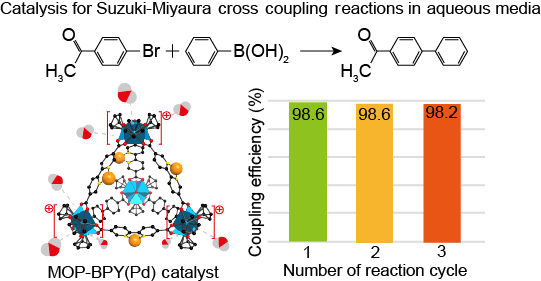
Abstract
The challenge for single-atom catalysts in various C—C cross coupling reaction exists in the development of solid supporting materials. It has been desired to find a supporting material designed in molecular level to anchor a single-atom catalyst and provide high degree of dispersion and substrate access in aqueous media. Here, we prepared discrete cages of metal-organic polyhedra anchoring single Pd atom (MOP-BPY(Pd)) and successfully performed a Suzuki-Miyaura cross coupling reaction with various substrates in aqueous media. It was revealed that each tetrahedral cage of MOP-BPY(Pd) has 4.5 Pd atoms on average and retained its high degree of dispersion up to 3 months in water. The coupling efficiencies of the Suzuki-Miyaura cross coupling reaction exhibited more than 90.0% for various substrates we have tested in the aqueous media, which is superior to those of the molecular Pd complex and metal-organic framework (MOF) anchoring Pd atoms. Moreover, MOP-BPY(Pd) was successfully recovered and recycled without performance degradation.
Similar content being viewed by others
References
Zhang, L. L.; Wang, A. Q.; Miller, J. T.; Liu, X. Y.; Yang, X. F.; Wang, W. T.; Li, L.; Huang, Y. Q.; Mou, C. Y.; Zhang, T. Efficient and durable Au alloyed Pd single-atom catalyst for the ullmann reaction of aryl chlorides in water. ACS Catal.2014, 4, 1546–1553.
Zhang, X. Y.; Sun, Z. C.; Wang, B.; Tang, Y.; Nguyen, L.; Li, Y. T.; Tao, F. F. C—C coupling on single-atom-based heterogeneous catalyst. J. Am. Chem. Soc.2018, 140, 954962.
Fernández, E.; Rivero-Crespo, M. A.; Domínguez, I.; Rubio-Marqués, P.; Oliver-Meseguer, J.; Liu, L. C.; Cabrero-Antonino, M.; Gavara, R.; Hernández-Garrido, J. C.; Boronat, M. et al. Base-controlled Heck, Suzuki, and Sonogashira reactions catalyzed by ligand-free platinum or palladium single atom and sub-nanometer clusters. J. Am. Chem. Soc.2019, 141, 1928–1940.
Yin, L. X.; Liebscher, J. Carbon-carbon coupling reactions catalyzed by heterogeneous palladium catalysts. Chem. Rev.2007, 107, 133–173.
Veerakumar, P.; Thanasekaran, P.; Lu, K. L.; Lin, K. C.; Rajagopal, S. Computational studies of versatile heterogeneous palladium-catalyzed Suzuki, Heck, and Sonogashira coupling reactions. ACS Sustain. Chem. Eng.2017, 5, 8475–8490.
Bernini, R.; Cacchi, S.; Fabrizi, G.; Forte, G.; Petrucci, F.; Prastaro, A.; Niembro, S.; Shafir, A.; Vallribera, A. Perfluoro-tagged, phosphine-free palladium nanoparticles supported on silica gel: Application to alkynylation of aryl halides, Suzuki-Miyaura cross-coupling, and Heck reactions under aerobic conditions. Green Chem.2010, 12, 150–158.
Biffis, A.; Zecca, M.; Basato, M. Palladium metal catalysts in Heck C—C coupling reactions. J. Mol. Catal. A Chem.2001, 173, 249–274.
Suzaki, Y.; Kobayashi, Y.; Tsuchido, Y.; Osakada, K. Pd-catalyzed Sonogashira coupling in aqueous media. Observation of micelles that contain substrates and catalyst. Mol. Catal.2019, 466, 106–111.
Christoffel, F.; Ward, T. R. Palladium-catalyzed heck cross-coupling reactions in water: A comprehensive review. Catal. Lett.2018, 148, 489–511.
Lakshminarayana, B.; Mahendar, L.; Ghosal, P.; Satyanarayana, G.; Subrahmanyam, C. Nano-sized recyclable PdO supported carbon nanostructures for Heck reaction: Influence of carbon materials. ChemistrySelect2017, 2, 2700–2707.
Tran, T. P. N.; Thakur, A.; Trinh, D. X.; Dao, A. T. N.; Taniike, T. Design of Pd@graphene oxide framework nanocatalyst with improved activity and recyclability in Suzuki-Miyaura cross-coupling reaction. Appl. Catal. A: Gen2018, 549, 60–67.
Diyarbakir, S.; Can, H. S.; Metin, O. Reduced graphene oxide-supported CuPd alloy nanoparticles as efficient catalysts for the Sonogashira cross-coupling reactions. ACS Appl. Mater. Interfaces2015, 7, 3199–3206.
Sharavath, V.; Ghosh, S. Palladium nanoparticles on noncovalently functionalized graphene-based heterogeneous catalyst for the Suzuki-Miyaura and Heck-Mizoroki reactions in water. RSC Adv.2014, 4, 48322–48330.
Scheuermann, G M.; Rumi, L.; Steurer, P.; Bannwarth, W.; Mülhaupt, R. Palladium nanoparticles on graphite oxide and its functionalized graphene derivatives as highly active catalysts for the Suzuki-Miyaura coupling reaction. J. Am. Chem. Soc.2009, 131, 8262–8270.
Balaswamy, K.; Pullaiah, P. C.; Srinivas, K.; Rao, M. M. Polystyrene-supported palladium(II) N,N-dimethylethylenediamine complex: A recyclable catalyst for Suzuki-Miyaura cross-coupling reactions in water. Inorg. Chim. Acta2014, 423, 95–100.
Moussa, S.; Siamaki, A. R.; Gupton, B. F.; El-Shall, M. S. Pd-partially reduced graphene oxide catalysts (Pd/PRGO): Laser synthesis of Pd nanoparticles supported on PRGO nanosheets for carbon-carbon cross coupling reactions. ACS Catal.2012, 2, 145–154.
De Castro, K. A.; Rhee, H. Resin-immobilized palladium nanoparticle catalysts for Suzuki-Miyaura cross-coupling reaction in aqueous media. J. Incl. Phenom. Macrocycl. Chem.2015, 82, 13–24.
Chen, L. Y.; Rangan, S.; Li, J.; Jiang, H. F.; Li, Y. W. A molecular Pd(II) complex incorporated into a MOF as a highly active singlesite heterogeneous catalyst for C—Cl bond activation. Green Chem.2014, 16, 3978–3985.
Nagai, D.; Goto, H. Effective heterogeneous catalyst for Suzuki-Miyaura cross-coupling in aqueous media: Melamine cyanurate complex containing Pd species. Bull. Chem. Soc. Jpn.2018, 91, 147–152.
Yang, Y.; Reber, A. C.; Gilliland III, S. E.; Castano, C. E.; Gupton, B. F.; Khanna, S. N. More than just a support: Graphene as a solid-state ligand for palladium-catalyzed cross-coupling reactions. J. Catal.2018, 360, 20–26.
Dighe, M. G.; Lonkar, S. L.; Degani, M. S. Mechanistic insights into palladium leaching in novel Pd/C-catalyzed boron-Heck reaction of arylboronic acid. Synlett2013, 24, 347–350.
Veerakumar, P.; Thanasekaran, P.; Lu, K. L.; Liu, S. B.; Rajagopal, S. Functionalized silica matrices and palladium: A versatile heterogeneous catalyst for Suzuki, Heck, and Sonogashira reactions. ACS Sustain. Chem. Eng.2017, 5, 6357–6376.
MacQuarrie, S.; Horton, J. H.; Barnes, J.; McEleney, K.; Loock, H. P.; Crudden, C. M. Visual observation of redistribution and dissolution of palladium during the Suzuki-Miyaura reaction. Angew. Chem., Int. Ed.2008, 47, 3279–3282.
Richardson, J. M.; Jones, C. W. Strong evidence of solution-phase catalysis associated with palladium leaching from immobilized thiols during Heck and Suzuki coupling of aryl iodides, bromides, and chlorides. J. Catal.2007, 251, 80–93.
Huang, Y. B.; Zheng, Z. L.; Liu, T. F.; Lü, J.; Lin, Z. J.; Li, H. F.; Cao, R. Palladium nanoparticles supported on amino functionalized metal-organic frameworks as highly active catalysts for the Suzuki-Miyaura cross-coupling reaction. Catal. Commun.2011, 14, 27–31.
Kardanpour, R.; Tangestaninejad, S.; Mirkhani, V.; Moghadam, M.; Mohammadpoor-Baltork, I.; Khosropour, A. R.; Zadehahmadi, F. Highly dispersed palladium nanoparticles supported on amino functionalized metal-organic frameworks as an efficient and reusable catalyst for Suzuki cross-coupling reaction. J. Organomet. Chem.2014, 761, 127–133.
Puthiaraj, P.; Ahn, W. S. Highly active palladium nanoparticles immobilized on NH2-MIL-125 as efficient and recyclable catalysts for Suzuki-Miyaura cross coupling reaction. Catal. Commun.2015, 65, 91–95.
Fei, H. H.; Cohen, S. M. A robust, catalytic metal-organic framework with open 2,2’-bipyridine sites. Chem. Commun.2014, 50, 4810–4812.
Nam, D.; Huh, J.; Lee, J.; Kwak, J. H.; Jeong, H. Y.; Choi, K.; Choe, W. Cross-linking Zr-based metal-organic polyhedra via postsynthetic polymerization. Chem. Sci.2017, 8, 7765–7771.
Mollick, S.; Fajal, S.; Mukherjee, S.; Ghosh, S. K. Stabilizing metal-organic polyhedra (MOP): Issues and strategies. Chem. Asian J.2019, 14, 3096–3108.
Wang, W.; Wang, Y. X.; Yang, H. B. Supramolecular transformations within discrete coordination-driven supramolecular architectures. Chem. Soc. Rev.2016, 45, 2656–2693.
Li, J. R.; Zhou, H. C. Bridging-ligand-substitution strategy for the preparation of metal-organic polyhedra. Nat. Chem.2010, 2, 893–898.
Tranchemontagne, D. J.; Ni, Z.; O’Keeffe, M.; Yaghi, O. M. Reticular chemistry of metal-organic polyhedra. Angew. Chem., Int. Ed.2008, 47, 5136–5147.
Lu, Z.; Knobler, C. B.; Furukawa, H.; Wang, B.; Liu, G. N.; Yaghi, O. M. Synthesis and structure of chemically stable metal-organic polyhedra. J. Am. Chem. Soc.2009, 131, 12532–12533.
Xing, W. H.; Li, H. Y.; Dong, X. Y.; Zang, S. Q. Robust multifunctional Zr-based metal-organic polyhedra for high proton conductivity and selective CO2 capture. J. Mater. Chem. A2018, 6, 7724–7730.
Ju, Z. F.; Liu, G. L.; Chen, Y. S.; Yuan, D. Q.; Chen, B. L. From coordination cages to a stable crystalline porous hydrogen-bonded framework. Chem.—Eur. J.2017, 23, 4774–4777.
Liu, G. L.; Ju, Z. F.; Yuan, D. Q.; Hong, M. C. In situ construction of a coordination zirconocene tetrahedron. Inorg. Chem.2013, 52, 13815–13817.
Liu, G. L.; Zeller, M.; Su, K. Z.; Pang, J. D.; Ju, Z. F.; Yuan, D. Q.; Hong, M. C. Controlled orthogonal self-assembly of heterometal-decorated coordination cages. Chem.—Eur. J.2016, 22, 17345–17350.
Lee, H. S.; Jee, S.; Kim, R.; Bui, H. T.; Kim, B.; Kim, J. K.; Park, K. S.; Choi, W.; Kim W.; Choi, K. M. A highly active, robust photocatalyst heterogenized in discrete cages of metal-organic polyhedra for CO2 reduction. Energy Environ. Sci.2020, 13, 519–526
Choi, K. M.; Jeong, H. M.; Park, J. H.; Zhang, Y. B.; Kang, J. K.; Yaghi, O. M. Supercapacitors of nanocrystalline metal-organic frameworks. ACS Nano2014, 8, 7451–7457.
Kang, Y. H.; Liu, X. D.; Yan, N.; Jiang, Y.; Liu, X. Q.; Sun, L. B.; Li, J. R. Fabrication of isolated metal-organic polyhedra in confined cavities: Adsorbents/catalysts with unusual dispersity and activity. J. Am. Chem. Soc.2016, 138, 6099–6102.
Lin, W. G.; Yuan, D. Q.; Yakovenko, A.; Zhou, H. C. Surface functionalization of metal-organic polyhedron for homogeneous cyclopropanation catalysis. Chem. Commun.2011, 47, 4968–4970.
Vardhan, H.; Verpoort, F. Metal-organic polyhedra: Catalysis and reactive intermediates. Adv. Synth. Catal.2015, 357, 1351–1368.
Maity, K.; Karan, C. K.; Biradha, K. Porous metal-organic polyhedral framework containing cuboctahedron cages as SBUs with high affinity for H2 and CO2 sorption: A heterogeneous catalyst for chemical fixation of CO2. Chem.—Eur. J.2018, 24, 10988–10993.
Ahmad, N.; Younus, H. A.; Chughtai, A. H.; Van Hecke, K.; Danish, M.; Gaoke, Z.; Verpoort, F. Development of mixed metal metal-organic polyhedra networks, colloids, and MOFs and their pharmacokinetic applications. Sci. Rep.2017, 7, 832.
Sherwood, J.; Clark, J. H.; Fairlamb, I. J. S.; Slattery, J. M. Solvent effects in palladium catalysed cross-coupling reactions. Green Chem.2019, 21, 2164–2213.
Lima, C. F. R. A. C.; Rodrigues, A. S. M. C.; Silva, V. L. M.; Silva, A. M. S.; Santos, L. M. N. B. F. Role of the base and control of selectivity in the Suzuki-Miyaura cross-coupling reaction. ChemCatChem.2014, 6, 1291–1302.
Zhang, H. C.; Kwong, F. Y.; Tian, Y.; Chan, K. S. Base and cation effects on the Suzuki cross-coupling of bulky arylboronic acid with halopyridines: Synthesis of pyridylphenols. J. Org. Chem.1998, 63, 6886–6890.
Remya, G. S.; Suresh, C. H. Quantification and classification of substituent effects in organic chemistry: A theoretical molecular electrostatic potential study. Phys. Chem. Chem. Phys.2016, 18, 20615–20626.
Littke, A. F.; Dai, C. Y.; Fu, G. C. Versatile catalysts for the Suzuki cross-coupling of arylboronic acids with aryl and vinyl halides and triflates under mild conditions. J. Am. Chem. Soc.2000, 122, 4020–4028.
Watanabe, T.; Miyaura, N.; Suzuki, A. Synthesis of sterically hindered biaryls via the palladium-catalyzed cross-coupling reaction of arylboronic acids or their esters with haloarenes. Synlett1992, 1992, 207–210.
Fournier, J. H.; Maris, T.; Wuest, J. D.; Guo, W. Z.; Galoppini, E. Molecular tectonics. Use of the hydrogen bonding of boronic acids to direct supramolecular construction. J. Am. Chem. Soc.2003, 125, 1002–1006.
Dhakshinamoorthy, A.; Alvaro, M.; Hwang, Y. K.; Seo, Y. K.; Corma, A.; Garcia, H. Intracrystalline diffusion in metal organic framework during heterogeneous catalysis: Influence of particle size on the activity of MIL-100 (Fe) for oxidation reactions. Dalton Trans.2011, 40, 10719–10724.
Acknowledgements
This research was supported by the Basic Science Research Program (No. NRF-2019R1A2C4069764) and by Convergent Technology R&D Program for Human Augmentation (No. 2019M3C1B8077549) through the National Research Foundation of Korea (NRF) funded by Ministry of Science and ICT.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
12274_2020_2885_MOESM1_ESM.pdf
Single-atom Pd catalyst anchored on Zr-based metal-organic polyhedra for Suzuki-Miyaura cross coupling reactions in aqueous media
Rights and permissions
About this article
Cite this article
Kim, S., Jee, S., Choi, K.M. et al. Single-atom Pd catalyst anchored on Zr-based metal-organic polyhedra for Suzuki-Miyaura cross coupling reactions in aqueous media. Nano Res. 14, 486–492 (2021). https://doi.org/10.1007/s12274-020-2885-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-020-2885-7




