Abstract
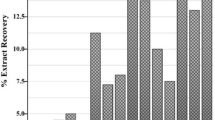
The Nigella sativa pharmacological properties are mainly ascribed to its volatile oil, of which thymoquinone is an important bioactive component. Surprisingly, till date, no standard formulation or thymoquinone rich N. sativa extract is under clinical use probably due to its poor extraction and lesser stability in the already used solvents. In the present investigation solubility, extraction, percent composition and total antioxidant activity from the seeds of N. sativa was explored using five solvents. An HPLC method was standardized in an isocratic system (C-18 column, flow rate of 1.0 ml/min, mobile phase—water:methanol: 30:70, detection wavelength—254 nm, retention time—8.77 min) for quantification of thymoquinone. To further confirm the presence of thymoquinone in the respective extracts absorbance spectra analysis has been carried out and compared with pure thymoquinone. Additionally total antioxidant activity of Nigella sativa extracts has been evaluated using ascorbic acid as standard. Our results showed maximum percentage yield in aqueous extract while methanol having the least yield and the ethanol, benzene and hexane extracts exhibited moderate yields. A linear standard calibration curve of thymoquinone showed R2 as 0.999 and % RSD as 7.166. The HPLC analysis revealed maximum percentage composition of thymoquinone in the benzene extract, whereas in the hexane and methanol extracts the content was less. Aqueous and ethanol extracts displayed insignificant thymoquinone content. Absorbance spectra analysis confirms the presence of thymoquinone peak in the benzene, hexane and methanol extracts while aqueous and ethanol extracts showed minimal absorbance. Maximum total antioxidant activity was observed in the aqueous extract while minimum was observed in the methanolic extract. Weak positive (+ 0.3676) correlation was established between percent composition of thymoquinone and antioxidant activity among different extracts indicating that thymoquinone may not be the only factor for antioxidant activity, but other phytochemicals might also contribute. However, we for the first time demonstrated that the benzene extract of N. sativa has better solubility and percent composition of thymoquinone as compared to other solvents. It can be concluded that the solubility, differential composition of bioactive components among these extracts may have diverse effects on the total antiradical activity. Thus, our study provides insights on optimization and standardization of bioactive rich formulation of N. sativa.







Similar content being viewed by others
References
Abdelwahab SI, Sheikh BY, Taha MM, How CW, Abdullah R, Yagoub U, El-Sunousi R, Eid EE (2013) Thymoquinone-loaded nanostructured lipid carriers: preparation, gastroprotection, in vitro toxicity, and pharmacokinetic properties after extravascular administration. Int J Nanomed 8:2163–2172
Alam S, Khan ZI, Mustafa G, Kumar M, Islam F, Bhatnagar A, Ahmad FJ (2012) Development and evaluation of thymoquinone-encapsulated chitosan nanoparticles for nose-to-brain targeting: a pharmacoscintigraphic study. Int J Nanomed 7:5705–5718
Al-Ani NK (2008) Thymol production from callus culture of Nigella sativa L. Plant Tissue Cult Biotechnol 18(2):181–185
Al-Said M, El-Olemy MM, Elhag HM (2002) Biotechnological production of biologically active metabolites by plant cell culture techniques. KACST, Riyadh, pp 218–256
Ashraf SS, Madduri VR, Fatima SK, Shahnaz Q, Al-Marzouqi AH, Irwin S, Chandranath Abdu A (2011) Nigella sativa extract as a potent antioxidant for petrochemical-induced oxidative stress. J Chromatogr Sci 49:321–326
Badary OA, Taha RA, Gamal El-Din AM, Abdel-Wahab MH (2003) Thymoquinone is a potent superoxide anion scavenger. Drug Chem Toxicol 26:87–98
Banerjee S, Padhye S, Azmi A, Wang Z, Philip PA, Kucuk O, Sarkar FH, Mohammad RM (2010) Review on molecular and therapeutic potential of thymoquinone in cancer. Nutr Cancer 62:938–946
Basha LI, Abou MSR, Aboul-Enein HY (2006) TLC Assay of thymoquinone in black seed oil (Nigella sativa linn) and identification of dithymoquinone and thymol. J Liq Chromatogr 18(1):105–115
Belete Y, Dagne E (2014) HPTLC assay of thymoquinone in black seed and black seed oil (Nigella sativa linn) and identification of thymoquinone conversion with UV-Vis. J Drug Deliv Ther 4(4):1–5
Burits M, Bucar F (2000) Antioxidant activity of Nigella sativa essential oil. Phytother Res 14:323–328
Cacig S, Szabo MR, Lupea AX, Ardelean A (2005) Determination of the antioxidant activity of Ziziphus jujuba and Hydrangea incognita aqueous extracts. Studia Univ Vasile Goldis Ser st Vietii 15:69–72
Cardoso T, Galhano CIC, Ferreira Marques MF, da Silva AM (2012) Thymoquinone β-cyclodextrin nanoparticles system: a preliminary study. Spectroscopy 27:329–336
Dergarabetian EM, Ghattass KI, El-Sitt SB, Al-Mismar RM, El-Baba CO, Itani WS, Melhem NM, El-Hajj HA, Bazarbachi AA, Schneider-Stock R (2013) Thymoquinone induces apoptosis in malignant T-cells via generation of ROS. Front Biosci (Elite Ed) 5:706–719
El-Tahir K, El-Din H, Bakeet DM (2006) The black seed Nigella sativa Linnaeus—a mine for multi cures: a plea for urgent clinical evaluation of its volatile oil. J Taibah Univ Med Sci 1(1):1–19
Gali-Muhtasib H, Kuester D, Mawrin C, Bajbouj K, Diestel A, Ocker M, Habold C, Foltzer-Jourdainne C, Schoenfeld P, Peters B (2008) Thymoquinone triggers inactivation of the stress response pathway sensor CHEK1 and contributes to apoptosis in colorectal cancer cells. Cancer Res 68:5609–5618
Ganea GM, Fakayode SO, Losso JN, van Nostrum CF, Sabliov CM, Warner IM (2010) Delivery of phytochemical thymoquinone using molecular micelle modified poly(D, L lactide-coglycolide) (PLGA) nanoparticles. Nanotechnology 21:1–10
Ghada MH, Randa A, Abdel S, Rabab MS, Mostafa KM (2013) HPLC–DAD determination of seven antioxidants and caffeine in different phytopharmaceuticals. J Chromatogr Sci 52:1–7
Ghosheh OA, Houdi AA, Crooks PA (1999) High performance liquid chromatographic analysis of the pharmacologically active quinones and related compounds in the oil of the black seed (Nigella sativa L.). J Pharm Biomed Anal 19:757–762
Gismondi A, Di Marco G, Canuti L, Canini A (2017) Antiradical activity of phenolic metabolites extracted from grapes of white and red Vitis vinifera L. cultivars. Vitis 56:19–26
Guleria S, Tiku AK, Singh G, Koul A, Gupta S, Rana S (2013) In vitro antioxidant activity and phenolic contents in methanol extracts from medicinal plants. J Plant Biochem Biotechnol 22(1):9–15
Helena S, Marie S, Martin T (2015) Supercritical CO2 extraction of volatile thymoquinone from Monarda didyma and M. fistulosa herbs. J Supercrit Fluids. https://doi.org/10.1016/j.supflu.2015.01.004
Houghton PJ, Zarka R, de las Heras B, Hoult JR (1995) Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Med 61:33–36
Impei S, Gismondi A, Canuti L, Canini A (2015) Metabolic and biological profile of autochthonous Vitis vinifera L. ecotypes. Food Funct 6(5):1526–1538
Iqbal MS, Ansari MI, Ahmad I, Pandey B (2015) Morpho-physiological characterization of seeds and seedlings of Nigella sativa Linn.: study on Indian germplasm. Int Res J Biol Sci 4(4):38–42
Iqbal Z, Iqbal MS, Mishra K (2017) Screening of antioxidant property in medicinal plants belonging to the family Apocynaceae. Asian J Pharm Clin Res 10(12):415–418
Ismail M, Al-Naqeep G, Chan KW (2010) Nigella sativa thymoquinone-rich fraction greatly improves plasma antioxidant capacity and expression of antioxidant genes in hypercholesterolemic rats. Free Radic Biol Med 48(5):664–672
Johnson MA, Krishnaveni E (2012) UV-VIS spectroscopic and HPLC studies on Dictyota bartayresiana lamour. Asian Pac J Trop Biomed S5:14-S518
Jumah MMS, Sajid A, Huixia L, Jianping Z (2014) Aqueous solubility and degradation kinetics of the phytochemical anticancer thymoquinone; probing the effects of solvents, pH and light. Molecules 19(5):5925–5939
Khader M, Bresgen N, Eckl PM (2009) In vitro toxicological properties of thymoquinone. Food ChemToxicol 47(1):129–133
Khoddami A, Meredith AW, Thomas HR (2013) Techniques for analysis of plant phenolic compounds. Molecules 18:2328–2375
Matsuura HN, Malik S, de Costa F, Yousefzadi M, Mirjalili MH, Arroo R, Bhambra AS, Strnad M, Bonfill M, Fett-Neto AG (2018) Specialized plant metabolism characteristics and impact on target molecule biotechnological production. Mol Biotechnol 60(2):169–183
Ncube B, Staden JV (2015) Tilting plant metabolism for improved metabolite biosynthesis and enhanced human benefit. Molecules 20:12698–12731
Odeh F, Ismail SI, Abu-Dahab R, Mahmoud IS, Bawab A (2012) Thymoquinone in liposomes: a study of loading efficiency and biological activity towards breast cancer. Drug Deliv 19:317–371
Pagola S, Benavente A, Raschi A, Romano E, Molina MAA, Stephens PW (2003) Crystal structure determination of thymoquinone by high-resolution X-ray powder diffraction. AAPS PharmSciTech 5:1–8
Petrucci R, Marrosu G, Astolfi P, Lupidi G, Greci L (2012) Cyclic voltammetry, spectroelectrochemistry and electron spin resonance as combined tools to study thymoquinone in aprotic medium. Electrochim Acta 60:230–238
Rajesh H, Rao SN, Prathima KS, Megha RN, Rejeesh EP, Lovelyn J (2013) Phytochemical analysis of aqueous extract of Ocimum Sanctum Linn. Int J Univers Pharm Bio Sci 2(2):462–468
Rao MV, Al-Mazrouqi AH, Fatima-Shad K, Ashraf S, Adem A (2007) Comparative evaluation of SFE and solvent extraction methods on the yield and composition of Black seeds (Nigella sativa). J Liq Chromatgr Relat Technol 30:2545–2555
Raschi AB, Romano E, Benavente AM, Ben Altabef A, Tuttolomondo ME (2010) Structural and vibrational analysis of thymoquinone. Spectrochim Acta Part A Mol Biomol Spectrosc 77:497–505
Ravindran J, Nairb HB, Sunga B, Prasada S, Tekmalb RR, Aggarwala BB (2010) Thymoquinone poly (lactide-co-glycolide) nanoparticles exhibit enhanced anti-proliferative, anti-inflammatory, and chemosensitization potential. Biochem Pharmacol 79:1640–1647
Salem ML (2005) Immunomodulatory and therapeutic properties of the Nigella sativa L. seed. Int Immunopharmacol 5(13–14):1749–1770
Salmani JMM, Asghar S, Lv H, Zhou J (2014) Aqueous solubility and degradation kinetics of the phytochemical anticancer thymoquinone; probing the effects of solvents, pH and light. Molecules 19:5925–5939
Schneider-Stock R, Fakhoury IH, Zaki AM, El-Baba CO, Gali-Muhtasib HU (2014) Thymoquinone: fifty years of success in the battle against cancer models. Drug Discov Today 19:18–30
Severina II, Severin FF, Korshunova GA, Sumbatyan NV, Ilyasova TM, Simonyan RA, Rogov AG, Trendeleva TA, Zvyagilskaya RA, Dugina VB (2013) In search of novel highly active mitochondria-targeted antioxidants: thymoquinone and its cationic derivatives. FEBS Lett 587:2018–2024
Singh A, Ahmad I, Akhter S, Jain GK, Iqbal Z, Talegaonkar S, Ahmad FJ (2013) Nanocarrier based formulation of thymoquinone improves oral delivery: stability assessment, in-vitro and in-vivo studies. Colloids Surf B 102:822–832
Thabrew MI, Mitry RR, Morsy MA, Hughes RD (2005) Cytotoxic effects of a decoction of Nigella sativa, Hemidesmus indicus and Smilax glabra on human hepatoma HepG2 cells. Life Sci 77:1319–1330
Velho-Pereira RM, Barhate CR, Kulkarni SR, Jagtap AG (2011) Validated high-performance thin-layer chromatographic method for the quantification of thymoquinone in Nigella Sativa extracts and formulations. Phytochem Anal 22:367–373
Woo CC, Kumar AP, Sethi G, Tan KHB (2012) Thymoquinone: potential cure for inflammatory disorders and cancer. Biochem Pharmacol 83:443–451
Zubair H, Khan HY, Sohail A, Azim S, Ullah MF, Ahmad A, Sarkar FH, Hadi SM (2013) Redox cycling of endogenous copper by thymoquinone leads to ROS-mediated DNA breakage and consequent cell death: putative anticancer mechanism of antioxidants. Cell Death Dis 4:660
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors worked for this manuscript declares that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent is not applicable.
Rights and permissions
About this article
Cite this article
Iqbal, M.S., Ahmad, A. & Pandey, B. Solvent based optimization for extraction and stability of thymoquinone from Nigella sativa Linn. and its quantification using RP-HPLC. Physiol Mol Biol Plants 24, 1209–1219 (2018). https://doi.org/10.1007/s12298-018-0593-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-018-0593-5