Abstract
Introduction
The objective of this study was to compare the relative bioavailability of the US Food and Drug Administration-approved formulation of colchicine after a single 0.6 mg dose in young (18–30 years of age) and elderly (≥60 years of age) healthy subjects to determine whether dosing adjustments are required in elderly patients.
Methods
A single-dose, single-drug, parallel-group study was performed in 20 young subjects with normal renal function (defined as creatinine clearance [CrCl] ≥80 mL/min) and 18 elderly subjects with normal or mild renal impairment (CrCl ≥50 mL/min) in otherwise good health. Blood samples were collected for up to 72 hours postdose and analyzed for colchicine using a validated liquid chromatography/tandem mass spectrometry method. Noncompartmental pharmacokinetic parameters were compared using analysis of variance methods.
Results
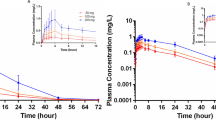
There were no statistically significant (P < 0.05) differences in mean colchicine pharmacokinetic parameters between young and elderly subjects, including peak plasma concentration (C max) (2.53 vs. 2.56 ng/mL), time to C max (1.25 vs. 1.25 hours), area under the plasma concentration-time curve to infinity (22.29 vs. 25.01 ng/h/mL), elimination half-life (25.4 vs. 30.1 hours), oral clearance (0.40 vs. 0.35 L/h/kg), and apparent volume of distribution (14.3 vs. 14.8 L/kg), respectively.
Conclusion
The lack of any significant differences in colchicine pharmacokinetic parameters between young and elderly healthy subjects, with some of the latter including mild renal impairment, suggests that dose modification of colchicine may not be necessary in healthy elderly patients. However, when evaluating the use of colchicine dosing in an elderly patient, the confounding effect on overall exposure and safety from comorbid conditions, the use of concomitant medications, and the administration of multiple doses should be considered.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Choi HK, Mount DB, Reginato AM. Pathogenesis of gout. Ann Intern Med. 2005;143:499–516.
Roddy E, Zhang W, Doherty M. The changing epidemiology of gout. Nat Clin Pract Rheumatol. 2007;3:443–449.
Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum. 2011;63:3136–3141.
Colcrys(colchicine, USP) tablets for oral use: prescribing information, 14 March 2012. Available at: www.colcrys.com/assets/pdf/COLCRYS_Full_Prescribing_Information.pdf. Accessed Apr 25 2012.
Niel E, Schermann J-M. Colchicine today. Joint Bone Spine. 2006;73:672–678.
Terkeltaub RA. Colchicine update: 2008. Semin Arthritis Rheum. 2009;38:411–419.
Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–241.
Rochdi M, Sabouraud A, Girre C, Venet R, Scherrmann JM. Pharmacokinetics and absolute bioavailability of colchicine after i.v. and oral administration in healthy human volunteers and elderly subjects. Eur J Clin Pharmacol. 1994;46:351–354.
Nuki G. Colchicine: its mechanism of action and efficacy in crystal-induced inflammation. Curr Rheumatol Rep. 2008;10:218–227.
Chappey ON, Niel E, Wautier JL, et al. Colchicine disposition in human leukocytes after single and multiple oral administration. Clin Pharmacol Ther. 1993;54:360–367.
Terkeltaub RA, Furst DE, Bennett K, Kook KA, Crockett RS, Davis MW. High versus low dosing of oral colchicine for early acute gout flare: twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum. 2010;62:1060–1068.
Terkeltaub RA. Gout. N Engl J Med. 2003;349:1647–1655.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
This article is published with open access at Springerlink.com
ClinicalTrials.Gov #NCT01001052.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Wason, S., Faulkner, R.D. & Davis, M.W. Are Dosing Adjustments Required for Colchicine in the Elderly Compared with Younger Patients?. Adv Therapy 29, 551–561 (2012). https://doi.org/10.1007/s12325-012-0028-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-012-0028-6