L, Sundström J, et al. Use of multiple biomarkers toimprove the prediction of death from cardiovascular causes. N Engl J Med 2008;358(20):2107–16.
Willer CJ, Schmidt EM, Sengupta S, et al. Discovery and refinement of loci associated with lipid levels. Nat Genet. 2013;45(11):1274.
Boettger LM, Salem RM, Handsaker RE, et al. Recurring exon deletions in the HP (haptoglobin) gene contribute to lower blood cholesterol levels. Nat Genet. 2016;48(4):359–66.
Zheng NS, Bastarache LA, Bastarache JA, et al. A common deletion in the haptoglobin gene associated with blood cholesterol levels among Chinese women. J Hum Genet. 2017;62(10):911–4.
Salvatore A, Cigliano L, Carlucci A, et al. Haptoglobin binds apolipoprotein E and influences cholesterol esterification in the cerebrospinal fluid. J Neurochem. 2009;110(1):255–63.
Yang Y, Cao Z, Tian L, et al. VPO1 mediates ApoE oxidation and impairs the clearance of plasma lipids. PLoS One. 2013;8(2):e57571.
Acknowledgements
Funding
Sponsorship for this study, development of this manuscript, and article processing charges were funded by the National Natural Science Foundation (81700510, 81570524), the Natural Science Foundation of Zhejiang Province (LY15H070004, LQ15H070006), the Science and Technology Planning Project of Hangzhou City (20170533B43, 20172016A02), the Nature Science Foundation of Min hang District of Shanghai (2018MHZ089, 2018MHZ027), and the Key Project of The Fifth People's Hospital of Shanghai (2018wyzd04). All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Editorial Assistance
Our thanks go to Dr Zhenjie Zhuang for assistance in preparation of this manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Authorship Contributions
Shufei Zang and Junping Shi designed experiments; Jingxin Zhou, Huiping Sheng, and Ningning You carried out experiments (Hp genotyping), Jun Liu analyzed experimental results and helped revise the manuscript (statistical analysis). Jin Chen and Xiaoxiao Mi performed physical examination and Fibroscan tests, Wenjun Yang performed histopathologic analyses. Jingxin Zhou and Shufei zang wrote the manuscript.
Disclosures
All authors (Jingxin Zhou, Jun Liu, Huiping Sheng, Ningning You, Jin Chen, Xiaoxiao Mi, Wenjun Yang, Shufei Zang, and Junping Shi) declare that they have nothing to disclose.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the Ethics Committee of Hangzhou Normal University Affiliated Hospital (ethics no. 2015-HS-001) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Data Availability
The data sets generated and analyzed during the current study are not publicly available because of information security, but are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Consortia
Corresponding authors
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.7643432.
Rights and permissions
About this article
Cite this article
Zhou, J., Liu, J., Sheng, H. et al. Haptoglobin 2-2 Genotype is Associated with More Advanced Disease in Subjects with Non-Alcoholic Steatohepatitis: A Retrospective Study. Adv Ther 36, 880–895 (2019). https://doi.org/10.1007/s12325-019-00902-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-00902-z