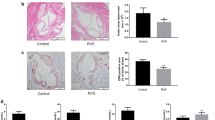
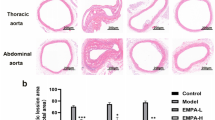
Abstract
It has been shown that SGLT2 suppresses atherosclerosis (AS). Recent studies indicate that autophagy widely participates in atherogenesis. This study aimed to assess the effect of canagliflozin (CAN) on atherogenesis via autophagy. Macrophages and ApoE − / − mice were used in this study. In macrophages, the results showed that CAN promoted LC3II expression and autophagosome formation. Furthermore, the cholesterol efflux assay demonstrated that CAN enhanced cholesterol efflux from macrophages via autophagy, resulting in lower lipid droplet concentrations in macrophages. The western blot revealed that CAN regulated autophagy via the AMPK/ULK1/Beclin1 signaling pathway. CAN resulted in increased macrophage autophagy in atherosclerotic plaques of ApoE − / − mice, confirming that CAN could inhibit the progression of AS via promoting macrophage autophagy. The current study found that CAN reduced the production of atherosclerotic lesions, which adds to our understanding of how SGLT2 inhibitors function to delay the progression of AS.
Graphical abstract






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Abbreviations
- CAD:
-
Coronary artery disease
- AS:
-
Atherosclerosis
- SGLT2:
-
Sodium-Glucose Co-Transporter 2
- ApoE:
-
Apolipoprotein E
- ApoE − / − :
-
Apolipoprotein E deficient
- HFD:
-
High-fat diet
- ox-LDL:
-
Oxidized low-density lipoprotein
- AMPK:
-
Adenosine 5′-monophosphate-activated protein kinase
- ULK1:
-
UNC-51-like autophagy-activating kinase 1
- ATG:
-
Autophagy-related gene
References
Moore KJ, Tabas I. Macrophages in the pathogenesis of atherosclerosis. Cell. 2011;145(3):341–55.
Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999;340(2):115–26.
Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015. 385(9963): 117–71.
Cervelli T, Borghini A, Galli A, Andreassi MG. DNA damage and repair in atherosclerosis: current insights and future perspectives. Int J Mol Sci. 2012;13(12):16929–44.
Rieg T, Masuda T, Gerasimova M, et al. Increase in SGLT1-mediated transport explains renal glucose reabsorption during genetic and pharmacological SGLT2 inhibition in euglycemia. Am J Physiol Renal Physiol. 2014;306(2):F188–93.
Kang A, Jardine MJ. SGLT2 inhibitors may offer benefit beyond diabetes. Nat Rev Nephrol. 2021;17(2):83–4.
Khunti K. SGLT2 inhibitors in people with and without T2DM. Nat Rev Endocrinol. 2021;17(2):75–6.
Cowie MR, Fisher M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat Rev Cardiol. 2020;17(12):761–72.
Xu L, Nagata N, Nagashimada M, et al. SGLT2 inhibition by empagliflozin promotes fat utilization and browning and attenuates inflammation and insulin resistance by polarizing M2 macrophages in diet-induced obese mice. EBioMedicine. 2017;20:137–49.
Xu L, Ota T. Emerging roles of SGLT2 inhibitors in obesity and insulin resistance: focus on fat browning and macrophage polarization. Adipocyte. 2018;7(2):121–8.
Kitada M, Koya D. Autophagy in metabolic disease and ageing. Nat Rev Endocrinol. 2021;17(11):647–61.
Liao X, Sluimer JC, Wang Y, et al. Macrophage autophagy plays a protective role in advanced atherosclerosis. Cell Metab. 2012;15(4):545–53.
Jeong SJ, Lee MN, Oh GT. The role of macrophage lipophagy in reverse cholesterol transport. Endocrinol Metab (Seoul). 2017;32(1):41–6.
Martinet W, De Meyer I, Verheye S, Schrijvers DM, Timmermans JP, De Meyer GR. Drug-induced macrophage autophagy in atherosclerosis: for better or worse. Basic Res Cardiol. 2013;108(1):321.
Stawowy P, Meyborg H, Stibenz D, et al. Furin-like proprotein convertases are central regulators of the membrane type matrix metalloproteinase-pro-matrix metalloproteinase-2 proteolytic cascade in atherosclerosis. Circulation. 2005;111(21):2820–7.
Klionsky DJ, Elazar Z, Seglen PO, Rubinsztein DC. Does bafilomycin A1 block the fusion of autophagosomes with lysosomes. Autophagy. 2008;4(7):849–50.
Daugherty A, Tall AR, Daemen M, et al. Recommendation on design, execution, and reporting of animal atherosclerosis studies: a scientific statement from the American Heart Association. Circ Res. 2017;121(6):e53–79.
He J, Zhang G, Pang Q, et al. SIRT6 reduces macrophage foam cell formation by inducing autophagy and cholesterol efflux under ox-LDL condition. FEBS J. 2017;284(9):1324–37.
Ouimet M, Franklin V, Mak E, Liao X, Tabas I, Marcel YL. Autophagy regulates cholesterol efflux from macrophage foam cells via lysosomal acid lipase. Cell Metab. 2011;13(6):655–67.
Hong MK, Mintz GS, Lee CW, et al. Comparison of coronary plaque rupture between stable angina and acute myocardial infarction: a three-vessel intravascular ultrasound study in 235 patients. Circulation. 2004;110(8):928–33.
Shiomi M, Ito T, Hirouchi Y, Enomoto M. Fibromuscular cap composition is important for the stability of established atherosclerotic plaques in mature WHHL rabbits treated with statins. Atherosclerosis. 2001;157(1):75–84.
Tomic D, Shaw JE, Magliano DJ. The burden and risks of emerging complications of diabetes mellitus. Nat Rev Endocrinol. 2022 : 1–15.
Pennig J, Scherrer P, Gissler MC, et al. Glucose lowering by SGLT2-inhibitor empagliflozin accelerates atherosclerosis regression in hyperglycemic STZ-diabetic mice. Sci Rep. 2019;9(1):17937.
Lauritsen KM, Voigt JH, Pedersen SB, et al. Effects of SGLT2 inhibition on lipid transport in adipose tissue in type 2 diabetes. Endocr Connect. 2022. 11(4).
Han JH, Oh TJ, Lee G, et al. The beneficial effects of empagliflozin, an SGLT2 inhibitor, on atherosclerosis in ApoE (-/-) mice fed a western diet. Diabetologia. 2017;60(2):364–76.
Xu J, Hirai T, Koya D, Kitada M. Effects of SGLT2 inhibitors on atherosclerosis: lessons from cardiovascular clinical outcomes in type 2 diabetic patients and basic researches. J Clin Med. 2021. 11(1).
Ha J, Guan KL, Kim J. AMPK and autophagy in glucose/glycogen metabolism. Mol Aspects Med. 2015;46:46–62.
Lin SC, Hardie DG. AMPK: Sensing glucose as well as cellular energy status. Cell Metab. 2018;27(2):299–313.
Wei L, Chen W, Zou Y, et al. AMP-activated protein kinase regulates autophagic protection against cisplatin-induced tissue injury in the kidney. Genet Mol Res. 2015;14(4):12006–15.
Hawley SA, Ford RJ, Smith BK, et al. The Na+/glucose cotransporter inhibitor canagliflozin activates AMPK by inhibiting mitochondrial function and increasing cellular AMP levels. Diabetes. 2016;65(9):2784–94.
Xie Z, Klionsky DJ. Autophagosome formation: core machinery and adaptations. Nat Cell Biol. 2007;9(10):1102–9.
Chen YC, Jandeleit-Dahm K, Peter K. Sodium-glucose co-transporter 2 (SGLT2) inhibitor dapagliflozin stabilizes diabetes-induced atherosclerotic plaque instability. J Am Heart Assoc. 2022;11(1):e022761.
Terasaki M, Hiromura M, Mori Y, et al. Amelioration of hyperglycemia with a sodium-glucose cotransporter 2 inhibitor prevents macrophage-driven atherosclerosis through macrophage foam cell formation suppression in type 1 and type 2 diabetic mice. PLoS ONE. 2015;10(11):e0143396.
Al-Sharea A, Murphy AJ, Huggins LA, Hu Y, Goldberg IJ, Nagareddy PR. SGLT2 inhibition reduces atherosclerosis by enhancing lipoprotein clearance in Ldlr(-/-) type 1 diabetic mice. Atherosclerosis. 2018;271:166–76.
Nasiri-Ansari Ν, Dimitriadis GK, Agrogiannis G, et al. Canagliflozin attenuates the progression of atherosclerosis and inflammation process in APOE knockout mice. Cardiovasc Diabetol. 2018;17(1):106.
Mori K, Tsuchiya K, Nakamura S, et al. Ipragliflozin-induced adipose expansion inhibits cuff-induced vascular remodeling in mice. Cardiovasc Diabetol. 2019;18(1):83.
Seneviratne A, Hulsmans M, Holvoet P, Monaco C. Biomechanical factors and macrophages in plaque stability. Cardiovasc Res. 2013;99(2):284–93.
Acknowledgements
We thank Jiahui Wang, Peng Zhu, and Huishan Zhao (Central Laboratory, The Affiliated Yantai Yuhuangding Hospital of Qingdao University) for their technical assistance.
Funding
Key Research and Development Program of Shandong Province (No. 2019GSF108142), Natural Science Foundation of Shandong Province (No. ZR2022MH151), National Natural Science Foundation of China (No. 81900310) (No. 82200503), Science and Technology Program of Yantai City (No. 2021MSGY042 and), Science and Technology Program of Yantai City (No. 2021MSGY044), and Youth Research Foundation of Yuhuangding Hospital (No. 2021–03).
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration. Informed consent was obtained from all patients before being included in the study. The human research protocol was approved by the medical ethics committee of Yantai Yuhuangding Hospital, affiliated with Qingdao University (NO.2021–346). All Institutional and National Guidelines for the care and use of animals (fisheries) were followed. The animal research was approved by the Institutional Animal Care and Use Committee at Qingdao University (NO.QDU-AEC-2021176).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration. Informed consent was obtained from all patients before being included in the study. The human research protocol was approved by the medical ethics committee of Yantai Yuhuangding Hospital, affiliated with Qingdao University (NO.2021-346). All Institutional and National Guidelines for the care and use of animals (fisheries) were followed. The animal research was approved by the Institutional Animal Care and Use Committee at Qingdao University (NO.QDU-AEC-2021176).
Conflict of Interest
The authors declare no competing interests.
Additional information
Associate Editor Guoping Li oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, H., Teng, D., Xu, B. et al. The SGLT2 Inhibitor Canagliflozin Reduces Atherosclerosis by Enhancing Macrophage Autophagy. J. of Cardiovasc. Trans. Res. 16, 999–1009 (2023). https://doi.org/10.1007/s12265-023-10390-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-023-10390-w