Abstract
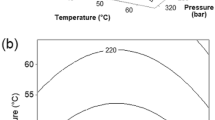
Supercritical fluid extraction (SFE) using cosolvent-modified supercritical carbon dioxide and a two-step separation/purification method was investigated as a way to improve the purity of glabridin, one of the many bioactive components of Glycyrrhiza glabra (licorice). The SFE parameters were optimized using an analytical-scale SFE system in the temperature range 40–80°C and the pressure range 10–50 MPa. The extraction was then scaled up by 100 times using a preparative SFE system under the following set of optimized conditions: 40°C, 30 MPa, and SCCO2 modified with ethanol equivalent to its 25% (v/v) concentration. The glabridin purity obtained through the scaled-up SFE system was 6.2%, a much higher level than that obtained through organic solvent extraction. The licorice extract obtained by scaling up the SFE system was isolated and purified by applying alcohol precipitation/ filtration and adsorption chromatography with 80% aqueous ethanol to obtain a purer product. It was confirmed that the glabridin purity of the final extract product was increased by up to 37%, without significant loss of glabridin after two separation/purification steps.
Similar content being viewed by others
References
Kaur, R., H. Kaur, and A. S. Dhindsa (2013) Glycyrrhiza glabra: A Phytopharmacological review. Int. J. Pharm. Sci. Res. 4: 2470–2477.
Fenwick, G. R., J. Lutomski, and C. Nieman (1990) Liquorice, Glycyrrhiza glabra L.- composition, uses and analysis. Food Chem. 38: 119–143.
Ahn, J., H. Lee, J. Jang, S. Kim, and T. Ha (2013) Anti-obesity effects of glabridin-rich supercritical carbon dioxide extract of licorice in high-fat-fed obese mice. Food Chem. Toxicol. 51: 439–445.
Aoki, F., K. Nakagawa, A. Tanaka, K. Matsuzaki, N. Arai, and T. Mae (2005) Determination of glabridin in human plasma by solid-phase extraction and LC-MS/MS. J. Chromatogr. B 828: 70–74.
Ito, C., N. Oi, T. Hashimoto, H. Nakabayashi, F. Aoki, Y. Tominaga, S. Yokota, K. Hosoe, and K. Kanazawa (2007) Absorption of dietary licorice isoflavan glabridin to blood circulation in rats. J. Nutr. Sci. Vitaminol. 53: 358–365.
Lim, T. K. (2016) Edible Medicinal and Non-medicinal Plants: Modified Stems, Roots, Bulbs. pp. 354–457. Springer, Dordrecht, Netherlands.
Aviram, M., M. Rosenblat, S. Billecke, J. Erogul, R. Sorenson, C. L. Bisgaier, R. S. Newton, and B. L. Du (1999) Human serum paraoxonase (PON1) is inactivated by oxidized low density lipoprotein and preserved by antioxidants. Free Radic. Biol. Med. 26: 892–904.
Belinky, P. A., M. Aviram, S. Mahmood, and J. Vaya (1998) Structural aspects of the inhibitory effect of glabridin on LDL oxidation. Free Radic. Biol. Med. 24: 1419–1429.
Nerya, O., J. Vaya, R. Musa, S. Izrael, R. Ben-Arie, and S. Tamir (2003) Glabrene and isoliquiritigenin as tyrosinase inhibitors from licorice roots. J. Agric. Food Chem. 51: 1201–1207.
Yokota, T., H. Nishio, Y. Kubota, and M. Mizoguchi (1998) The inhibitory effect of glabridin from licorice extracts on melanogenesis and inflammation. Pigment Cell Res. 11: 355–361.
Tamir, S., M. Eizenberg, D. Somjen, S. Izrael, and J. Vaya (2001) Estrogen-like activity of glabrene and other constituents isolated from licorice root. J. Steroid Biochem. Mol. Biol. 78: 291–298.
Ofir, R., S. Tamir, S. Khatib, and J. Vaya (2003) Inhibition of serotonin re-uptake by licorice_constituents. J. Mol. Neurosci. 20: 135–140.
Sawada, K., Y. Yamashita, T. Zhang, K. Nakagawa, and H. Ashida (2014) Glabridin induces glucose uptake via the AMP-activated protein kinase pathway in muscle cells. Mol. Cell. Endocrinol. 393: 99–108.
Nakagawa, K., H. Kishida, N. Arai, T. Nishiyama, and T. Mae (2004) Licorice flavonoids suppress abdominal fat accumulation and increase in blood glucose level in obese diabetic KK-Ay mice. Biol. Pharm. Bull. 27: 1775–1778.
Lee, J. W., S. S. Choe, H. Jang, J. Kim, H. W. Jeong, H. Jo, K. H. Jeong, S. Tadi, M. G. Park, T. H. Kwak, J. M. Kim, D. H. Hyun, and J. B. Kim (2012) AMPK activation with glabridin ameliorates adiposity and lipid dysregulation in obesity. J. Lipid Res. 53: 1277–1286.
Aoki, F., S. Honda, H. Kishida, M. Kitano, N. Arai, H. Tanaka, S. Yokota, K. Nakagawa, T. Asakura, Y. Nakai, and T. Mae (2007) Suppression by licorice flavonoids of abdominal fat accumulation and body weight gain in high-fat diet-induced obese C57BL/6J mice. Biosci. Biotechnol. Biochem. 71: 206–214.
Haraguchi, H., N. Yoshida, H. Ishikawa, Y. Tamura, K. Mizutani, and T. Kinoshita (2000) Protection of mitochondrial functions against oxidative stresses by isoflavans from Glycyrrhiza glabra. J. Pharm. Pharmacol. 52: 219–223.
Shang, H., S. Cao, J. Wang, H. Zheng, and R. Putheti (2010) Glabridin from Chinese herb licorice inhibits fatigue in mice. Afr. J. Tradit Complement. Altern. Med. 7: 17–23.
Fukai, T., K. Satoh, T. Nomura, and H. Sakagami (2003) Preliminary evaluation of antinephritis and radical scavenging activities of glabridin from Glycyrrhiza glabra. Fitoterapia. 74: 624–629.
Fukai, T., A. Marumo, K. Kaitou, T. Kanda, S. Terada, and T. Nomura (2002) Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 71: 1449–1463.
Simmler, C., G. F. Pauli, and S.-N. Chen (2013) Phytochemistry and biological properties of glabridin. Fitoterapia 90: 160–184.
Pastorino, G., L. Cornara, S. Soares, F. Rodrigues, and M. B. P. P. Oliveira (2018) Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytother. Res. 32: 2323–2339.
Zadeh, J. B., Z. M. Kor, and M. K. Goftar (2013) Licorice (Glycyrrhiza glabra Linn) as a valuable medicinal plant. Int. J. Adv. Biol. Biomed. Res. 1: 1281–1288.
Tian, M., H. Yan, and K. Row (2008) Extraction of glycyrrhizic acid and glabridin from licorice. Int. J. Mol. Sci. 9: 571–577.
Viswanathan, V., R. Pharande, A. Bannalikar, P. Gupta, U. Gupta, and A. Mukne (2019) Inhalable liposomes of Glycyrrhiza glabra extract for use in tuberculosis: formulation, in vitro characterization, in vivo lung deposition, and in vivo pharmacodynamic studies. Drug Dev. Ind. Pharm. 45: 11–20.
Modarresi, M., Y. Manoochehri, F. Ahmadi, and L. Hosseinzadeh (2017) Protective effects of glabridin against cytotoxicity and oxidative stress induced by doxorubicin in PC12 cells. J. Rep. Pharm. Sci. 6: 1–12.
Da Silva, R. P. F. F., T. A. P. Rocha–Santos, and A. C. Duarte (2016) Supercritical fluid extraction of bioactive compounds. Trends Analyt. Chem. 76: 40–51.
Durante, M., M. S. Lenucci, and G. Mita (2014) Supercritical carbon dioxide extraction of carotenoids from pumpkin (Cucurbita spp.): A review. Int. J. Mol. Sci. 15: 6725–6740.
Patil, P. D., K. P. R. Dandamudi, J. Wang, Q. Deng, and S. Deng (2018) Extraction of bio-oils from algae with supercritical carbon dioxide and co-solvents. J. Supercrit. Fluids 135: 60–68.
Sajilata, M. G., R. S. Singhal, and M. Y. Kamat (2008) Supercritical CO2 extraction of γ-linolenic acid (GLA) from Spirulina platensis ARM 740 using response surface methodology. J. Food Eng. 84: 321–326.
Charpe, T. W. and V. K. Rathod (2015) Separation of glycyrrhizic acid from licorice root extract using macroporous resin. Food Bioprod. Process. 93: 51–57.
Garcia-Vaquero, M., G. Rajauria, J. V. O’Doherty, and T. Sweeney (2017) Polysaccharides from macroalgae: Recent advances, innovative technologies and challenges in extraction and purification. Food Res. Int. 99: 1011–1020.
Xu, Y., Q. Yuan, X. Hou, and Y. Lin (2009) Preparative separation of glabridin from glycyrrhiza glabra L. extracts with macroporous resins. Sep. Sci. Technol. 44: 3717–3734.
Cho, Y.-K., H.-S. Kim, J.-W. Kim, S.-Y. Lee, W.-S. Kim, J.-H. Ryu, and G.-B. Lim (2004) Extraction of glabridin from licorice using supercritical carbon dioxide. KSBB J. 19: 427–432.
Markom, M., M. Hasan, W. R. W. Daud, H. Singh, and J. M. Jahim (2007) Extraction of hydrolysable tannins from Phyllanthus niruri Linn.: Effects of solvents and extraction methods. Sep. Purif. Technol. 52: 487–496.
Kim, H.-S., B.-Y. Kim, S.-Y. Lee, W.-S. Kim, E.-K. Lee, J.-H. Ryu, and G.-B. Lim (2003) Extraction of glycyrrhizic acid from licorice using supercritical carbon dioxide/aqueous ethanol. KSBB J. 18: 347–351.
Castro-Vargas, H. I., L. I. Rodríguez-Varela, S. R. S. Ferreira, and F. Parada-Alfonso (2010) Extraction of phenolic fraction from guava seeds (Psidium guajava L.) using supercritical carbon dioxide and co-solvents. J. Supercrit. Fluids 51: 319–324.
Gañán, N. A., A. M. A. Dias, F. Bombald, J. A. Zygadlo, E. A. Brignole, H. C. de Sousa, and M. E. M. Braga (2016) Alkaloids from Chelidonium majus L.: Fractionated supercritical CO2 extraction with co-solvents. Sep. Purif. Technol. 165: 199–207.
Tonthubthimthong, P., S. Chuaprasert, P. Douglas, and W. Luewisutthichat (2001) Supercritical CO2 extraction of nimbin from neem seeds - an experimental study. J. Food Eng. 47: 289–293.
Rincón, J., R. Camarillo, and V. Ancillo (2016) Cosolvent effect on the recovery of triglycerides from used frying oil with modified supercritical ethane. J. Supercrit. Fluids 110: 83–89.
Korea Food & Drug Administration, Licorice extract. https://www.foodsafetykorea.go.kr/foodcode
Acknowledgement
The paper was supported by The University of Suwon in 2015.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hong, JH., Jung, II., Cho, YK. et al. Preparation of High-quality Glabridin Extract from Glycyrrhiza glabra. Biotechnol Bioproc E 24, 666–674 (2019). https://doi.org/10.1007/s12257-019-0121-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-019-0121-7