Abstract
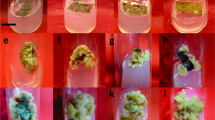
An experiment was designed to evaluate the effect of various adenine derived cytokinins (kinetin and 6-benzylaminopurine) and diphenyl urea cytokinin (thidiazuron) on the postharvest performance of cut scapes of Iris germanica. Flower scapes were harvested with the oldest bud at ‘1 day before anthesis stage’, brought to laboratory under water, cut to a uniform length of 35 cm, divided into three sets viz., kinetin (KIN), 6-benzyl aminopurine (BAP) and thidiazuron (TDZ). Each set of scapes was treated with a particular cytokinin alone or in combination with 0.1 M sucrose. TDZ was effective than KIN and BAP in improving the postharvest life of the I. germanica scapes by 5.4 days as compared to the control (untreated scapes held in distilled water). This was because of the minimum percentage of bud abortion by TDZ application. Cytokinin application resulted in increased antioxidant activity, higher protein and phenolic content, besides a decrease in specific protease activity and α-amino acids in the tepal tissues. Application of TDZ resulted in the maximum increase in the superoxide dismutase, catalase and ascorbate peroxidase activity in the tepal tissues. The scapes treated with BAP and KIN maintained higher carbohydrate content in the tissue samples as compared to control and TDZ treated scapes. TDZ and BAP application resulted in increased membrane stability because of the decreased lipoxygenase activity which prevented membrane lipid peroxidation. Among the cytokinins tested, TDZ proved to be the promising cytokinin in improving the postharvest performance of beautiful flowers of I. germanica scapes.









Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Ahmad SS, Tahir I (2015) Storage protocol for improving the postharvest performance in cut scapes of Iris versicolor. Acta Hortic 1060:71–79
Ahmad SS, Tahir I (2016a) Increased oxidative stress, lipid peroxidation and protein degradation trigger senescence in Iris versicolor L. flowers. Physiol Mol Biol Plants 22(4):507–514
Ahmad SS, Tahir I (2016b) How and why of flower senescence: understanding from models to ornamentals. Ind J Plant Physiol 21(4):446–456
Ahmad SS, Tahir I (2017) Regulatory role of phenols in flower development and senescence in the genus Iris. Ind J Plant Physiol 22(1):135–140
Ahmad SS, Tahir I, Shahri W (2013) Effect of different storage treatments on physiology and postharvest performance in cut scapes of three Iris Species. J Agric Sci Technol 15:323–331
Arrom L, Munne-Bosch S (2012) Hormonal changes during flower development in floral tissues of Lilium. Planta 236:343–354
Axerold B, Chesbrough TM, Laakso S (1981) Lipoxygenase from soybean. In: Lowenstein JM (ed) Methods enzymology. Academic Press, New York, pp 441–451
Bartrina I, Jensen H, Novak O, Strnad M, Werner T, Schmulling T (2017) Gain-of-function mutants of the cytokinin receptors AHK2 and AHK3 regulate plant organ size, flowering time and plant longevity. Plant Physiol. https://doi.org/10.1104/pp.16.01903
Celikel FG, van Doorn WG (1995) Solute leakage, lipid peroxidation, and protein degradation during the senescence of Iris tepals. Plant Physiol 94:515–521
Celikel FG, van Doorn WG (2012) Endogenous ethylene does not regulate opening of unstressed Iris flowers but strongly inhibits it in water-stressed flowers. J Plant Physiol 169:1425–1429
Chen GX, Asada K (1989) Ascorbate peroxidase in tea leaves: occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant Cell Physiol 30:987–998
Cvikrova M, Sukhova LS, Eder J, Korableva NP (1994) Possible involvement of abscisic acid, ethylene and phenolic acids in potato tuber dormancy. Plant Physiol Biochem 32:685–691
Danilova MN, Kudryakova NV, Doroshenko AS, Zabrodin DA, Rakhmankulova ZF, Oelmuller R, Kusnetsov VV (2017) Opposite roles of the Arabidopsis cytokinin receptors AHK2 and AHK3 in the expression of plastid genes and genes for the plastid transcriptional machinery during senescence. Plant Mol Biol 93(5):533–546
Dar RA, Tahir I, Ahmad SS (2014a) Sugars and sugar alcohol have their say in the regulation of flower senescence in Dianthus chinensis L. Sci Hortic 174:24–28
Dar RA, Tahir I, Ahmad SS (2014b) Physiological and biochemical changes associated with flower development and senescence in Dianthus chinensis. Ind J Plant Physiol 19:215–221
Dek MSP, Padmanabhan P, Sherif S, Subramanium J, Paliyath G (2017) Upregulation of phosphatidylinositol 3- Kinase (P13K) enhances ethylene biosynthesis and accelerates flower senescence in transgenic Nicotiana tabacum L. Intl J Mol Sci 18(7):1533
Dhindsa RS, Plumb-Dhindsa D, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101
Ferrante A, Sodi AM, Serra G (2009) Effect of thidiazuron and gibberellic acid on leaf yellowing of cut stock flowers. Cent Eur J Biol 4(4):461–468
Fukuchi-Mizutani M, Ishiguro K, Nakayama T, Utsunomiya Y, Tanaka Y, Kusumi T, Ueda T (2000) Molecular and functional characterization of a rose lipoxygenase cDNA related to flower senescence. Plant Sci 160:129–137
Have AT, Woltering EJ (1997) Ethylene biosynthetic genes are differentially expressed during carnation (Dianthus caryophyllus L.) flower senescence. Plant Mol Biol 34:89–97
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast I. Kinetics and stochiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hunter DA, Steele BC, Reid MS (2002) Identification of genes associated with perianth senescence in daffodil (Narcissus Pseudonarcissus L. “Dutch master”). Plant Sci 163:13–21
Hunter DA, Ferranti A, Vernieri P, Reid MS (2004) Role of abscisic acid in perianth senescence of daffodil (Narcissus Pseudonarcissus “Dutch master”). Physiol Plant 121:313–321
Ichimura K, Shimizu-Yumoto H, Goto R (2009) Ethylene production by the gynoecium and receptacle is associated with sepal abscission in cut Delphinium flowers. Postharvest Biol Technol 52:262–267
Imsabai W, van Doorn WG (2013) Effects of auxin, gibberellin, and cytokinin on petal blackening and flower opening in cut lotus flowers (Nelumbo nucifera). Postharvest Biol Technol 75:54–57
Iqbal N, Khan NA, Ferrante A, Trivellini A, Francini A, Khan MIR (2017) Ethylene role in plant growth, development and senescence. Interaction with other phytohormones. Front Plant Sci 8:475. https://doi.org/10.3389/tpls.2017.004575
Javid MG, Sorooshzadeh A, Sanavy SAMM, Allahdadi I, Moradi F (2011) Effects of the exogenous application of auxin and cytokinin on carbohydrate accumulation in grains of rice under salt stress. Plant Growth Regul 65:305–313
Jones ML, Chaffin GS, Eason JR, Clark DG (2005) Ethylene sensitivity regulates proteolytic activity and cysteine protease gene expression in Petunia corollas. J Exp Bot 56:2733–2744
Lattanzio M, Lattanzio VMT, Cardinali A (2006) Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. Phytochem Adv Res 661:23–67
Lee AK, Rhee SR, Suh JK, Cha HC (2005) Development of floral organ and physiochemical changes of cut Iris hollandica ‘Blue Magic’ according to plant growth regulators and storage temperature. Acta Hortic 673:315–321
Liu C, Li F, Gai S, Zhang Y, Zhah X, Lu X, Zheng G (2016a) Screening and identification of genes associated with flower senescence in tree Peony (Paeonia × Suffnuticosa Andrews) using suppression subtractive hybridization. J Hortic Sci Biotechnol 92(2):146–154
Liu T, Longhurst AD, Talavera-Rauh F, Hokin SA, Barton MK (2016b) The Arabidopsis transcription factor ABIG1 relays ABA signaled growth inhibition and drought induced senescence. eLife 5:1–19
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Lukaszewski TA, Reid MS (1989) Bulb-type flower senescence. Acta Hortic 261:59–62
Macnish AJ, Jiang CZ, Negre-Zakharov F, Reid MS (2010a) Physiological and molecular changes during opening and senescence of Nicotiana mutabilis flowers. Plant Sci 179(3):267–272
Macnish AJ, Jiang CZ, Reid MS (2010b) Treatment with thidiazuron improves opening and vase life of iris flowers. Postharvest Biol Technol 56:77–84
Mittler R, Vanderauwera S, Gollery M, Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Mor Y, Spiegelstein H, Halevy AH (1983) Inhibition of ethylene biosynthesis in carnation petals by cytokinin. Plant Physiol 71:541–546
Mortazavi SN, Talebi SF, Naderi RA, Sharafi Y (2011) Regulation of ethylene biosynthesis by nitric acid and thidiazuron during postharvest of rose. J Med Plant Res 5(20):5177–5183
Mwangi M, Chatterjee SR, Bhattacharjee SK (2003) Changes in the biochemical constituents of “Golden gate” cut rose petals as affected by precooling with ice cold water spray, pulsing and packaging. J Plant Biol 30:95–97
Nelson N (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Nisar S, Tahir I, Ahmad SS (2015) Modulation of flower senescence in Nicotiana plumabinifolia by polyamines. Ind J Plant Physiol 20:186–190
Pak C, van Doorn WG (2005) Delay of Iris flower senescence by protease inhibitors. New Phytol 165:473–480
Panavas T, Rubinstein B (1998) Oxidative events during programmed cell death of daylily (Hemerocallis hybrid) petals. Plant Sci 133:25–138
Petit-Paly G, Franck T, Brisson L, Kevers C, Chenieux C, Rideau M (1999) Cytokinin modulates catalase activity and coumarin accumulation in in vitro cultures of tobacco. J Plant Physiol 155:9–15
Price AM, Orellana DFA, Salleh FM, Stevens R, Acock R, Buchanan-Wollaston V, Stead AD, Rogers HJ (2008) A comparison of leaf and petal senescence in wallflower reveals common and distinct patterns of gene expression and physiology. Plant Physiol 147(4):1898–1912
Radio MC, Arrom L, Puig S, Munne-Bosch S (2017) Hormonal sensitivity decreases during the progression of flower senescence in Lilium longiflorum. J Plant Growth Regul 36(2):402–412
Reid MS, Wu MJ (2018) Ethylene in flower development and senescence. In: Mattoo AK, Suttle JC (eds) The plant hormone ethylene. CRC Press Taylor and Francis Group, Boca Raton, pp 4–32
Rogers HJ (2013) From models to ornamentals: how is flower senescence regulated? Plant Mol Biol 82:563–574
Rosen H (1957) A modified ninhydrin colorimetric analysis for amino acids. Arch Biochem Biophys 67(1):10–15
Saeed T, Hassan I, Abbasi NA, Jilani G (2014) Effect of gibberellic acid on the vase life and oxidative activities in senescing cut gladiolus flowers. Plant Growth Regul 72:89–95
Sairam RK (1994) Effect of moisture stress on physiological activities of two contrasting wheat genotypes. Indian J Exp Biol 32:584–593
Sankhla N, Mackay WA, Davis TD (2005) Effect of thidiazuron on senescence of flowers in cut inflorescences of Lupinus densiflorus Benth. Acta Hortic 669:239–244
Schmitzer V, Veberic R, Osterc G, Stampar F (2010) Color and phenolic content changes during flower development in groundcover rose. J Am Soc Hortic Sci 135(3):195–202
Schnablova R, Synkova H, Vicankova A, Burketova L, Ederc J, Cvikrova M (2006) Transgenic ipt tobacco overproducing cytokinins over accumulates phenolic compounds during in vitro growth. Plant Physiol Biochem 44:526–534
Shahri W, Tahir I (2014) Flower senescence: some molecular aspects. Planta 239:277–297
Shibuya K, Ichimura K (2016) Physiology and molecular biology of flower senescence. In: Pareek S (ed) Postharvest ripening physiology of crops. CRC Press, Boca Raton, pp 109–129
Siranidou E, Kang Z, Buchnauer H (2002) Studies on symptom development, phenolic compounds and morphological defense responses in wheat cultivars differing in resistance to Fusarium head blight. J Phytopathol 150:200–208
Sui S, Luo J, Liu D, Ma J, Men W, Fan L, Bai Y, Li M (2015) Effects of hormone treatments on cut flower opening and senescence in Wintersweet (Chimonanthus praecox). HortScience 50:1365–1369
Swain T, Hillis WE (1959) The phenolic constituents of Prunus domestica I. The quantitative analysis of phenolic constituents. J Sci Food Agric 10(1):63–68
Synkova H, Semoradova S, Schnablova R, Witters E, Husak M, Valcke R (2006) Cytokinin-induced activity of antioxidant enzymes in transgenic Pssu-ipt tobacco during plant ontogeny. Biol Plant 50(1):31–41
Tassoni A, Accettulli P, Bagni N (2006) Exogenous spermidine delays senescence of Dianthus caryophyllus flowers. Plant Biosyst 140:107–114
Tayyab S, Qamar S (1992) A look into enzyme kinetics: some introductory experiments. Biochem Educ 20(2):116–118
Towne G, Owensby C (1983) Cytokinins effect on protein and chlorophyll content of big bluestem leaves. J Range Manag 36(1):75–77
Tripathi SK, Tuteja N (2007) Integrated signalling in flower senescence. Plant Signal Behav 2(6):437–445
Trivellini A, Cocetta G, Vernieri P, Mensuali-Sodi A, Ferrante A (2014) Effect of cytokinins on delaying petunia flower senescence: a transcriptomic approach. Plant Mol Biol 87:169–180
van Doorn WG (2004) Is petal senescence due to sugar starvation? Plant Physiol 134:35–42
van Doorn WG, Woltering EJ (2008) Physiology and molecular biology of petal senescence. J Exp Bot 59(3):453–480
van Doorn WG, Harkema H, Song JS (1995) Water relations and senescence of cut Iris flowers: effects of cycloheximide. Postharvest Biol Technol 5:345–351
van Doorn WG, Sinz A, Tomassen MM (2003) Daffodil flowers delay senescence in cut Iris flowers. Phytochemistry 65:571–577
van Doorn WG, Celikel FG, Pak C, Harkema H (2013) Delay of Iris flower senescence by cytokinins and jasmonates. Physiol Plant 148:105–120
Williamson VG, Hepwonth G (2018) An investigation of ethylene sensitivity in three Australian native of cut flower genera Calothamnus, Grevillea and Philotheca. Sci Hortic 230:149–154
Xu X, Gookin T, Jiang C, Reid MS (2007) Genes associated with opening and senescence of Mirabilis jalapa flowers. J Exp Bot 58:2193–2201
Acknowledgements
The authors thank Prof. S. Farooq for his influence through the opportunities he provided and insights he conveyed. Dr. Syed Sabhi Ahmad thanks University Grants Commission, Govt. of India for providing SRF under (UGC-BSR) SRF scheme.
Author’s contribution
Syed Sabhi Ahmad carried out the experiments, obtained results, analyzed, compiled the data and drafted the manuscript. Prof. Inayatullah Tahir helped in designing the experiment, supervised the laboratory work, took the photographs and edited the manuscript. Arif Shafi Wani, Riyaz Ahmad Dar and Shaziya Nisar helped in statistical analysis of the data and in the laboratory work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors don’t have any conflict of interest regarding this manuscript.
Rights and permissions
About this article
Cite this article
Ahmad, S.S., Tahir, I., Wani, A.S. et al. Adenine type and diphenyl urea derived cytokinins improve the postharvest performance of Iris germanica L. cut scapes. Physiol Mol Biol Plants 24, 1127–1137 (2018). https://doi.org/10.1007/s12298-018-0554-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-018-0554-z