Abstract
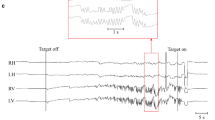
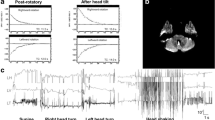
The cerebellar flocculus is a critical structure involved in the control of eye movements. Both static and dynamic abnormalities of the vestibulo-ocular reflex (VOR) have been described in animals with experimental lesions of the flocculus/paraflocculus complex. In humans, lesions restricted to the flocculus are rare so they can become an exceptional model to contrast with the clinical features in experimental animals or in patients with more generalized cerebellar diseases. Here, we examined a 67-year-old patient with an acute vestibular syndrome due to an isolated infarct of the right flocculus. We evaluated him multiple times over 6 months—to follow the changes in eye movements and vestibular function—with caloric testing, video-oculography and head-impulse testing, and the anatomical changes on imaging. Acutely, he had an ipsilateral-beating spontaneous nystagmus, bilateral gaze-evoked nystagmus, borderline impaired smooth pursuit, and a complete contraversive ocular tilt reaction. The VOR gain was reduced for head impulses directed contralateral to the lesion, and there was also an ipsilesional caloric weakness. All abnormalities progressively improved at follow-up visits but with a considerable reduction in volume of the affected flocculus on imaging. The vestibular and ocular motor findings, qualitatively similar to a previously reported patient, further clarify the “acute floccular syndrome” in humans. We also add new information about the pattern of recovery from such a lesion with corresponding changes in the size of the affected flocculus on imaging.





Similar content being viewed by others
References
Rambold H, Churchland A, Selig Y, Jasmin L, Lisberger SG. Partial ablations of the flocculus and ventral paraflocculus in monkeys cause linked deficits in smooth pursuit eye movements and adaptive modification of the VOR. J Neurophysiol. 2002;87:912–24.
Takemori S, Cohen B. Loss of visual suppression of vestibular nystagmus after flocculus lesions. Brain Res. 1974;72:213–24.
Zee DS, Yamazaki A, Butler PH, Gucer G. Effects of ablation of flocculus and paraflocculus of eye movements in primate. J Neurophysiol. 1981;46:878–99.
Lisberger SG, Miles FA, Zee DS. Signals used to compute errors in monkey vestibuloocular reflex: possible role of flocculus. J Neurophysiol. 1984;52:1140–53.
Lee SH, Park SH, Kim JS, Kim HJ, Yunusov F, Zee DS. Isolated unilateral infarction of the cerebellar tonsil: ocular motor findings. Ann Neurol. 2014;75:429–34.
Mai JK, Paxinos G. The human nervous system. Amsterdam: Elsevier Academic Press; 2012.
Park HK, Kim JS, Strupp M, Zee DS. Isolated floccular infarction: impaired vestibular responses to horizontal head impulse. J Neurol. 2013;260:1576–82. https://doi.org/10.1007/s00415-013-6837-y.
Newman-Toker DE, Kattah JC, Alvernia JE, Wang DZ. Normal head impulse test differentiates acute cerebellar strokes from vestibular neuritis. Neurology. 2008;70:2378–85. https://doi.org/10.1212/01.wnl.0000314685.01433.0d.
Casani AP, Dallan I, Cerchiai N, Lenzi R, Cosottini M, Sellari-Franceschini S. Cerebellar infarctions mimicking acute peripheral vertigo: how to avoid misdiagnosis? Otolaryngol Head Neck Surg. 2013;148:475–81. https://doi.org/10.1177/0194599812472614.
Kattah JC, Talkad AV, Wang DZ, Hsieh YH, Newman-Toker DE. HINTS to diagnose stroke in the acute vestibular syndrome: three-step bedside oculomotor examination more sensitive than early MRI diffusion-weighted imaging. Stroke. 2009;40:3504–10. https://doi.org/10.1161/STROKEAHA.109.551234.
Halmagyi GM, Chen L, MacDougall HG, Weber KP, McGarvie LA, Curthoys IS. The video head impulse test. Front Neurol. 2017; https://doi.org/10.3389/fneur.2017.00258.
Zwergal A, Rettinger N, Frenzel C, Dieterich M, Brandt T, Strupp M. A bucket of static vestibular function. Neurology. 2009;72:1689–92. https://doi.org/10.1212/WNL.0b013e3181a55ecf.
Huh YE, Kim JS. Bedside evaluation of dizzy patients. J Clin Neurol. 2013;9:203–13. https://doi.org/10.3988/jcn.2013.9.4.203.
Koo JW, Kim JS, Hong SK. Vibration-induced nystagmus after acute peripheral vestibular loss: comparative study with other vestibule-ocular reflex tests in the yaw plane. Otol Neurotol. 2011;32:466–71. https://doi.org/10.1097/MAO.0b013e31820d9685.
Luis L, Costa J, Munoz E, de Carvalho M, Carmona S, Schneider E, et al. Vestibulo-ocular reflex dynamics with head-impulses discriminates spinocerebellar ataxias types 1, 2 and 3 and Friedreich ataxia. J Vestib Res. 2016;26:327–34. https://doi.org/10.3233/VES-160579.
Mossman B, Mossman S, Purdie G, Schneider E. Age dependent normal horizontal VOR gain of head impulse test as measured with video-oculography. J Otolaryngol Head Neck Surg. 2015;44:29. https://doi.org/10.1186/s40463-015-0081-7.
Piker EG, Jacobson GP, McCaslin DL, Hood LJ. Normal characteristics of the ocular vestibular evoked myogenic potential. J Am Acad Audiol. 2011;22:222–30. https://doi.org/10.3766/jaaa.22.4.5.
McCaslin DL, Jacobson GP, Hatton K, Fowler AP, DeLong AP. The effects of amplitude normalization and EMG targets on cVEMP interaural amplitude asymmetry. Ear Hear. 2013;34:482–90. https://doi.org/10.1097/AUD.0b013e31827ad792.
Schmahmann JD. MRI atlas of the human cerebellum. San Diego: Academic Press; 2000.
Schmahmann JD, Doyon J, McDonald D, Holmes C, Lavoie K, Hurwitz AS, et al. Three-dimensional MRI atlas of the human cerebellum in proportional stereotaxic space. NeuroImage. 1999;10:233–60. https://doi.org/10.1006/nimg.1999.0459.
Luis L, Lehnen N, Munoz E, de Carvalho M, Schneider E, Valls-Sole J, et al. Anticompensatory quick eye movements after head impulses: a peripheral vestibular sign in spontaneous nystagmus. J Vestib Res. 2016;25:267–71. https://doi.org/10.3233/VES-160566.
Tarnutzer AA, Berkowitz AL, Robinson KA, Hsieh YH, Newman-Toker DE. Does my dizzy patient have a stroke? A systematic review of bedside diagnosis in acute vestibular syndrome. CMAJ. 2011;183:E571–92. https://doi.org/10.1503/cmaj.100174.
Kremmyda O, Kirchner H, Glasauer S, Brandt T, Jahn K, Strupp M. False-positive head-impulse test in cerebellar ataxia. Front Neurol. 2012;3:162. https://doi.org/10.3389/fneur.2012.00162.
Huh YE, Kim JS, Kim HJ, Park SH, Jeon BS, Kim JM, et al. Vestibular performance during high-acceleration stimuli correlates with clinical decline in SCA6. Cerebellum. 2015;14:284–91. https://doi.org/10.1007/s12311-015-0650-3.
Kim SH, Zee DS, du Lac S, Kim HJ, Kim JS. Nucleus prepositus hypoglossi lesions produce a unique ocular motor syndrome. Neurology. 2016;87:2026–33. https://doi.org/10.1212/WNL.0000000000003316.
Reutern GM, Dichgans J. Oculomotor disturbances as cerebellar symptoms in pontine angle tumors. Contralateral diminution of optokinetic nystagmus as early sign of floccular lesions (author’s transl). Arch Psychiatr Nervenkr (1970). 1977;223:117–30.
Waespe W. Deficits of smooth-pursuit eye movements in two patients with a lesion in the (para-)floccular or dorsolateral pontine region. Neuro-Ophthalmology. 1992;12:91–6. https://doi.org/10.3109/01658109209058123.
Baek SH, Choi JY, Jung JM, Kwon DY, Park MH, Choi J, et al. Abnormal head impulse test in a unilateral cerebellar lesion. J Clin Neurol. 2015;11:279–82. https://doi.org/10.3988/jcn.2015.11.3.279.
Ohki M, Kitazawa H, Hiramatsu T, Kaga K, Kitamura T, Yamada J, et al. Role of primate cerebellar hemisphere in voluntary eye movement control revealed by lesion effects. J Neurophysiol. 2009;101:934–47. https://doi.org/10.1152/jn.90440.2009.
Xiong G, Nagao S, Kitazawa H. Mossy and climbing fiber collateral inputs in monkey cerebellar paraflocculus lobulus petrosus and hemispheric lobule VII and their relevance to oculomotor functions. Neurosci Lett. 2010;468:282–6. https://doi.org/10.1016/j.neulet.2009.11.013.
Baier B, Bense S, Dieterich M. Are signs of ocular tilt reaction in patients with cerebellar lesions mediated by the dentate nucleus? Brain. 2008;131:1445–54. https://doi.org/10.1093/brain/awn086.
Wong AM. New understanding on the contribution of the central otolithic system to eye movement and skew deviation. Eye (Lond). 2015;29:153–6. https://doi.org/10.1038/eye.2014.243.
Brodsky MC, Donahue SP, Vaphiades M, Brandt T. Skew deviation revisited. Surv Ophthalmol. 2006;51:105–28. https://doi.org/10.1016/j.survophthal.2005.12.008.
Mossman S, Halmagyi GM. Partial ocular tilt reaction due to unilateral cerebellar lesion. Neurology. 1997;49:491–3.
Choi SY, Lee SH, Kim HJ, Kim JS. Impaired modulation of the otolithic function in acute unilateral cerebellar infarction. Cerebellum. 2014;13:362–71. https://doi.org/10.1007/s12311-013-0544-1.
Shaikh AG, Marti S, Tarnutzer AA, Palla A, Crawford TO, Straumann D, et al. Ataxia telangiectasia: a “disease model” to understand the cerebellar control of vestibular reflexes. J Neurophysiol. 2011;105:3034–41. https://doi.org/10.1152/jn.00721.2010.
Weber KP, Rosengren SM, Michels R, Sturm V, Straumann D, Landau K. Single motor unit activity in human extraocular muscles during the vestibulo-ocular reflex. J Physiol. 2012;590:3091–101. https://doi.org/10.1113/jphysiol.2011.226225.
Acknowledgments
The authors thank Marcelo Duperre for his generous assistance in the production of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the local review board and the patient gave written informed consent.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yacovino, D.A., Akly, M.P., Luis, L. et al. The Floccular Syndrome: Dynamic Changes in Eye Movements and Vestibulo-ocular Reflex in Isolated Infarction of the Cerebellar Flocculus. Cerebellum 17, 122–131 (2018). https://doi.org/10.1007/s12311-017-0878-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-017-0878-1