Abstract
Extranodal natural killer/T-cell lymphoma (ENKTCL), nasal type, is rare and aggressive and often involves the nose, nasopharynx, and upper aerodigestive tract. The non-nasal type can affect the skin, salivary glands, gut, testes, brain, salivary glands, and other sites. Primary ENKTCL of the lung is rare. Here, we report a 68-year-old non-smoking female who presented with fever, dry cough, and night sweats. The chest image showed lung consolidation in the right lower lung field. Pulmonary biopsy showed diffuse abnormal lymphocyte infiltrate in the necrotic exudate. Immunohistochemical data indicated that the tumour cells were positive for CD56, granzyme B, CD3, and TIA. Using in situ hybridization, Epstein–Barr virus-encoded ribonucleic acid (EBER) was detected. There was no evidence to indicate extrathoracic lymphoma involvement. Primary pulmonary ENKTCL was therefore diagnosed. The patient underwent chemotherapy using the P-GEMOX regimen (pegaspargase, gemcitabine, and oxaliplatin) and is still alive.
Similar content being viewed by others
Introduction
Extranodal NK/T-cell lymphoma (ENKTCL), nasal type, is a rare and invasive neoplasm with a poor prognosis that is closely associated with Epstein–Barr virus (EBV) infection. While lymphoma can occur in the nasal and upper aerodigestive regions, the skin, salivary gland, testis, or gastrointestinal tract, and causes lymphadenopathy and hepatosplenomegaly, the nasal and upper aerodigestive regions are the most common invasive sites [1, 2]. Primary pulmonary ENKTCL is very rare, with no more than twenty cases reported in the English literature [3,4,5,6,7,8,9,10,11,12,13,14,15]. Here, we describe a case of primary pulmonary ENKTCL and review the related literature.
Clinical history
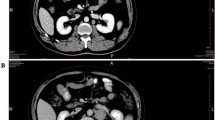
A 68-year-old non-smoking female presented with fever, dry cough, and night sweats in the clinic. The chest computed tomography (CT) scan revealed a soft tissue mass in the right lung (Fig. 1a). The T-SPOT, bone scan, and head CT did not show any abnormalities. Her temperature was 36.1 °C, pulse 78 bpm, breathing 18 bpm, and blood pressure 124/75 mmHg. No palpable lymph nodes were found on the neck, supraclavicular area, axillary, or inguinal lymph nodes. Laboratory examination results showed the following: white blood cell count, 6.7 × 109/L; neutrophil percentage, 74.6%; haemoglobin, 111 g/L; platelet count, 296 × 109/L; total protein (TP), 66.3 g/L; blood urea nitrogen (BUN), 3.88 mmol/L; creatinine (Cr), 43.9 μmol/L; C-reactive protein (CRP), 46.92 mg/L; LDH, 194 U/L; procalcitonin (PCT), 0.03 ng/mL; and serum ferritin, 610.95 ng/mL. Tumour markers were all normal. She was then diagnosed with pneumonia and treated with cephalosporins. However, her clinical condition did not obviously improve. Bronchoscopic biopsy was not performed because she declined the procedure. Then, CT-guided needle biopsy was performed, and a final diagnosis of ENKTCL was made based on immunohistochemistry, T-cell receptor (TCR) gene rearrangement and EBER analysis results. A positron emission tomography-CT (PET-CT) scan showed significant fluorodeoxyglucose (FDG) uptake in the right lung (Fig. 1b). No other organ involvement was found by PET/CT. Based on the neoplastic morphology and the immunohistochemical examination, a diagnosis of primary pulmonary ENKTCL was made. There was no obvious abnormality in bone marrow biopsy. According to the National Comprehensive Cancer Network (NCCN) guidelines, patients are treated using the pegaspargase, gemcitabine, and oxaliplatin (P-GEMOX) regimen [16].
Imaging features. a Computed tomography (CT) scan of the chest showed dense consolidation in the right lung. b Positron emission tomography–computed tomography (PET/CT) scan showed a mass in the right lung with high-18F-fluorodeoxyglucose uptake. c After 5 cycles of treatment, chest CT scan revealed a reduction in the area of consolidation
Materials and methods
Morphology and immunohistochemistry
Lung specimens were fixed in formalin (10%) and processed routinely. Paraffin-embedded blocks were sectioned (4 μm thick) and stained with haematoxylin-eosin (H&E). Immunohistochemical analysis was carried out with a Ventana BenchMark GX system using the following primary antibodies: anti-CD56 (123C3.D5), anti-CD3 (SP7), anti-TIA (TIA-1), and anti-granzyme B (GZB01). Working solutions of the above antibodies were purchased from Fuzhou Maxim, China. Additionally, EBER in situ hybridization (EBER1 DNP Probe, Ventana) was performed.
Multiplex polymerase chain reaction for TCR gene rearrangement
Human peripheral blood mononuclear cells (PBMCs) from patients were isolated by density gradient centrifugation using Ficoll-Paque (BD, USA), and genomic DNA was extracted using the Blood Genomic DNA Kit (Fastagen, China). The multiplex polymerase chain reaction (PCR) protocols and the 56 primers used for TCR gene analysis were standardised BIOMED-2 protocols. The 56 primers used for TCR clonal rearrangement were selected from BIOMED-2 [17]. Nine groups of PCRs were conducted, including five groups for the VDJ domains of TCRβ, two groups for the VJ domains of TCRγ, one group for the VDJ/DD/VD/DJ domains of TCRδ, and one group for the positive control. The PCR cycling conditions were as follows: denaturation at 95 °C for 7 min; 45 cycles of denaturation at 95 °C for 45 s, annealing at 60 °C for 45 s, and extension at 72 °C for 90 s; and a final extension at 72 °C for 10 min. All PCR products were separated via 6% polyacrylamide gel electrophoresis in a DG-600C electrophoresis system (Dingguo, China) according to the manufacturer’s instructions. The gels were collected and analysed using a ChemiDoc XRS+ imager (Bio-Rad, USA).
Results
Histopathologic sections revealed massive prominent necrosis with diffuse dense medium-sized lymphoma cells infiltrate. These cells were markedly atypical, and nuclei were irregular, exhibiting an angiocentric angiodestructive quality (Fig. 2a). Immunohistochemical analysis revealed that the cells had the following immunotypes: CD56(+), granzyme B(+), CD3(+), TIA(+), CD20(−), CD5(−), CD8(−), CD79a(−), CD10(−), BCL-6(−) (Fig. 2b–e). Furthermore, in situ hybridization showed that these cells were strongly positive for EBER (Fig. 2f), and EBV DNA tests showed 1.24 × 105 copies/mL (normal, 5 × 103 copies/mL). TCR gene rearrangement studies for TCRβ, TCRγ, and TCRδ were negative.
a Lung biopsy showed diffuse infiltrative atypical medium-sized tumour cells had irregular nuclei, moderate in amount of cytoplasm and pale to clear in extensive coagulative necrosis (H&E, × 400 magnification). b–e Immunohistochemical analysis showed that these cells were positive for CD56 (× 400 magnification), granzyme B (× 400 magnification), CD3 (× 400 magnification), and TIA (magnification, × 400). f Epstein–Barr virus-encoded RNA was proven by in situ hybridization
Following 5-cycle P-GEMOX chemotherapy, the patient’s symptoms were significantly alleviated, and a repeat CT scan showed decreasing consolidation in the right lung (Fig. 1c). The patient achieved partial remission (PR) and is still alive.
Discussion
ENKTCL, nasal type, is rare and accounts for 10% of all peripheral T-cell lymphomas (PTCLs) worldwide. Primary pulmonary NK/T-cell lymphoma represents 0.5 to 1% of all primary pulmonary lymphomas [12, 18]. ENKTCL is geographically predominant in East Asia and southern North America but rare in Europe and Africa. In the World Health Organization (WHO) records, most patients with ENKTCL have a median age of 44–54 years, and it affects more males than females. Primary tumour sites of ENKTCL are located in the upper airway region. Usually, in the nasal area, the lesion presents as an invasive mass with surrounding tissue hyperaemia and oedema. It erodes the hard palate, leading to extensive midfacial lesions, and then extends to other adjacent and nonadjacent facial tissues, but bone marrow involvement is uncommon. The presentation of non-nasal NK/T-cell lymphoma can vary, depending on the involved sites. Histologically, features of ENKTCL show no obvious difference irrespective of their anatomical location. Lymphomatous infiltrate often exhibits diffuse and permeative infiltration. Growth patterns present angiocentricity and angiodestruction, leading to coagulative necrosis. Cytologically, most neoplastic cells are medium-sized or comprise a mixture of small and large cells with irregularly folded nuclei. In other types of lymphoma, coagulation necrosis and vascular invasion are not obvious. Its typical immunophenotype is CD56(+), cytoplasmic CD3ε(+), and CD2(+), and it is positive for cytotoxic molecules (TIA-1, granzyme B, and perforin) but negative for surface CD3 and TCR gene rearrangement. In most cases, EBER is detected by in situ hybridization, which indicates that EBV plays a highly important role in its pathogenesis [2, 19]. In our case, medium-sized atypical lymphoid and coagulative necrosis were easily found. Immunohistochemical staining confirmed that these cells were reactive for CD56, granzyme B, CD3, and TIA but were negative for CD20, CD5, CD8, CD79a, CD10, BCL-6, and TCR rearrangement. EBER was positive, and EBV DNA levels were 1.24 × 105 copies/mL. The combination of histopathology and immunophenotyping along with EBV status led to a diagnosis of ENKTCL. Based on the PET-CT results, there were no other involvements except the lung; thus, we can confirm that NK/T-cell lymphoma originated from the lung. Because of the unique pathological characteristics of the disease, it is often misdiagnosed as other diseases, such as Wegener granulomatosis (GPA) and PTCL-not otherwise specified (NOS). GPA is a systemic autoimmune condition that often occurs in the upper and lower respiratory tract and kidney. Patients can experience recurrent sinusitis and exhibit lung nodules, inflammatory infiltration, glomerulonephritis, and mucocutaneous lesions. The histological characteristics of this disease are vasculitis and non-caseating granulomas. In the initial stage, the clinical features of ENKTCL and GPA are very similar. However, the sera of most patients with GPA contain anti-neutrophil cytoplasmic antibodies (ANCA) and lack EBER. Furthermore, the prognosis of GPA is much better than that of ENKTCL. PTCL-NOS represents a heterogeneous category of mature T-cell origin lymphomas affecting nodal and extranodal tissues. As the clinical manifestations are non-specific, we should rely on immunophenotyping for diagnosis. These neoplastic cells have the following markers: surface CD3(+), cytoplasmic CD3ε(−), CD56(−), and EBER(−), and TCR rearrangement (+). Thus, repeat biopsy and testing for CD56 or EBV-positive lymphocytes can increase the sensitivity and specificity for ENKTCL diagnoses.
Primary pulmonary ENKTCL is extremely rare. A list of retrospective clinical studies on similar English cases is provided in Table 1, encompassing studies from China, Japan, Korea, and the USA. Regarding the patients with these available cases, nine were male, and five were female, for a male-to-female ratio of approximately 1.8:1, and ranged in age from 31 to 80 years. Most patients presented with fever, cough, expectoration, and dyspnoea. The most common imaging characteristics included alveolar infiltration, pleural effusion, nodules, and ground glass opacities (GGOs). Immunologically, the patients were positive for CD56, cytoplasmic CD3ε, TIA-1, granzyme B, and perforin and negative for surface CD3, CD20, and CD79, and 64.3% were positive for EBV. The prognosis of these patients was generally poor. Three of the patients improved, but most died soon after diagnosis. Cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP)–based chemotherapy has been reported in some studies [8, 10, 14], but the patients’ symptoms did not obviously improve. Our patient underwent 5 cycles of chemotherapy using the P-GEMOX regimen according to the NCCN guidelines for the treatment of ENKTCL. Currently, the patient’s symptoms have improved. She is still alive and will continue to receive further treatment.
Conclusion
We report a rare case of primary pulmonary ENKTCL and summarise previous reported articles. If a patient presents with persistent cough, sputum, and fever, the possibility of this disease should not be ignored. Because of the non-specific clinical symptoms and imaging findings, the correct diagnosis of primary pulmonary ENKTCL depends on histopathology and immunopathological data. Currently, therapeutic strategies are based on conventional ENKTCL treatment, but the prognosis of this disease originating from the lung is poor. Based on the NCCN guidelines, patients with extranasal involvement of NK/T-cell lymphoma should be treated with haematopoietic stem cell transplantation (HSCT) if they are in good health. However, because our patient was older and generally in poor condition (Eastern Cooperative Oncology Group (ECOG) score > 2), we did not recommend HSCT [16].
References
Kwong YL, Kim WS, Lim ST, Kim SJ, Tang T, Tse E, Leung AY, Chim CS (2012) SMILE for natural killer/T-cell lymphoma: analysis of safety and efficacy from the Asia lymphoma study group. Blood 120:2973–2980. https://doi.org/10.1182/blood-2012-05-431460
Tse E, Kwong YL (2013) How I treat NK/T-cell lymphomas. Blood 121:4997–5005. https://doi.org/10.1182/blood-2013-01-453233
Lee S, Shin B, Yoon H, Lee JY, Chon GR (2016) A case of primary pulmonary NK/T cell lymphoma presenting as pneumonia. Respir Med Case Rep 17:1–4. https://doi.org/10.1016/j.rmcr.2015.11.003
Zhou P, Fan S, Dai Y, Tang F, Zhang H (2017) Case report primary pulmonary NK/T cell lymphoma: report of a rare entity and review of literature. Int J Clin Exp Pathol 10:3570–3576
Lee BH, Kim SY, Kim MY, Hwang YJ, Han YH, Seo JW, Kim YH, Cha SJ, Hur G (2006) CT of nasal-type T/NK cell lymphoma in the lung. J Thorac Imaging 21:37–39. https://doi.org/10.1097/01.rti.0000179472.46877.28
Gong L, Wei LX, Huang GS, Zhang WD, Wang L, Zhu SJ, Han XJ, Yao L, Lan M, Li YH, Zhang W (2013) Identification of genuine primary pulmonary NK cell lymphoma via clinicopathologic observation and clonality assay. Diagn Pathol 8:140–147. https://doi.org/10.1186/1746-1596-8-140
Laohaburanakit P, Hardin KA (2006) NK/T cell lymphoma of the lung: a case report and review of literature. Thorax 61:267–270. https://doi.org/10.1136/thx.2004.025767
Chien CC, Lee HS, Lin MH, Hsieh PP (2016) Primary extranodal natural killer/T-cell lymphoma of bronchus and lung: a case report and review of literature. Thorac Cancer 7:140–144. https://doi.org/10.1111/1759-7714.12254
Liu CH, Wang HH, Perng CL, Peng CK, Chian CF, Shen CH (2014) Primary extranodal NK/T-cell lymphoma of the lung: mimicking bronchogenic carcinoma. Thorac Cancer 5:93–96. https://doi.org/10.1111/1759-7714.12021
Song M, Kim JY, Choi JS, Yoon B, Kim M, Kim SJ, Kim SY (2017) Primary pulmonary extranodal natural killer/T-cell lymphoma, nasal type presenting as diffuse ground glass opacities: a case report. J Korean Med Sci 32:1727–1730. https://doi.org/10.3346/jkms.2017.32.10.1727
Oshima K, Tanino Y, Sato S, Inokoshi Y, Saito J, Ishida T, Fukuda T, Watanabe K, Munakata M (2012) Primary pulmonary extranodal natural killer/T-cell lymphoma: nasal type with multiple nodules. Eur Respir J 40:795–798. https://doi.org/10.1183/09031936.00123911
Zhang J, Wang M, Yang X, Liu C, He X (2017) Primary pulmonary extranodal NK/T-cell lymphoma of nasal type misdiagnosed as pneumonia: a case report and literature review. Medicine (Baltimore) 96:e8914. https://doi.org/10.1097/md.0000000000008914
Qiu Y, Hou J, Hao D, Zhang D (2018) Primary pulmonary NK/T-cell lymphoma: a case report and literature review. Mol Clin Oncol 8:753–756. https://doi.org/10.3892/mco.2018.1613
Gui W, Yang B, Shen Q, Bai M, Wang J, Guan T, Zhao J, Wang J, Su L (2015) Successful treatment with L-asparaginase-based regimen for primary pulmonary NK/T cell lymphoma: a case report and review of the literature. Clin Respir J 9:493–496. https://doi.org/10.1111/crj.12156
Davis BW, Beasley MB, Dua S (2010) Primary pulmonary natural killer/T-Cell lymphoma presenting as nonresolving pneumonia. Chest 138:18A. https://doi.org/10.1378/chest.10067
National Comprehensive Cancer Network (2016) NCCN clinical practice guidelines in oncology. The National Comprehensive Cancer Network (NCCN), Plymouth Meeting, PA
van Dongen JJ, Langerak AW, Bruggemann M et al (2003) Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: report of the BIOMED-2 concerted action BMH4-CT98-3936. Leukemia 17:2257–2317. https://doi.org/10.1038/sj.leu.2403202
Devins K, Schuster SJ, Caponetti GC, Bogusz AM (2018) Rare case of low-grade extranodal NK/T-cell lymphoma, nasal type, arising in the setting of chronic rhinosinusitis and harboring a novel N-terminal KIT mutation. Diagn Pathol 13:92–97. https://doi.org/10.1186/s13000-018-0765-1
Tse E, Kwong YL (2017) The diagnosis and management of NK/T-cell lymphomas. J Hematol Oncol 10:85. https://doi.org/10.1186/s13045-017-0452-9
Funding
We are supported by the University Natural Science Key Project of Anhui Province (KJ2018A0269).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Informed consent
Informed consent was obtained from all participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, C., Lu, Z., Bai, X. et al. Primary pulmonary extranodal NK/T cell lymphoma of an elderly adult: a case report and literature review. J Hematopathol 12, 207–211 (2019). https://doi.org/10.1007/s12308-019-00380-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-019-00380-w