Abstract
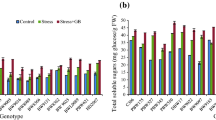
Faba bean (Vicia faba L.) is the major food legume crop in Tunisia. However, its growth and yield is strongly affected by water‐limited environments. In this study, osmotic stress exhibited a negative effect on Bachar and Badii cultivar. Nevertheless, the deteriorating effects of osmotic stress were relatively low on studied parameters of Bachar due to its better efficiency to reduce oxidative damage by increasing enzymatic activities such as catalase (CAT), superoxide dismutase (SOD) and ascorbate peroxidase (APX), accumulation of total chlorophyll (Chlt), soluble sugars and leaf relative water content (RWC). GC–MS analysis determined a total of 11 soluble carbohydrates induced by osmotic stress and differentially accumulated in the both cultivars. Bachar showed elevated levels of mannose, glucose, galactose, ribose, rhamnose and myo-inositol which might help to maintain osmotic adjustment, membranes and proteins protection from the damaging effect of reactive oxygen species. Sugar metabolism related genes (VfNINV3, VfPHS2, VfFRK4, VfHXK1, VfGPI1, VfSTP1.1, VfpGlcT1.1, VfSTP5.1, VfpGlcT1.2, VfSWEET2.1, VfVINV2, VfSUS1, VfPGM1, VfSUT1.1, VfGPT1, VfSPS1, VfSPP1, VfPHS1, VfSUT4.1 and VfTMT1.1) were differentially expressed in both cultivars demonstrating their important roles in sugar accumulation. Most of these genes were upregulated in the leaves of Bachar under moderate and severe stress, which could lead to increase glycolysis and tricarboxylic acid cycle in order to accelerate energy production, necessary to increase osmotic regulation and consequently enhancing the osmotic stress tolerance in that cultivar. Overall, sugars accumulation ability can be used as a useful indicator for the osmotic stress tolerant potential in faba bean breeding programs.





Similar content being viewed by others
References
Abid G, Hessini K, Aouida M, Aroua I, Baudoin JP, Muhovski Y, Mergeai G, Sassi K, Machraoui M, Souissi F, Jebara M (2017) Agro-physiological and biochemical responses of faba bean (Vicia faba L. var. ‘minor’) genotypes to water deficit stress. Biotechnol Agron Soc Environ 21:146–159
Abid M, Ali S, Qi KL, Zahoor R, Tian Z, Jiang D, Snider JL, Da T (2018) Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci Rep 8:4615. https://doi.org/10.1038/s41598-018-21441-7
Ali HM, Siddiqui MH, Al-Whaibi MH, Basalah MO, Sakran AM, El-Zaidy M (2013) Effect of proline and abscisic acid on the growth and physiological performance of faba bean under water stress. Pak J Bot 45:933–940
Aliche EB, Theeuwena TPJM, Oortwijn M, Visser RGF, van der Linden CG (2020) Carbon partitioning mechanisms in POTATO under drought stress. Plant Physiol Biochem 146:211–219
Amede T, Schubert S (2003) Mechanisms of drought resistance in seed legumes. I.-Osmotic adjustment. Ethiop J Sci 26:37–46
Andrade ER, Ribeiro VN, Azevedo CVG, Chiorato AF, Williams TCR, Carbonell SAM (2016) Biochemical indicators of drought tolerance in the common bean (Phaseolus vulgaris L.). Euphytica 210:277–289
Arabzadeh N (2012) The effect of drought stress on soluble carbohydrates (sugars) in two species of Haloxylon persicum and Haloxylon aphyllum. Asian Journal of Plant Sciences 11:44–51
Bai T, Li Z, Song C, Song S, Jiao J, Liu Y, Dong Z, Zheng X (2019) Contrasting drought tolerance in two apple cultivars associated with difference in leaf morphology and anatomy. American Journal of Plant Sciences 10:709–722
Barrs HD, Weatherleyt PE (1962) A re-examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci 15:413–428
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bogdan J, Zagdanska B (2006) Changes in the pool of soluble sugars induced by dehydration at the heterotrophic phase of growth of wheat seedlings. Plant Physiol Biochem 44:787–794
Bohnert HJ, Jensen RG (1996) Strategies for engineering water-stress tolerance in plants. Trends Biotechnol 14:89–97
Bradford MM (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116
Cakmak I, Marschner H (1992) Magnesium deficiency and highlight intensity enhance activities of superoxide dismutase ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol 98:1222–1227
Claeyssen E, Dorion S, Clendenning A, He JZ, Wally O, Chen J, Auslender EL, Moisan MC, Jolicoeur M, Rivoal J (2013) The futile cycling of hexose phosphates could account for the fact that hexokinase exerts a high control on glucose phosphorylation but not on glycolytic rate in transgenic potato (Solanum tuberosum) roots. PLoS ONE 8:e53898. https://doi.org/10.1371/journal.pone.0053898
Del Longo OT, Gonzalez CA, Pastori GM, Trippi VS (1993) Antioxidant defenses under hyperoxygenic and hyperosmotic conditions in leaves of two lines of maize with differential sensitivity to drought. Plant Cell Physiol 34:1023–1028
Dien DC, Mochizuki T, Yamakaw T (2019) Effect of various drought stresses and subsequent recovery on proline, total soluble sugar and starch metabolisms in rice (Oryza sativa L.) varieties. Plant Prod Sci 22:530–545
Du Y, Zhao Q, Chen L, Yao X, Zhang W, Zhang B, Xie F (2020) Effect of drought stress on sugar metabolism in leaves and roots of soybean seedlings. Plant Physiol Biochem 146:1–12
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Durand M, Porcheron B, Hennion N, Maurousset L, Lemoine R, Pourtau N (2016) Water deficit enhances C export to the roots in Arabidopsis thaliana plants with contribution of sucrose transporters in both shoot and roots. Plant Physiol 170:1460–1479
El Ayed M, Kadri S, Smine S, Elkahoui S, Limam F, Aouani E (2017) Protective effects of grape seed and skin extract against high-fat-diet-induced lipotoxicity in rat lung. Lipids Health Dis 16:174. https://doi.org/10.1186/s12944-017-0561-z
Fabregas N, Fernie AR (2019) The metabolic response to drought. J Expt Bot 70:1077–1085
Fang Y, Xiong L (2015) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 72:673–689
FAOSTAT (2018) http://www.fao.org/faostat/en/#data/QC
Ferner E, Rennenberg H, Kreuzwieser J (2012) Effect of flooding on C metabolism of flood-tolerant (Quercus robur) and non-tolerant (Fagus sylvatica) tree species. Tree Physiol 32:135–145
Guo R, Shi LX, Jiao Y, Li MX, Zhong XL, Gu FX, Liu Q, Xia X, Li HR (2018) Metabolic responses to drought stress in the tissues of drought-tolerant and drought-sensitive wheat genotype seedlings. AOB Plants. https://doi.org/10.1093/aobpla/ply016
Gupta A, Kaur N (2005) Sugar signalling and gene expression in relation to carbohydrate metabolism under abiotic stresses in plants. J Biosci 30:761–776
Hayano-Kanashiro C, Calderón-Vázquez C, Ibarra-Laclette E, Herrera-Estrella L, Simpson J (2009) Analysis of gene expression and physiological responses in three Mexican maize landraces under drought stress and recovery irrigation. PLoS ONE 4:e7531. https://doi.org/10.1371/journal.pone.0007531
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Station 347:1–32
Hou J, Huang X, Sun W, Du C, Wang C, Xie Y, Ma Y, Ma D (2018) Accumulation of water-soluble carbohydrates and gene expression in wheat stems correlates with drought resistance. J Plant Physiol 231:182–191
Jiang Y, Huang B (2001) Drought and heat stress injury to two cool-season turfgrasses in relation to antioxidant metabolism and lipid peroxidation. Crop Sci 41:436–442
Jing L, Mei-hong S, Qi-jun M, Hui K, Ya-jing L, Yu-jin H, Chun-xiang Y (2019) MdSWEET17, a sugar transporter in apple, enhances drought tolerance in tomato. J Integr Agric 18:2041–2051
Kang Z, Babar MA, Khan N, Guo J, Khan J, Islam S, Shrestha S, Shahi D (2019) Comparative metabolomic profiling in the roots and leaves in contrasting genotypes reveals complex mechanisms involved in post-anthesis drought tolerance in wheat. PLoS ONE 14:e0213502
Karkanis A, Ntatsi G, Lepse L, Fernández JA, Vågen IM, Rewald B, Alsina I, Kronberga A, Balliu A, Olle M, Bodner G, Dubova L, Rosa E, Savvas D (2018) Faba bean cultivation–revealing novel managing practices for more sustainable and competitive European cropping systems. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01115
Khan HR, Paull JG, Siddique KHM, Stoddard FL (2010) Faba bean breeding for drought-affected environments: a physiological and agronomic perspective. Field Crops Res 115:279–286
Khan MS, Ahmad D, Khan MA (2015) Utilization of genes encoding osmoprotectants in transgenic plants for enhanced abiotic stress tolerance. Electron J Biotechnol 18:257–266
Kholová J, Hash CT, Kakkera A, Kočová M, Vadez V (2010) Constitutive water conserving mechanisms are correlated with the terminal drought tolerance of pearl millet (Pennisetum glaucum (L.) R. Br.). J Exp Bot 61:369–377
Kozminska A, Wiszniewska A, Hanus-Fajerska E, Boscaiu M, Al Hassan M, Halecki W, Vicente O (2019) Identification of salt and drought biochemical stress markers in several Silene vulgaris populations. Sustainability 11:800. https://doi.org/10.3390/su11030800
Krasensky J, Jonak C (2012) Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J Exp Bot 63:1593–1608
Kunz HH, Hausler RE, Fettke J, Herbst K, Niewiadomski P, Gierth M, Bell K, Steup M, Flugge UI, Schneider A (2010) The role of plastidial glucose-6-phosphate/phosphate translocators in vegetative tissues of Arabidopsis thaliana mutants impaired in starch biosynthesis. Plant Biol 12:115–128
Laxa M, Liebthal M, Telman W, Chibani K, Dietz KJ (2019) The role of the plant antioxidant system in drought tolerance. Antioxidants 8:94. https://doi.org/10.3390/antiox8040094
Legay S, Lefevre I, Lamoureux D, Barreda C, Luz RT, Gutierrez R, Quiroz R, Hoffmann L, Hausman JF, Bonierbale M, Evers D, Schafleitner R (2011) Carbohydrate metabolism and cell protection mechanisms differentiate drought tolerance and sensitivity in advanced potato clones (Solanum tuberosum L). Funct Integr Genom 11:275–291
Lemoine R, La Camera S, Atanassova R, Dédaldéchamp F, Allario T, Pourtau N, Bonnemain JL, Laloi M, Coutos-Thévenot P, Maurousset L, Faucher M, Girousse C, Lemonnier P, Parrilla J, Durand M (2013) Source-to-sink transport of sugar and regulation by environmental factors. Front Plant Sci 4:272
Lenka SK, Katiyar A, Chinnusamy V, Bansal KC (2011) Comparative analysis of drought-responsive transcriptome in Indica rice genotypes with contrasting drought tolerance. Plant Biotechnol J 9:315–327
Li M, Feng F, Cheng L (2012) Expression patterns of genes involved in sugar metabolism and accumulation during apple fruit development. PLoS ONE 7:e33055. https://doi.org/10.1371/journal.pone.0033055
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lin Q, Yang J, Wang Q, Zhu H, Chen Z, Dao Y, Wang K (2019) Overexpression of the trehalose-6-phosphate phosphatase family gene AtTPPF improves the drought tolerance of Arabidopsis thaliana. BMC Plant Biol 19:381. https://doi.org/10.1186/s12870-019-1986-5
Loka DA, Oosterhuis DM (2014) Water-deficit stress effects on pistil biochemistry and leaf physiology in cotton (Gossypium hirsutum L.). S Afr J Bot 93:131–136
Marček T, Hamow KA, Vegh B, Janda T, Darko E (2019) Metabolic response to drought in six winter wheat genotypes. PLoS ONE 14:e0212411. https://doi.org/10.1371/journal.pone.0212411
Medici A, Laloi M, Atanassova R (2014) Profiling of sugar transporter genes in grapevine coping with water deficit. FEBS Lett 588:3989–3997
Mohammadkhani N, Heidari R (2008) Drought-induced accumulation of soluble sugars and proline in two maize varieties. World Appl Sci J 3:448–453
Murray MB, Cape JN, Fowler D (1989) Quantification of frost damage in plant tissues by rates of electrolyte leakage. New Phytol 113:307–311
Muscolo A, Sidari M, Anastasi U, Santonoceto C, Maggio A (2014) Effect of PEG-induced drought stress on seed germination of four lentil genotypes. J Plant Interact 9:354–363
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Okunlola GO, Akinwale RO, Adelusi AA (2016) Proline and soluble sugars accumulation in three pepper species (Capsicum spp) in response to water stress imposed at different stages of growth. Sci Cold Arid Reg 8:205–211
Osakabe Y, Yamaguchi-Shinozaki K, Shinozaki K, Tran LSP (2014) ABA control of plant macroelement membrane transport systems in response to water deficit and high salinity. New Phytol 202:35–49
Ouji A, Naouari M, Mouelhi M, Ben Younes M (2017) Yield and yield components of faba bean (Vicia faba L.) as influenced by supplemental irrigation under semi-arid region of Tunisia. World J Agric Res 5:52–57
Polle A, Otter T, Seifert F (1994) Apoplastic peroxidases and lignification in needles of norway spruce Picea abies L. Plant Physiol 106:53–60
Rolland F, Baena-Gonzalez E, Sheen J (2006) Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu Rev Plant Biol 57:675–709
Rosa M, Prado C, Podazza G, Interdonato R, González JA, Hilal M, Prado FE (2009) Soluble sugars-Metabolism, sensing and abiotic stress. Plant Signal Behav 4:388–393
Saglam A, Terzi R, Demiralay M (2014) Effect of polyethylene glycol induced drought stress on photosynthesis in two chickpea genotypes with different drought tolerance. Acta Biol Hung 65:178–188
Sallam A, Alqudah AM, Dawood MFA, Baenziger PS, Börner A (2019) Drought stress tolerance in wheat and barley: advances in physiology, breeding and genetics research. Int J Mol Sci 20:3137. https://doi.org/10.3390/ijms20133137
Sami F, Yusuf M, Faizan M, Faraz A, Hayat S (2016) Role of sugars under abiotic stress. Plant Physiol Biochem 109:54–61
Sarkar KK, Mannan MA, Haque MM, Ahmed JU (2015) Physiological basis of water stress tolerance in soybean. Bangladesh Agron J 18:71–78
Sharma A, Shahzad B, Kumar V, Kohli SK, Sidhu GPS, Bali AS, Handa N, Kapoor D, Bhardwaj R, Zheng B (2019) Phytohormones regulate accumulation of osmolytes under abiotic stress. Biomolecules 9:285. https://doi.org/10.3390/biom9070285
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Sheen J, Zhou L, Jang JC (1999) Sugars as signaling molecules. Curr Opin Plant Biol 2:410–418
Siddiqui MH, Al-Khaishany MY, Al-Qutami MA, Al-Whaibi MH, Grover A, Ali HM, Al-Wahibi MS, Bukhari NA (2015) Response of different genotypes of faba bean plant to drought stress. Int J Mol Sci 16:10214–10227
Stein O, Granot D (2019) An overview of sucrose synthases in plants. Front Plant Sci 10:95. https://doi.org/10.3389/fpls.2019.00095
Thalmann M, Santelia D (2017) Starch as a determinant of plant fitness under abiotic stress. New Phytol 214:943–951
Trouvelot S, Héloir MC, Poinssot B, Gauthier A, Paris F, Guillier C, Combier M, Trdá L, Daire X, Adrian M (2014) Carbohydrates in plant immunity and plant protection: roles and potential application as foliar sprays. Front Plant Sci 5:592. https://doi.org/10.3389/fpls.2014.00592
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid raintreated bean plants. Plant Sci 151:59–66
Wang W, Zhou H, Ma H, Owiti A, Korban SS, Han Y (2016) Divergent evolutionary pattern of sugar transporter genes is associated with the difference in sugar accumulation between Grasses and Eudicots. Sci Rep 6:29153. https://doi.org/10.1038/srep29153
Wang Z, Wei X, Yang J, Li H, Ma B, Zhang K, Zhang Y, Cheng L, Ma F, Li M (2020) Heterologous expression of the apple hexose transporter MdHT2.2 altered sugar concentration with increasing cell wall invertase activity in tomato fruit. Plant Biotechnol J 18:540–552
Wenzel A, Frank T, Reichenberger G, Herz M, Engel KH (2015) Impact of induced drought stress on the metabolite profiles of barley grain. Metabolomics 11:454–467
Xue GP, McIntyre CL, Glassop D, Shorter R (2008) Use of expression analysis to dissect alterations in carbohydrate metabolism in wheat leaves during drought stress. Plant Mol Biol 67:197–214
Xu W, Cui K, Xu A, Nie L, Huang J, Peng S (2015) Drought stress condition increases root to shoot ratio via alteration of carbohydrate partitioning and enzymatic activity in rice seedlings. Acta Physiol Plant 37:1–11
Yang DL, Jing RL, Chang XP, Li W (2007) Identification of quantitative trait loci and environmental interactions for accumulation and remobilization of water-soluble carbohydrates in wheat (Triticum aestivum L.) stems. Genetics 176:571–584
Yang D, Zhang J, Li M, Shi L (2017) Metabolomics analysis reveals the salt-tolerant mechanism in Glycine soja. J Plant Growth Regul 36:460–471
Yang J, Zhang J, Li C, Zhang Z, Ma F, Li M (2019) Response of sugar metabolism in apple leaves subjected to short-term drought stress. Plant Physiol Biochem 141:164–171
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Ghouili, E., Sassi, K., Jebara, M. et al. Physiological responses and expression of sugar associated genes in faba bean (Vicia faba L.) exposed to osmotic stress. Physiol Mol Biol Plants 27, 135–150 (2021). https://doi.org/10.1007/s12298-021-00935-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-021-00935-1