Abstract
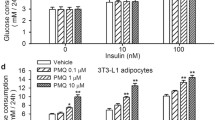
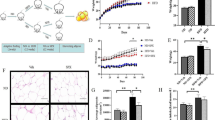
Recently, pharmacological activation of thermogenesis in brown fat and induction of white fat browning (beiging) have been considered as promising strategies in the development of anti-obesity drugs. During the screening of natural compounds that may stimulate thermogenesis, urolithin A (UroA), which is metabolized from pomegranate ellagitannins by gut microflora, was identified as a potent anti-obesity candidate. In the present study, we elucidated the role of UroA to induce the brown-like phenotype in 3T3-L1 white adipocytes. UroA treatments of up to 50 µM were non-toxic to cells. UroA at 15 µM significantly increased the protein expression levels of brown-fatspecific markers such as UCP1, PRDM16, PGC-1α, C/EBPβ, and PPARα. In addition, it remarkably increased the expression of beige-specific genes, including Cd137, Cidea, Cited1, Tbx1, and Tmen26, in 3T3-L1 white adipocytes and significantly elevated expressions of the brown-fatspecific genes (Ppargc1, Prdm16, and Ucp1) in white adipocytes. Furthermore, UroA treatment of 3T3-L1 white adipocytes cells reduced the expression of key adipogenic transcription factors, whereas enhanced lipolysis and the fat oxidation process. Mechanistic study revealed that UroA treatment induces browning in white adipocytes via activation of β3-AR- and p38 MAPK-dependent signaling pathways. Taken together, UroA has the potential to treat obesity by its capacity to recruit beige fat cells in white adipocyte tissue, thereby contributing to an increase in thermogenesis.
Similar content being viewed by others
References
Flegal, K. M., D. Kruszon-Moran, M. D. Carroll, C. D. Fryar, and C. L. Ogden (2016) Trends in obesity among adults in the United States, 2005 to 2014. JAMA. 315: 2284–2291.
Jia, W. (2015) Obesity in China: its characteristics, diagnostic criteria, and implications. Front Med. 9: 129–133.
Wang, S. and J. Ren (2018) Obesity paradox in aging: from prevalence to pathophysiology. Prog. Cardiovasc. Dis. 61: 182–189.
World Health Organisation. Obesity and overweight (2018) http://www.who.int/mediacentre/factsheets/fs311/en/.
Jung, U. J. and M. S. Choi (2014) Obesity and its metabolic complications: the role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 15: 6184–6223.
Kang, J. G. and C. Y. Park (2012) Anti-obesity drugs: A review about their effects and safety. Diabetes Metab. J. 36: 13–25.
Derosa, G. and P. Maffioli (2012) Anti-obesity drugs: a review about their effects and their safety. Expert. Opin. Drug Saf. 11: 459–471.
Tan, C. Y., K. Ishikawa, S. Virtue, and A. Vidal-Puig (2011) Brown adipose tissue in the treatment of obesity and diabetes: Are we hot enough? J. Diabetes Investig. 2: 341–350.
Kaur, K. K., G. Allahbadia, and M. Singh (2018) Advances in BAT physiology for understanding and translating into Pharmacotherapies for obesity and comorbidities. MOJ. Drug Des. Develop. Ther. 2: 166–176.
Saito, M. (2013) Brown adipose tissue as a regulator of energy expenditure and body fat in humans. Diabetes Metab. J. 37: 22–29.
Van Marken Lichtenbelt, W. D., J. W. Vanhommerig, N. M. Smulders, J. M. Drossaerts, G. J. Kemerink, N. D. Bouvy, P. Schrauwen, and G. J. Teule (2009) Cold-activated brown adipose tissue in healthy men. N. Engl. J. Med. 360: 1500–1508.
Cypess, A. M. and C. R. Kahn (2010) Brown fat as a therapy for obesity and diabetes. Curr. Opin. Endocrinol Diabetes Obes. 17: 143–149.
Wang, W. and P. Seale (2016) Control of brown and beige fat development. Nat. Rev. Mol. Cell. Biol. 17: 691–702.
Karri, S., S. Sharma, K. Hatware, and K. Patil (2019) Natural anti-obesity agents and their therapeutic role in management of obesity: A future trend perspective. Biomed. Pharmacother. 110: 224–238.
Bonet, M. L., P. Oliver, and A. Palou (2013) Pharmacological and nutritional agents promoting browning of white adipose tissue. Biochim. Biophys. Acta. 1831: 969–985.
Jang, M. H., N. H. Kang, S. Mukherjee, and J. W. Yun (2018) Theobromine, a methylxanthine in cocoa bean, stimulates thermogenesis by inducing white fat browning and activating brown adipocytes. Biotechnol. Bioprocess Eng. 23: 617–626.
Mukherjee, S., K. R. Aseer, and J. W. Yun (2020) Roles of macrophage colony stimulating factor in white and brown adipocytes. Biotechnol. Bioprocess Eng. 25: 29–38.
Veeresham, C. (2012) Natural products derived from plants as a source of drugs. J. Adv. Pharm. Technol. Res. 3: 200–201.
Mohamed, G. A., S. R. M. Ibrahim, E. S. Elkhayat, and R. S. El Dine (2014) Natural anti-obesity agents. Bull. Fac. Pharm. Cairo Univ. 52: 269–284.
Mopuri, R. and M. S. Islam (2017) Medicinal plants and phytochemicals with anti-obesogenic potentials: A review. Biomed. Pharmacother. 89: 1442–1452.
Azhar, Y., A. Parmar, C. N. Miller, J. S. Samuels, and S. Rayalam (2016) Phytochemicals as novel agents for the induction of browning in white adipose tissue. Nutr. Metab. (Lond). 13: 89.
Silvester, A. J., K. R. Aseer, and J. W. Yun (2019) Dietary polyphenols and their roles in fat browning. J. Nutr. Biochem. 64: 1–12.
Espín, J. C., R. González-Barrio, B. Cerdá, C. López-Bote, A. I. Rey, and F. A. Tomás-Barberán (2007) Iberian pig as a model to clarify obscure points in the bioavailability and metabolism of ellagitannins in humans. J. Agric. Food Chem. 55: 10476–10485.
Gimenez-Bastida, J. A., A. Gonzalez-Sarrias, M. Larrosa, F. Tomas-Barberan, J. C. Espin, and M. T. Garcia-Conesa (2012) Ellagitannin metabolites, urolithin A glucuronide and its aglycone urolithin A, ameliorate TNF-α-induced inflammation and associated molecular markers in human aortic endothelial cells. Mol. Nutr. Food Res. 56: 784–796.
Piwowarski, J. P., S. Granica, M. Zwierzyńska, J. Stefańska, P. Schopohl, M. F. Melzig, and A. K. Kiss (2014) Role of human gut microbiota metabolism in the anti-inflammatory effect of traditionally used ellagitannin-rich plant materials. J. Ethnopharmacol. 155: 801–809.
Heber, D. (2008) Multitargeted therapy of cancer by ellagitannins. Cancer Lett. 269: 262–268.
Li, Z., S. M. Henning, R. P. Lee, Q. Y. Lu, P. H. Summanen, G. Thames, K. Corbett, J. Downes, C. H. Tseng, S. M. Finegold, and D. Heber (2015) Pomegranate extract induces ellagitannin metabolite formation and changes stool microbiota in healthy volunteers. Food Funct. 6: 2487–2495.
Puupponen-Pimiä, R., T. Seppänen-Laakso, M. Kankainen, J. Maukonen, R. Törrönen, M. Kolehmainen, T. Leppänen, E. Moilanen, L. Nohynek, A. M. Aura, K. Poutanen, F. A. Tómas-Barberán, J. C. Espín, and K. M. Oksman-Caldentey (2013) Effects of ellagitannin-rich berries on blood lipids, gut microbiota, and urolithin production in human subjects with symptoms of metabolic syndrome. Mol. Nutr. Food Res. 57: 2258–2263.
Tomás-Barberán, F. A., A. González-Sarrías, R. García-Villalba, M. A. Núñez-Sánchez, M. V. Selma, M. T. García-Conesa, and J. C. Espín (2017) Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 61: 1500901.
Espin, J. C., M. Larrosa, M. T. Garcia-Conesa, and F. Tomas-Barberan (2013) Biological significance of urolithins, the gut microbial ellagic acid-derived metabolites: the evidence so far. Evid. Based Complement Alterna. Med. 2013: 270418.
Selma, M. V., A. González-Sarrías, J. Salas-Salvadó, C. Andrés-Lacueva, C. Alasalvar, A. Örem, F. A. Tomás-Barberán, and J. C. Espín (2018) The gut microbiota metabolism of pomegranate or walnut ellagitannins yields two urolithin-metabotypes that correlate with cardiometabolic risk biomarkers: Comparison between normoweight, overweight-obesity and metabolic syndrome. Clin. Nutr. 37: 897–905.
Ishimoto, H., M. Shibata, Y. Myojin, H. Ito, Y. Sugimoto, A. Tai, and T. Hatano (2011) In vivo anti-inflammatory and antioxidant properties of ellagitannin metabolite urolithin A. Bioorg. Med. Chem. Lett. 21: 5901–5904.
Cerda, B., P. Periago, J. C. Espín, and F. A. Tomas-Barberan (2005) Identification of urolithin A as a metabolite produced by human colon microflora from ellagic acid and related compounds. J. Agric. Food Chem. 53: 5571–5576.
Singh, R., S. Chandrashekharappa, S. R. Bodduluri, B. V. Baby, B. Hegde, N. G. Kotla, A. A. Hiwale, T. Saiyed, P. Patel, M. Vijay-Kumar, M. G. Langille, G. M. Douglas, X. Cheng, E. C. Rouchka, S. J. Waigel, G. W. Dryden, H. Alatassi, H. G. Zhang, B. Haribabu, P. K. Vemula, and V. R. Jala (2019) Enhancement of the gut barrier integrity by a microbial metabolite through the Nrf2 pathway. Nat. Commun. 10: 89.
Kang, I., Y. Kim, F. A. Tomas-Barberan, J. C. Espin, and S. Chung (2016) Urolithin A, C, and D, but not iso-urolithin A and urolithin B, attenuate triglyceride accumulation in human cultures of adipocytes and hepatocytes. Mol. Nutr. Food Res. 60: 1129–1138.
Les, F., J. M. Arbones-Mainar, M. S. Valero, and V. Lopez (2018) Pomegranate polyphenols and urolithin A inhibit α-glucosidase, dipeptidyl peptidase-4, lipase, triglyceride accumulation and adipogenesis related genes in 3T3-L1 adipocyte-like cells. J. Ethnopharmacol. 220: 67–74.
Mosmann, T. (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65: 55–63.
Kang, B., C. Y. Kim, J. Hwang, K. Jo, S. Kim, H. J. Suh, and H. S. Choi (2019) Punicalagin, a pomegranate-derived ellagitannin, suppresses obesity and obesity-induced inflammatory responses via the Nrf2/Keap1 signaling pathway. Mol. Nutr. Food Res. 63: e1900574.
Wang, L., Y. Wei, C. Ning, M. Zhang, P. Fan, D. Lei, J. Du, M. Gale, Y. Ma, and Y. Yang (2019) Ellagic acid promotes browning of white adipose tissues in high-fat diet-induced obesity in rats through suppressing white adipocyte maintaining genes. Endocr. J. 66: 923–936.
Han, Q. A., C. Yan, L. Wang, G. Li, Y. Xu, and X. Xia (2016) Urolithin A attenuates ox-LDL-induced endothelial dysfunction partly by modulating microRNA-27 and ERK/PPAR-γ pathway. Mol. Nutr. Food Res. 60: 1933–1943.
Seale, P., S. Kajimura, W. Yang, S. Chin, L. M. Rohas, M. Uldry, G. Tavernier, D. Langin, and B. M. Spiegelman (2007) Trans-criptional control of brown fat determination by PRDM16. Cell Metab. 6: 38–54.
Sharp, L. Z., K. Shinoda, H. Ohno, D. W. Scheel, E. Tomoda, L. Ruiz, H. Hu, L. Wang, Z. Pavlova, V. Gilsanz, and S. Kajimura (2012) Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS One. 7: e49452.
Tiraby, C., G. Tavernier, C. Lefort, D. Larrouy, F. Bouillaud, D. Ricquier, and D. Langin (2003) Acquirement of brown fat cell features by human white adipocytes. J. Biol. Chem. 278: 33370–33376.
Vega, R. B., J. M. Huss, and D. P. Kelly (2000) The coactivator PGC-1 cooperates with peroxisome proliferator-activated receptor α in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes. Mol. Cell. Biol. 20: 1868–1876.
Toney, A. M., R. Fan, Y. Xian, V. Chaidez, A. E. Ramer-Tait, and S. Chung (2019) Urolithin A, a gut metabolite, improves insulin sensitivity through augmentation of mitochondrial function and biogenesis. Obesity. 27: 612–620.
Farmer, S. R. (2006) Transcriptional control of adipocyte formation. Cell Metab. 4: 263–273.
Tung, Y. C., P. H. Hsieh, M. H. Pan, and C. T. Ho (2017) Cellular models for the evaluation of the antiobesity effect of selected phytochemicals from food and herbs. J. Food Drug Anal. 25: 100–110.
Hardie, D. G., F. A. Ross, and S. A. Hawley (2012) AMPK: A nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13: 251–262.
Viollet, B., S. Horman, J. Leclerc, L. Lantier, M. Foretz, M. Billaud, S. Giri, and F. Andreelli (2010) AMPK inhibition in health and disease. Crit. Rev. Biochem. Mol. Biol. 45: 276–295.
Duncan, R. E., M. Ahmadian, K. Jaworski, E. Sarkadi-Nagy, and H. S. Sul (2007) Regulation of lipolysis in adipocytes. Annu. Rev. Nutr. 27: 79–101.
Nielsen, T. S., N. Jessen, J. O. L. Jørgensen, N. Møller, and S. Lund (2014) Dissecting adipose tissue lipolysis: molecular regulation and implications for metabolic disease. J. Mol. Endocrinol. 52: R199–R222.
Hondares, E., R. Iglesias, A. Giralt, F. J. Gonzalez, M. Giralt, T. Mampel, and F. Villarroya (2011) Thermogenic activation induces FGF21 expression and release in brown adipose tissue. J. Biol. Chem. 286: 12983–12990.
Cao, W., K. W. Daniel, J. Robidoux, P. Puigserver, A. V. Medvedev, X. Bai, L. M. Floering, B. M. Spiegelman, and S. Collins (2004) p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell Biol. 24: 3057–3067.
Whittle, A. J., S. Carobbio, L. Martins, M. Slawik, E. Hondares, M. J. Vázquez, D. Morgan, R. I. Csikasz, R. Gallego, S. Rodriguez-Cuenca, M. Dale, S. Virtue, F. Villarroya, B. Cannon, K. Rahmouni, M. López, and A. Vidal-Puig (2012) BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell. 149: 871–885.
Choi, J. H., S. W. Kim, R. Yu, and J. W. Yun (2017) Monoterpene phenolic compound thymol promotes browning of 3T3-L1 adipocytes. Eur. J. Nutr. 56: 2329–2341.
Cao, W., A. V. Medvedev, K. W. Daniel, and S. Collines (2001) β-adrenergic activation of p38 MAPK kinase in adipocytes: cAMP induction of the uncoupling protein 1 (UCP1) gene requires p38 MAP kinase. J. Biol. Chem. 276: 27077–27082.
Acknowledgment
This study was supported by Daegu University Research Grant 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest The authors declared no conflicts of interest.
Ethical Statement This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manigandan, S., Yun, J.W. Urolithin A Induces Brown-like Phenotype in 3T3-L1 White Adipocytes via β3-adrenergic Receptor-p38 MAPK Signaling Pathway. Biotechnol Bioproc E 25, 345–355 (2020). https://doi.org/10.1007/s12257-020-0149-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-020-0149-8