Abstract
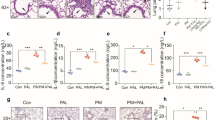
Inhalation of fine particulate matter (PM2.5) is associated with an increase in lung injury caused by the loss of integrity of the vascular barrier. The rare ginsenoside Rg4 is a main protopanaxatriol type ginsenoside of black ginseng (BG). The aim of this study was to investigate the beneficial effects of Rg4 on PM-induced lung endothelial cell (EC) barrier disruption and pulmonary inflammation. Permeability, leukocyte migration, activation of proinflammatory proteins, generation of reactive oxygen species (ROS), and histology were examined in PM2.5-treated EC and mice. Rg4 significantly scavenged PM2.5-induced ROS, inhibited ROS-induced activation of p38 mitogen-activated protein kinase (MAPK), activated Akt in purified pulmonary endothelial cells, which helped maintain endothelial integrity. Further, Rg4 reduced vascular protein leakage, leukocyte infiltration, and proinflammatory cytokine release in bronchoalveolar lavage fluids in PM-induced mouse lung tissues. Data suggested that Rg4 might exhibit protective effects in PM-induced inflammatory lung injury and vascular hyperpermeability.
Similar content being viewed by others
References
Liu, Q., C. Xu, G. Ji, H. Liu, W. Shao, C. Zhang, A. Gu, and P. Zhao (2017) Effect of exposure to ambient PM2.5 pollution on the risk of respiratory tract diseases: a meta-analysis of cohort studies. J. Biomed. Res. 31: 130–142.
Xing, Y. F., Y. H. Xu, M. H. Shi, and Y. X. Lian (2016) The impact of PM2.5 on the human respiratory system. J. Thorac. Dis. 8: E69–74.
Rao, X., J. Zhong, R. D. Brook, and S. Rajagopalan (2018) Effect of particulate matter air pollution on cardiovascular oxidative stress pathways. Antioxid. Redox Signal. 28: 797–818.
Brook, R. D., B. Franklin, W. Cascio, Y. Hong, G. Howard, M. Lipsett, R. Luepker, M. Mittleman, J. Samet, S. C. Smith, Jr., and I. Tager (2004) Air pollution and cardiovascular disease: a statement for healthcare professionals from the expert panel on population and prevention science of the American heart association. Circulation. 109: 2655–2671.
Araujo, J. A., and A. E. Nel (2009) Particulate matter and atherosclerosis: role of particle size, composition and oxidative stress. Part. Fibre Toxicol. 6: 24.
de Kok, T. M., L. G. Engels, E. J. Moonen, and J. C. Kleinjans (2005) Inflammatory bowel disease stimulates formation of carcinogenic N-nitroso compounds. Gut. 54: 731.
Sun, Y., Y. Yin, J. Zhang, H. Yu, X. Wang, J. Wu, and Y. Xue (2008) Hydroxyl radical generation and oxidative stress in Carassius auratus liver, exposed to pyrene. Ecotoxicol. Environ. Saf. 71: 446–453.
Wu, S., F. Deng, Y. Hao, X. Wang, C. Zheng, H. Lv, X. Lu, H. Wei, J. Huang, Y. Qin, M. Shima, and X. Guo (2014) Fine particulate matter, temperature, and lung function in healthy adults: findings from the HVNR study. Chemosphere 108: 168–174.
Shusterman, D. (2011) The effects of air pollutants and irritants on the upper airway. Proc. Am. Thorac. Soc. 8: 101–105.
Deng, X., F. Zhang, W. Rui, F. Long, L. Wang, Z. Feng, D. Chen, and W. Ding (2013) PM2.5-induced oxidative stress triggers autophagy in human lung epithelial A549 cells. Toxicol. In Vitro. 27: 1762–1770.
Kelly, F. J. and J. C. Fussell (2015) Linking ambient particulate matter pollution effects with oxidative biology and immune responses. Ann. N. Y. Acad. Sci. 1340: 84–94
Dong, H., L. P. Bai, V. K. Wong, H. Zhou, J. R. Wang, Y. Liu, Z. H. Jiang, and L. Liu (2011) The in vitro structure-related anticancer activity of ginsenosides and their derivatives. Molecules 16: 10619–10630.
Wu, J. Y., B. H. Gardner, C. I. Murphy, J. R. Seals, C. R. Kensil, J. Recchia, G. A. Beltz, G. W. Newman, and M. J. Newman (1992) Saponin adjuvant enhancement of antigen-specific immune responses to an experimental HIV-1 vaccine. J. Immunol. 148: 1519–1525.
Nag, S. A., J. J. Qin, W. Wang, M. H. Wang, H. Wang, and R. W. Zhang (2012) Ginsenosides as anticancer agents: in vitro and in vivo activities, structure-activity relationships, and molecular mechanisms of action. Front. Pharmacol. 3.
Park, J. D., D. K. Rhee, and Y. H. Lee (2005) Biological Activities and Chemistry of Saponins from Panax ginseng C. A. Meyer. Phytochem. Rev 4: 159–175.
Kang, O. H., M. Y. Shon, R. Kong, Y. S. Seo, T. Zhou, D. Y. Kim, Y. S. Kim, and D. Y. Kwon (2017) Anti-diabetic effect of black ginseng extract by augmentation of AMPK protein activity and upregulation of GLUT2 and GLUT4 expression in db/db mice. BMC Complem. Altern. M. 17: 341.
Park, J. Y., D. S. Lee, C. E. Kim, M. S. Shin, C. S. Seo, H. K. Shin, G. S. Hwang, J. M. An, S. N. Kim, and K. S. Kang (2018) Effects of fermented black ginseng on wound healing mediated by angiogenesis through the mitogen-activated protein kinase pathway in human umbilical vein endothelial cells. J. Ginseng Res. 42: 524–531.
Saba, E., Y. Y. Lee, M. Kim, S. H. Kim, S. B. Hong, and M. H. Rhee (2018) A comparative study on immune-stimulatory and antioxidant activities of various types of ginseng extracts in murine and rodent models. J. Ginseng Res. 42: 577–584.
Sun, B. S., L. J. Gu, Z. M. Fang, C. Y. Wang, Z. Wang, M. R. Lee, Z. Li, J. J. Li, and C. K. Sung (2009) Simultaneous quantification of19 ginsenosides in black ginseng developed from Panax ginseng by HPLC-ELSD. J. Pharm. Biomed. Anal. 50: 15–22.
Cho, J. H., H. Y. Chun, J. S. Lee, J. H. Lee, K. J. Cheong, Y. S. Jung, T. G. Woo, M. H. Yoon, A. Y. Oh, S. M. Kang, C. Lee, H. Sun, J. Hwang, G. Y. Song, and B. J. Park (2016) Prevention effect of rare ginsenosides against stress-hormone induced MTOC amplification. Oncotarget 7: 35144–35158.
Lee, J. H., H. Lim, O. Shehzad, Y. S. Kim, and H. P. Kim (2014) Ginsenosides from Korean red ginseng inhibit matrix metalloproteinase-13 expression in articular chondrocytes and prevent cartilage degradation. Eur. J. Pharmacol. 724: 145–151.
Chen, B., Y. P. Shen, D. F. Zhang, J. Cheng, and X. B. Jia (2013) The apoptosis-inducing effect of ginsenoside F4 from steamed notoginseng on human lymphocytoma JK cells. Nat. Prod. Res. 27: 2351–2354.
Wang, H., L. Song, W. Ju, X. Wang, L. Dong, Y. Zhang, P. Ya, C. Yang, and F. Li (2017) The acute airway inflammation induced by PM2.5 exposure and the treatment of essential oils in Balb/c mice. Sci. Rep-UK. 7: 44256.
Kovacs-Kasa, A., M. N. Varn, A. D. Verin, and J. N. Gonzales (2017) Method for the culture of mouse pulmonary microvascular endothelial cells. Sci. Pages Pulmonol. 1: 7–18.
Kim, J. E., W. Lee, S. Yang, S. H. Cho, M. C. Baek, G. Y. Song, and J. S. Bae (2019) Suppressive effects of rare ginsenosides, Rk1 and Rg5, on HMGB1-mediated septic responses. Food Chem. Toxicol. 124: 45–53.
Lee, I. C., and J. S. Bae (2019) Pelargonidin protects against renal injury in a mouse model of sepsis. J. Med. Food. 22: 57–61.
Zhang, L., and M. C. Wang (2018) Growth Inhibitory Effect of Mangiferin on Thyroid Cancer Cell Line TPC1. Biotechnol. Bioproc. E. 23: 649–654.
Jang, M. H., N. H. Kang, S. Mukherjee, and J. W. Yun (2018) Theobromine, a methylxanthine in cocoa bean, stimulates thermogenesis by inducing white fat browning and activating brown adipocytes. Biotechnol. Bioproc E. 23: 617–626.
Piao, M. J., M. J. Ahn, K. A. Kang, Y. S. Ryu, Y. J. Hyun, K. Shilnikova, A. X. Zhen, J. W. Jeong, Y. H. Choi, H. K. Kang, Y. S. Koh, and J. W. Hyun (2018) Particulate matter 2.5 damages skin cells by inducing oxidative stress, subcellular organelle dysfunction, and apoptosis. Arch. Toxicol. 92: 2077–2091.
Ozdulger, A., I. Cinel, O. Koksel, L. Cinel, D. Avlan, A. Unlu, H. Okcu, M. Dikmengil, and U. Oral (2003) The protective effect of N-acetylcysteine on apoptotic lung injury in cecal ligation and puncture-induced sepsis model. Shock 19: 366–372.
Wang, T., Y. Shimizu, X. Wu, G. T. Kelly, X. Xu, L. Wang, Z. Qian, Y. Chen, and J. G. N. Garcia (2017) Particulate matter disrupts human lung endothelial cell barrier integrity via Rhodependent pathways. Pulm. Circ. 7: 617–623.
Wang, T., E. T. Chiang, L. Moreno-Vinasco, G. D. Lang, S. Pendyala, J. M. Samet, A. S. Geyh, P. N. Breysse, S. N. Chillrud, V. Natarajan, and J. G. Garcia (2010) Particulate matter disrupts human lung endothelial barrier integrity via ROS- and p38 MAPK-dependent pathways. Am. J. Respir. Cell Mol. Biol. 42: 442–449.
Qin, Y. H., S. M. Dai, G. S. Tang, J. Zhang, D. Ren, Z. W. Wang, and Q. Shen (2009) HMGB1 enhances the proinflammatory activity of lipopolysaccharide by promoting the phosphorylation of MAPK p38 through receptor for advanced glycation end products. J. Immunol. 183: 6244–6250.
Sun, C., C. Liang, Y. Ren, Y. Zhen, Z. He, H. Wang, H. Tan, X. Pan, and Z. Wu (2009) Advanced glycation end products depress function of endothelial progenitor cells via p38 and ERK 1/2 mitogen-activated protein kinase pathways. Basic Res. Cardiol. 104: 42–49.
Zhao, Y., P. V. Usatyuk, I. A. Gorshkova, D. He, T. Wang, L. Moreno-Vinasco, A. S. Geyh, P. N. Breysse, J. M. Samet, E. W. Spannhake, J. G. Garcia, and V. Natarajan (2009) Regulation of COX-2 expression and IL-6 release by particulate matter in airway epithelial cells. Am. J. Respir. Cell Mol. Biol. 40: 19–30.
Xu, C., Q. Shi, L. Zhang, and H. Zhao (2018) High molecular weight hyaluronan attenuates fine particulate matter-induced acute lung injury through inhibition of ROS-ASK1-p38/JNK-mediated epithelial apoptosis. Environ. Toxicol. Pharmacol. 59: 190–198.
Gualtieri, M., E. Longhin, M. Mattioli, P. Mantecca, V. Tinaglia, E. Mangano, M. C. Proverbio, G. Bestetti, M. Camatini, and C. Battaglia (2012) Gene expression profiling of A549 cells exposed to Milan PM2.5. Toxicol. Lett. 209: 136–145.
Bellacosa, A., T. O. Chan, N. N. Ahmed, K. Datta, S. Malstrom, D. Stokoe, F. McCormick, J. N. Feng, and P. Tsichlis (1998) Akt activation by growth factors is a multiple-step process: the role of the PH domain. Oncogene 17: 313–325.
Singleton, P. A., S. Chatchavalvanich, P. Fu, J. Xing, A. A. Birukova, J. A. Fortune, A. M. Klibanov, J. G. Garcia, and K. G. Birukov (2009) Akt-mediated transactivation of the S1P1 receptor in caveolin-enriched microdomains regulates endothelial barrier enhancement by oxidized phospholipids. Circ. Res. 104: 978–986.
Shiojima, I. and K. Walsh (2002) Role of Akt signaling in vascular homeostasis and angiogenesis. Circ. Res. 90: 1243–1250.
Komarova, Y. A., D. Mehta, and A. B. Malik (2007) Dual regulation of endothelial junctional permeability. Sci. STKE. 2007: re8.
Curry, F. R. and R. H. Adamson (2013) Tonic regulation of vascular permeability. Acta Physiol. (Oxf) 207: 628–649.
Mehta, D., K. Ravindran, and W. M. Kuebler (2014) Novel regulators of endothelial barrier function. Am. J. Physiol. Lung Cell. Mol. Physiol. 307: L924–935.
Dominici, F., R. D. Peng, M. L. Bell, L. Pham, A. McDermott, S. L. Zeger, and J. M. Samet (2006) Fine particulate air pollution and hospital admission for cardiovascular and respiratory diseases. JAMA 295: 1127–1134.
Peng, R. D., H. H. Chang, M. L. Bell, A. McDermott, S. L. Zeger, J. M. Samet, and F. Dominici (2008) Coarse particulate matter air pollution and hospital admissions for cardiovascular and respiratory diseases among Medicare patients. JAMA 299: 2172–2179.
Schicker, B., M. Kuhn, R. Fehr, L. M. Asmis, C. Karagiannidis, and W. H. Reinhart (2009) Particulate matter inhalation during hay storing activity induces systemic inflammation and platelet aggregation. Eur. J. Appl. Physiol. 105: 771–778.
Mutlu, G. M., D. Green, A. Bellmeyer, C. M. Baker, Z. Burgess, N. Rajamannan, J. W. Christman, N. Foiles, D. W. Kamp, A. J. Ghio, N. S. Chandel, D. A. Dean, J. I. Sznajder, and G. R. Budinger (2007) Ambient particulate matter accelerates coagulation via an IL-6-dependent pathway. J. Clin. Invest. 117: 2952–2961.
Baccarelli, A., P. A. Cassano, A. Litonjua, S. K. Park, H. Suh, D. Sparrow, P. Vokonas, and J. Schwartz (2008) Cardiac autonomic dysfunction: effects from particulate air pollution and protection by dietary methyl nutrients and metabolic polymorphisms. Circulation 117: 1802–1809.
Wang, T., L. Moreno-Vinasco, Y. Huang, G. D. Lang, J. D. Linares, S. N. Goonewardena, A. Grabavoy, J. M. Samet, A. S. Geyh, P. N. Breysse, Y. A. Lussier, V. Natarajan, and J. G. Garcia (2008) Murine lung responses to ambient particulate matter: genomic analysis and influence on airway hyperresponsiveness. Environ. Health Perspect. 116: 1500–1508.
Garcon, G., Z. Dagher, F. Zerimech, F. Ledoux, D. Courcot, A. Aboukais, E. Puskaric, and P. Shirali (2006) Dunkerque City air pollution particulate matter-induced cytotoxicity, oxidative stress and inflammation in human epithelial lung cells (L132) in culture. Toxicol In Vitro 20: 519–528.
Pozzi, R., B. De Berardis, L. Paoletti, and C. Guastadisegni (2003) Inflammatory mediators induced by coarse (PM2.5–10) and fine (PM2.5) urban air particles in RAW 264.7 cells. Toxicology 183: 243–254.
Hulsmann, A. R., H. R. Raatgeep, J. C. den Hollander, T. Stijnen, P. R. Saxena, K. F. Kerrebijn, and J. C. de Jongste (1994) Oxidative epithelial damage produces hyperresponsiveness of human peripheral airways. Am. J. Respir. Crit. Care Med. 149: 519–525.
Kreyling, W. G., M. Semmler, F. Erbe, P. Mayer, S. Takenaka, H. Schulz, G. Oberdorster, and A. Ziesenis (2002) Translocation of ultrafine insoluble iridium particles from lung epithelium to extrapulmonary organs is size dependent but very low. J. Toxicol. Environ. Health A 65: 1513–1530.
Karoly, E. D., Z. Li, L. A. Dailey, X. Hyseni, and Y. C. Huang (2007) Up-regulation of tissue factor in human pulmonary artery endothelial cells after ultrafine particle exposure. Environ. Health Perspect. 115: 535–540.
Walters, D. M., P. N. Breysse, and M. Wills-Karp (2001) Ambient urban Baltimore particulate-induced airway hyperresponsiveness and inflammation in mice. Am. J. Respir. Crit. Care Med. 164: 1438–1443.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No.2018R1A5A2025272 and 2017R1A5A2015385).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, W., Ku, SK., Kim, JE. et al. Pulmonary Protective Functions of Rare Ginsenoside Rg4 on Particulate Matter-induced Inflammatory Responses. Biotechnol Bioproc E 24, 445–453 (2019). https://doi.org/10.1007/s12257-019-0096-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-019-0096-4