Abstract
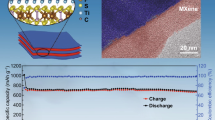
Electrochemical conversion reactions provide more selections for Na-storage materials, but the reaction suffers from low reversibility and poor cyclability. Introducing an electrochemically inactive component is a common strategy, but the effect is quite limited since it could not stabilize the structure during long-term cycling. In this study, a new approach is developed using an amino group-functioned hyperbranched polymer (AHP) as a template and electrode additive for the design of high-performance FeSe2-AHP composite with chemical interaction. The assembled FeSe2-AHP composite nanoneedles were prepared by the selenylation of FeS-AHP composite microflowers and entirely inherit the polymer network from the precursor. The amino groups of AHP in composite coordinate with iron cations to achieve uniform polymer dispersion in the composite, and maintain the molecular level mixed state during the long-term cycling. Moreover, the in-situ constructed uniform 3D elastic polymer network effectively accommodates volume expansion and alleviates nanoparticle aggregation during sodiation/de-sodiation. FeSe2-AHP composite provides a superior rate capability (584.8 mAh·g−1 at 20 A·g−1) and a remarkable cyclability with a capacity retention rate of 93.3% after 2,000 cycles. FeSe2-AHP composite shows a high pseudocapacitive behavior for the abundant nanometer interface established by AHP, enhancing the solid-state Na+ diffusion. The FeSe2-AHP anode is also compatible with Na3V2(PO4)3/C cathode in a full Na-ion battery, which provides a high-power performance (powering 51 LEDs). The work herein highlights an innovative and efficient strategy for conversion-type material design and demonstrates the function of chemical interaction of polymer additive in the enhancement of long-term cyclability for conversion electrode.

Similar content being viewed by others
References
Dunn, B.; Kamath, H.; Tarascon, J. M. Electrical energy storage for the grid: A battery of choices. Science 2011, 334, 928–935.
Etacheri, V.; Marom, R.; Elazari, R.; Salitra, G.; Aurbach, D. Challenges in the development of advanced Li-ion batteries: A review. Energy Environ. Sci. 2011, 4, 3243–3262.
Larcher, D.; Tarascon, J. M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 2015, 7, 19–29.
Yabuuchi, N.; Kubota, K.; Dahbi, M.; Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 2014, 114, 11636–11682.
Kundu, D.; Talaie, E.; Duffort, V.; Nazar, L. F. The emerging chemistry of sodium ion batteries for electrochemical energy storage. Angew. Chem., Int. Ed. 2015, 54, 3431–3448.
Hwang, J. Y.; Myung, S. T.; Sun, Y. K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614.
Zhang, H.; Hasa, I.; Passerini, S. Beyond insertion for Na-ion batteries: Nanostructured alloying and conversion anode materials. Adv. Energy Mater. 2018, 8, 1702582.
Kim, H.; Kim, H.; Ding, Z.; Lee, M. H.; Lim, K.; Yoon, G.; Kang, K. Recent progress in electrode materials for sodium-ion batteries. Adv. Energy Mater. 2016, 6, 1600943.
Kang, H. Y.; Liu, Y. C.; Cao, K. Z.; Zhao, Y.; Jiao, L. F.; Wang, Y. J.; Yuan, H. T. Update on anode materials for Na-ion batteries. J. Mater. Chem. A 2015, 3, 17899–17913.
Wang, A. N.; Hong, W. W.; Yang, L.; Tian, Y.; Qiu, X. J.; Zou, G. Q.; Hou, H. S.; Ji, X. B. Bi-based electrode materials for alkali metal-ion batteries. Small 2020, 16, 2004022.
Cabana, J.; Monconduit, L.; Larcher, D.; Palacín, M. R. Beyond intercalation-based Li-ion batteries: The state of the art and challenges of electrode materials reacting through conversion reactions. Adv. Mater. 2010, 22, E170–E192.
Klein, F.; Jache, B.; Bhide, A.; Adelhelm, P. Conversion reactions for sodium-ion batteries. Phys. Chem. Chem. Phys. 2013, 15, 15876–15887.
Wei, X. J.; Wang, X. P.; Tan, X.; An, Q. Y.; Mai, L. Q. Nanostructured conversion-type negative electrode materials for low-cost and high-performance sodium-ion batteries. Adv. Funct. Mater. 2018, 28, 1804458.
Wu, C.; Dou, S. X.; Yu, Y. The state and challenges of anode materials based on conversion reactions for sodium storage. Small 2018, 14, 1703671.
Zhang, Y.; Xia, X. H.; Liu, B.; Deng, S. J.; Xie, D.; Liu, Q.; Wang, Y. D.; Wu, J. B.; Wang, X. L.; Tu, J. P. Multiscale graphene-based materials for applications in sodium ion batteries. Adv. Energy Mater. 2019, 9, 1803342.
Lu, Y.; Lu, Y. Y.; Niu, Z. Q.; Chen, J. Graphene-based nanomaterials for sodium-ion batteries. Adv. Energy Mater. 2018, 8, 1702469.
Su, D. S.; Schlögl, R. Nanostructured carbon and carbon nanocomposites for electrochemical energy storage applications. ChemSusChem 2010, 3, 136–168.
Nishihara, H.; Kyotani, T. Templated nanocarbons for energy storage. Adv. Mater. 2012, 24, 4473–4498.
Xia, X. H.; Chao, D. L.; Zhang, Y. Q.; Zhan, J. Y.; Zhong, Y.; Wang, X. L.; Wang, Y. D.; Shen, Z. X.; Tu, J. P.; Fan, H. J. Generic synthesis of carbon nanotube branches on metal oxide arrays exhibiting stable high-rate and long-cycle sodium-ion storage. Small 2016, 12, 3048–3058.
Li, T.; Qin, A. Q.; Yang, L. L.; Chen, J.; Wang, Q. F.; Zhang, D. H.; Yang, H. X. In situ grown Fe2O3 single crystallites on reduced graphene oxide nanosheets as high performance conversion anode for sodium-ion batteries. ACS Appl. Mater. Interfaces 2017, 9, 19900–19907.
Zhang, Q. F.; Uchaker, E.; Candelariaza, S. L.; Cao, G. Z. Nanomaterials for energy conversion and storage. Chem. Soc. Rev. 2013, 42, 3127–3171.
Guo, Y. G.; Hu, J. S.; Wan, L. J. Nanostructured materials for electrochemical energy conversion and storage devices. Adv. Mater. 2008, 20, 2878–2887.
Liang, Y. R.; Lai, W. H.; Miao, Z. C.; Chou, S. L. Nanocomposite materials for the sodium-ion battery: A review. Small 2018, 14, 1702514.
Li, T.; Qin, A. Q.; Wang, H. T.; Wu, M. Y.; Zhang, Y. Y.; Zhang, Y. J.; Zhang, D. H.; Xu, F. A high-performance hybrid Mg2+/Li+ battery based on hierarchical copper sulfide microflowers conversion cathode. Electrochim. Acta 2018, 263, 168–175.
Qin, A. Q.; Wu, H.; Chen, J.; Li, T.; Chen, S. H.; Zhang, D. H.; Xu, F. Constructing hyperbranched polymers as a stable elastic framework for copper sulfide nanoplates for enhancing sodium-storage performance. Nanoscale 2019, 11, 7188–7198.
Li, T.; Li, H.; Qin, A. Q.; Wu, H.; Zhang, D. H.; Xu, F. Assembled NiS nanoneedles anode for Na-ion batteries: Enhanced the performance by organic hyperbranched polymer electrode additives. J. Power Sources 2020, 451, 227796.
Zhang, K.; Hu, Z.; Liu, X.; Tao, Z. L.; Chen, J. FeSe2 microspheres as a high-performance anode material for Na-ion batteries. Adv. Mater. 2015, 27, 3305–3309.
Wei, X. J.; Tang, C. J.; An, Q. Y.; Yan, M. Y.; Wang, X. P.; Hu, P.; Cai, X. Y.; Mai, L. Q. FeSe2 clusters with excellent cyclability and rate capability for sodium-ion batteries. Nano Res. 2017, 10, 3202–3211.
Cho, J. S.; Lee, J. K.; Kang, Y. C. Graphitic carbon-coated FeSe2 hollow nanosphere-decorated reduced graphene oxide hybrid nanofibers as an efficient anode material for sodium ion batteries. Sci. Rep. 2016, 6, 23699.
Fan, H. S.; Yu, H.; Zhang, Y. F.; Guo, J.; Wang, Z.; Wang, H.; Zhao, N.; Zheng, Y.; Du, C. F.; Dai, Z. F. et al. 1D to 3D hierarchical iron selenide hollow nanocubes assembled from FeSe2@C core-shell nanorods for advanced sodium ion batteries. Energy Storage Mater. 2018, 10, 48–55.
Ge, P.; Hou, H. S.; Li, S. J.; Yang, L.; Ji, X. B. Tailoring rod-like FeSe2 coated with nitrogen-doped carbon for high-performance sodium storage. Adv. Funct. Mater. 2018, 28, 1801765.
Zhao, F. P.; Shen, S. D.; Cheng, L.; Ma, L.; Zhou, J. H.; Ye, H. L.; Han, N.; Wu, T. P.; Li, Y. G.; Lu, J. Improved sodium-ion storage performance of ultrasmall iron selenide nanoparticles. Nano Lett. 2017, 17, 4137–4142.
Park, J. S.; Jeong, S. Y.; Jeon, K. M.; Kang, Y. C.; Cho, J. S. Iron diselenide combined with hollow graphitic carbon nanospheres as a high-performance anode material for sodium-ion batteries. Chem. Eng. J. 2018, 339, 97–107.
Tang, Y. C.; Zhao, Z. B.; Hao, X. J.; Wei, Y.; Zhang, H.; Dong, Y. F.; Wang, Y. W.; Pan, X.; Hou, Y. N.; Wang, X. Z. et al. Cellular carbon-wrapped FeSe2 nanocavities with ultrathin walls and multiple rooms for ion diffusion-confined ultrafast sodium storage. J. Mater. Chem. A 2019, 7, 4469–4479.
Fan, H. H.; Li, H. H.; Wang, Z. W.; Li, W. L.; Guo, J. Z.; Fan, C. Y.; Sun, H. Z.; Wu, X. L.; Zhang, J. P. Tailoring coral-like Fe7Se8@C for superior low-temperature Li/Na-ion half/full batteries: Synthesis, structure, and DFT studies. ACS Appl. Mater. Interfaces 2019, 11, 47886–47893.
Tian, W. Z.; Ma, W. Z.; Feng, Z. Y.; Tian, F.; Li, H. B.; Liu, J.; Xiong, S. L. Formation of hierarchical Fe7Se8 nanorod bundles with enhanced sodium storage properties. J. Energy Chem. 2020, 44, 97–105.
Chen, S.; Huang, S. Z.; Zhang, Y. F.; Fan, S.; Yan, D.; Shang, Y.; Pam, M. E.; Ge, Q.; Shi, Y. M.; Yang, H. Y. Constructing stress-release layer on Fe7Se8-based composite for highly stable sodium-storage. Nano Energy 2020, 69, 104389.
Qi, S. H.; Mi, L. W.; Song, K. M.; Yang, K. W.; Ma, J. M.; Feng, X. M.; Zhang, J. M.; Chen, W. H. Understanding shuttling effect in sodium ion batteries for the solution of capacity fading: FeS2 as an example. J. Phys. Chem. C 2019, 123, 2775–2782
Yang, D.; Chen, W. H.; Zhang, X. X.; Mi, L. M.; Liu, C. T.; Chen, L. J.; Guan, X. X.; Cao, Y. L.; Shen, C. Y. Facile and scalable synthesis of low-cost FeS@C as long-cycle anodes for sodium-ion batteries. J. Mater. Chem. A 2019, 7, 19709–19718.
Liu, Y.; Fang, Y. J.; Zhao, Z. W.; Yuan, C. Z.; Lou, X. W. A ternary Fe1−xS@porous carbon nanowires/reduced graphene oxide hybrid film electrode with superior volumetric and gravimetric capacities for flexible sodium ion batteries. Adv. Energy Mater. 2019, 9, 1803052.
Zhou, M.; Tao, H. W.; Wang, K. L.; Cheng, S. J.; Jiang, K. Nano-embedded microstructured FeS2@C as a high capacity and cycling-stable Na-storage anode in an optimized ether-based electrolyte. J. Mater. Chem. A 2018, 6, 24425–24432.
Zhang, D. H.; Liu, T. T.; Chen, S. F.; Miao, M. H.; Cheng, J.; Zhang, A. Q.; Chen, S. H. Amino-ended hyperbranched polyamide as template for tuning the morphology of self-assembled ZnS particles. Mater. Chem. Phys. 2016, 184, 162–171.
Fu, T. T.; Chen, Y. Y.; Hao, J. L.; Wang, X. Y.; Liu, G.; Li, Y. G.; Liu, Z.; Cheng, L. Facile preparation of uniform FeSe2 nanoparticles for PA/MR dual-modal imaging and photothermal cancer therapy. Nanoscale 2015, 7, 20757–20768.
Fang, J. Q.; Wang, S. Q.; Li, Z. T.; Chen, H. B.; Xia, L.; Ding, L. X.; Wang, H. H. Porous Na3V2(PO4)3@C nanoparticles enwrapped in three-dimensional graphene for high performance sodium-ion batteries. J. Mater. Chem. A 2016, 4, 1180–1185.
Zhang, Z.; Shi, X. D.; Yang, X.; Fu, Y.; Zhang, K.; Lai, Y. Q.; Li, J. Nanooctahedra particles assembled FeSe2 microspheres embedded into sulfur-doped reduced graphene oxide sheets as a promising anode for sodium ion batteries. ACS Appl. Mater. Interfaces 2016, 8, 13849–13856.
Li, J. C.; Xiao, F.; Zhong, H.; Li, T.; Xu, M. J.; Ma, L.; Cheng, M.; Liu, D.; Feng, S.; Shi, Q. R. et al. Secondary-atom-assisted synthesis of single iron atoms anchored on N-doped carbon nanowires for oxygen reduction reaction. ACS Catal. 2019, 9, 5929–5934.
Muller, G. A.; Cook, J. B.; Kim, H. S.; Tolbert, S. H.; Dunn, B. High performance pseudocapacitor based on 2D layered metal chalcogenide nanocrystals. Nano Lett. 2015, 15, 1911–1917.
Zhao, Y. L.; Cao, X. X.; Fang, G. Z.; Wang, Y. P.; Yang, H. L.; Liang, S. Q.; Pan, A. Q.; Cao, G. Z. Hierarchically carbon-coated Na3V2(PO4)3 nanoflakes for high-rate capability and ultralong cycle-life sodium ion batteries. Chem. Eng. J. 2018, 339, 162–169.
Acknowledgements
We gratefully acknowledge the financial support of Hubei Provincial Natural Science Foundation (Nos. 2019CFB620 and 2019CFB452), Innovation group of Hubei Natural Science Foundation (No. 2018CFA023), and the Fundamental Research Funds for the Central Universities, South-Central University for Nationalities (No. CZY20022).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
12274_2021_3320_MOESM1_ESM.pdf
Enhancing the long-term Na-storage cyclability of conversion-type iron selenide composite by construction of 3D inherited hyperbranched polymer buffering matrix
Rights and permissions
About this article
Cite this article
Li, H., Hu, C., Xia, Y. et al. Enhancing the long-term Na-storage cyclability of conversion-type iron selenide composite by construction of 3D inherited hyperbranched polymer buffering matrix. Nano Res. 14, 3952–3960 (2021). https://doi.org/10.1007/s12274-021-3320-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-021-3320-4