Abstract
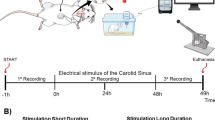
The splanchnic vascular compartment is the major reservoir for intravascular blood volume, and dysregulation of the compartment was implicated in a series of cardiovascular conditions. We explored feasibility and effectiveness of an implantable cuff system on the greater splanchnic nerve (GSN) in healthy canines for short- and long-term neuromodulation to affect the circulation. Five mongrel hounds underwent minimally invasive right-sided unilateral GSN cuff placement. All animals underwent same day GSN stimulation and repeat stimulation at 9–30 days. Stimulation parameter optimization was conducted both acutely and chronically. Parameters ranged from 1–250 Hz, 0.25 mA–35 mA, 0.1–0.5 ms, and 30-s pulse duration. Two animals were survived for 9 days and 3 animals for 30 days. Stimulation of the right GSN increased mean arterial blood pressure by 36.9 mmHg ± 13.4 (p < 0.0001), central venous pressure by 6.9 mmHg ± 1.7 (p < 0.0001), and mean pulmonary arterial pressure by 6.3 mmHg ± 2.0 (p < 0.0001). Peak effects were observed within 30 s, and magnitude of effects was comparable between stimulation cycles (p = 0.4). Stimulation-induced changes in hemodynamics were independent of afferent nerve fibers (pain response) or the adrenal gland. Necropsy showed no evidence of nerve damage on histologic studies up to 30 days after implantation. GSN stimulation via an implanted nerve cuff provided a reproducible and rapid method to increase arterial, central venous, and pulmonary arterial pressures. The neuromodulation cuff was well tolerated and elicited a response up to 30 days after implantation. The clinical application of GSN stimulation as a tool to change central and peripheral cardiovascular hemodynamics needs to be explored.










Similar content being viewed by others
References
Magder, S. (2016). Volume and its relationship to cardiac output and venous return. Critical Care, 20, 271.
Gelman, S. (2008). Venous function and central venous pressure: a physiologic story. Anesthesiology, 108(4), 735–748.
Barnes, R. J., Bower, E. A., & Rink, T. J. (1986). Haemodynamic responses to stimulation of the splanchnic and cardiac sympathetic nerves in the anaesthetized cat. The Journal of Physiology, 378, 417–436.
Greenway, C. V. (1991). Blockade of reflex venous capacitance responses in liver and spleen by hexamethonium, atropine, and surgical section. Canadian Journal of Physiology and Pharmacology, 69(9), 1284–1287.
Carneiro, J. J., & Donald, D. E. (1977). Change in liver blood flow and blood content in dogs during direct and reflex alteration of hepatic sympathetic nerve activity. Circulation Research, 40(2), 150–158.
Greenway, C. V. (1983). Role of splanchnic venous system in overall cardiovascular homeostasis. Federation Proceedings, 42(6), 1678–1684.
Fudim, M., et al. (2017). Raising the pressure: hemodynamic effects of splanchnic nerve stimulation. Journal of Applied Physiology (Bethesda, MD: 1985), jap 00069 2017.
Fudim, M., Hernandez, A. F., & Felker, G. M. (2017). Role of volume redistribution in the congestion of heart failure. Journal of the American Heart Association, 6(8).
Diedrich, A., & Biaggioni, I. (2004). Segmental orthostatic fluid shifts. Clinical Autonomic Research, 14(3), 146–147.
Fallick, C., Sobotka, P. A., & Dunlap, M. E. (2011). Sympathetically mediated changes in capacitance: redistribution of the venous reservoir as a cause of decompensation. Circulation. Heart Failure, 4(5), 669–675.
Fudim, M., et al. (2018). Splanchnic nerve block for acute heart failure. Circulation.
Radlinsky, M. G., et al. (2002). Thoracoscopic visualization and ligation of the thoracic duct in dogs. Vet Surg, 31(2), 138–46.
Parker, R. H., E. A. Kahn, & Iob V. (1953) Thoracic duct ligation during supradiaphragmatic splanchnic section; effect on hypertension and lipid transport. Med Bull (Ann Arbor), 19(11), 291–301.
Kahn, E. A. (1954). Twenty years’ experience with the surgery of hypertension. The New England Journal of Medicine, 251(16), 633–638.
Fudim, M., et al. (2018). Splanchnic nerve block for decompensated chronic heart failure: splanchnic-HF. European Heart Journal.
Brooksby, G. A., & Donald, D. E. (1971). Dynamic changes in splanchnic blood flow and blood volume in dogs during activation of sympathetic nerves. Circulation Research, 29(3), 227–238.
Martin, S. J., Burstein, C. L., & Rovenstine, E. A. (1942). Stimulation of the celiac plexus in the dog: I. Cardiovascular and respiratory effects. Archives of Surgery, 44(5), 943–952.
Pan, H. L., Zeisse, Z. B., & Longhurst, J. C. (1996). Role of summation of afferent input in cardiovascular reflexes from splanchnic nerve stimulation. The American Journal of Physiology, 270(3 Pt 2), H849–H856.
Pan, H. L., et al. (2001). Differential responses of regional sympathetic activity and blood flow to visceral afferent stimulation. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 280(6), R1781–R1789.
Greenway, C. V., & Innes, I. R. (1980). Effects of splanchnic nerve stimulation on cardiac preload, afterload, and output in cats. Circulation Research, 46(2), 181–189.
Rea, R. F., & Thames, M. D. (1993). Neural control mechanisms and vasovagal syncope. Journal of Cardiovascular Electrophysiology, 4(5), 587–595.
Kaufmann, H. (1997). Neurally mediated syncope and syncope due to autonomic failure: differences and similarities. Journal of Clinical Neurophysiology, 14(3), 183–196.
Shen, W. K., et al. (2017). 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology, 70(5), 620–663.
Stewart, J. M., et al. (2004). Relation of postural vasovagal syncope to splanchnic hypervolemia in adolescents. Circulation, 110(17), 2575–2581.
Denq, J. C., et al. (1997). Efficacy of compression of different capacitance beds in the amelioration of orthostatic hypotension. Clinical Autonomic Research, 7(6), 321–326.
Tanaka, H., Yamaguchi, H., & Tamai, H. (1997). Treatment of orthostatic intolerance with inflatable abdominal band. Lancet, 349(9046), 175.
Breslow, M. J., et al. (1987). Effect of vasopressors on organ blood flow during endotoxin shock in pigs. The American Journal of Physiology, 252(2 Pt 2), H291–H300.
Funding
This research was funded by NIH Grant 1R44HL132656-01A1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
AB is an employee at Coridea LLC, INC, serves as consultant to Axon Therapies and supported by R44-HL132656-02 grant.
ZE is an employee at Coridea LLC, INC, serves as consultant to Axon Therapies and supported by R44-HL132656-02 grant.
MF is supported by an American Heart Association Grant 17MCPRP33460225, and serves as a consultant to Coridea, Axon Therapies, Galvani and Daxor.
Additional information
Associate Editor Enrique Lara-Pezzi oversaw the review of this article
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Statement of Clinical Relevance
Splanchnic vascular compliance has been suggested to be central to the pathophysiology of heart failure. Splanchnic nerve stimulation via an implanted nerve cuff provided a reproducible and rapid method to increase arterial, central venous, and pulmonary arterial pressures. The present research opens up numerous avenues of splanchnic nerve modulation for the treatment of diseases such as neurally mediated syncope.
Rights and permissions
About this article
Cite this article
Bapna, A., Adin, C., Engelman, Z.J. et al. Increasing Blood Pressure by Greater Splanchnic Nerve Stimulation: a Feasibility Study. J. of Cardiovasc. Trans. Res. 13, 509–518 (2020). https://doi.org/10.1007/s12265-019-09929-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-019-09929-7