Abstract
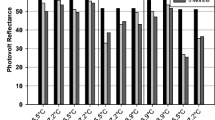
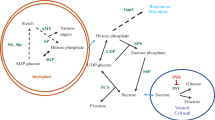
Past investigations have suggested that both UGPase and AcInv activities can be used as markers to screen genetically diverse potato clones for cold induced sweetening resistance (CIS-R). The goal of this study was to define their cooperative interaction in regulating sweetening. Inter- and intra-ploidy hybridizations of good (G) and poor (P) processing 24 or 48 chromosome potato clones were used to create 24 potato families. Potatoes were field grown and 460 progeny (≤20 each family) were stored for five months in the cold (4 C). Tubers from each progeny plant were evaluated for cold induced sweetening resistance (CIS-R) and correlated with the percentage of A-II isozymes of UDP-glucose pyrophosphorylase (UGPase; EC 2.7.7.9); and acid invertase activity (AcInv; EC 3.2.1.26). Each progeny was given a CIS-R score of 1–10 (1-most resistance, 10 least resistance). The families were grouped into four classes based on (1) high or low AcInv activity (low being a SA of 0.30 or less) (2) high or low percentage of A-II isozymes (low being 50% or less), and (3) CIS-R score. In high AcInv families, CIS-R was low regardless of the percentage of A-II isozymes present. In low AcInv activity families, there was a trend for average chip color to improve as the percentage of A-II isozymes increased from 0% to 40%. This increase in CIS-R in low AcInv families is likely due to the kinetic properties unique to the A-II forms of UGPase (principally UGP5) which limit the formation of sucrose via sucrose-6-phosphate synthase (SPS; EC 2.4.1.14). Lower concentrations of sucrose can lead to a decrease in reducing sugar production via vacuolar AcInv and lighter chip and fry colors. In selecting tetraploid parents, for the development of processing potato clones with improved CIS-R, it is recommended they have a basal AcInv SA of 0.30 or less and have A-II isozymes of UGPase.
Resumen
Investigaciones previas han sugerido que las actividades de la UGPasa y AcInv pueden usarse como marcadores para estudiar clones de papa genéticamente diversos para resistencia al endulzamiento inducido por el frío (CIS-R). La meta de este estudio fue definir su interacción cooperativa en la regulación del endulzamiento. Se usaron hibridaciones inter e intra-ploidía, de clones de bueno (G) y pobre (P) procesamiento, de 24 o 48 cromosomas, para crear 24 familias de papa. Las papas se cultivaron en el campo, y una progenie de 460 (≤20 cada familia) se almacenaron por cinco meses en frío (4 °C). Se evaluaron los tubérculos de la progenie de cada planta para resistencia al endulzamiento inducido por el frío (CIS-R) y se correlacionaron con el porcentaje de actividad de isoenzimas A-II de UDP-glucosa pirofosforilasa (UGPasa; EC 2.7.7.9) y de la ácido invertasa (AcInv; EC 3.2.1.26). A cada progenie se le dio una calificación de CIS-R de 1 a 10 (1, más resistencia, 10, menos resistencia). Se agrupó a las familias en cuatro clases con base en (1) alta o baja actividad de AcInv (siendo la baja un AS de 0.30 o menos). (2) alto o bajo porcentaje de isoenzimas A-II (siendo bajo 50% o menos), y (3) la calificación CIS-R. En las familias de alta AcInv, CIS-R fue bajo independientemente del porcentaje de las isoenzimas A-II presentes. En las familias de baja actividad AcInv hubo una tendencia a promediar el color de la hojuela por mejorar, mientras que el porcentaje de las isoenzimas A-II aumentó de 0% a 40%. Este aumento en CIS-R en las familias de baja AcInv es probable que se deba a las propiedades cinéticas únicas para las formas A-II de UGPasa (principalmente UGP5) que limita la formación de sacarosa vía sacarosa-6-fosfato sintetasa (SPS; EC 2.4.1.14). Concentraciones más bajas de sacarosa pueden conducir a una disminución de la producción de azúcar reductor via AcInv vacuolar y color más claro de hojuelas y de papa frita. En la selección de progenitores tetraploides, para el desarrollo de clones de papa para proceso con CIS-R mejorado, se recomienda que tengan un AS AcInv de 0.30 o menos y que tenga isoenzimas A-II de UGPasa.






Similar content being viewed by others
Abbreviations
- CIS-R:
-
Cold induced sweetening-resistance
- CIS-S:
-
Cold induced sweetening-sensitive
- RS:
-
Reducing sugars
- UGPase:
-
UDP-Glc pyrophosphorylase
- RH:
-
Relative humidity
- SPS:
-
Sucrose-6-phosphate synthase
- kDa:
-
Kilodalton
- AcInv:
-
Acid invertase
- PGM:
-
Phosphoglucomutase
- Units of AcInv:
-
μmol glc formed h−1
- SA:
-
Specific activity of AcInv = μmol glc formed h−1 mg−1 protein
References
Ashwell, G. 1957. Colorimetric analysis of sugar. In Methods of Enzymology, vol III, ed. S.P. Colwick and N.O. Kaplan, 73–105. New York: Academic Press.
Bhaskar, P.B., L. Wu, J.S. Busse, B.R. Whitty, A.J. Hamernik, S.H. Jansky, C.R. Buell, P.C. Bethke, and J. Jiang. 2010. Suppression of the vacuolar invertase gene prevents cold-induced sweetening in potato. Plant Physiology 154: 939–948.
Borovkov, A.Y., P.E. McClean, J.R. Sowokinos, S.H. Ruud, and G.A. Secor. 1995. Effect of expression of UDP-glucose pyrophosphorylase antisense and ribozyme RNAs on the enzyme activity and carbohydrate composition of transgenic potato plants. Journal of Plant Physiology 147: 644–652.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72: 248–254.
Greiner, S., T. Rausch, U. Sonnewald, and K. Herbers. 1999. Ectopic expression of a tobacco invertase inhibitor homolog prevents cold-induced sweetening sweetening of potato tubers. Nature Biotechnology 17: 708–711.
Gupta, S.K. 2017. Predictive markers for cold-induced sweetening resistance in cold stored potatoes (Solanum tuberosum L.). American Journal of Potato Research 94: 297–305.
Gupta, S.K., and J.R. Sowokinos. 2003. Physiochemical and kinetic properties of unique isozymes of UDP-glc pyrophosphorylase that are associated with resistance to sweetening in cold-stored potato tubers. Journal of Plant Physiology 160: 589–600.
Habib, A., and H.D. Brown. 1957. The role of reducing sugars and amino acids in browning of potato chips. Food Technology 11: 85–80.
Hill, L., R. Reimholz, R. Schroder, T.H. Nielsen, and M. Stitt. 1996. The onset of sucrose accumulation in cold-stored potato tubers is caused by an increased rate of sucrose synthesis and coincides with low levels of hexose-phosphates, an activation of sucrose phosphate synthase and the appearance of a new form of amylase. Plant, Cell and Environment 19: 1223–1237.
Huber, S.C., and J.L. Huber. 1996. Role and regulation of sucrose-phosphate synthase in higher plants. Annual Review of Physiology and Plant Molecular Biology 47: 431–444.
Krause, K.-P., L. Hill, R. Reimholz, T.H. Nielsen, U. Sonnewald, and M. Stitt. 1998. Sucrose metabolism in cold-stored tubers with decreased expression of sucrose phosphate synthase. Plant, Cell and Environment 21: 285–299.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Liu, X., C. Zhang, Y. Ou, Y. Lin, B. Song, C. Xie, J. Liu, and X.Q. Li. 2011. Systematic analysis of potato acid invertase genes reveals that a cold-responsive member StvacINV1, regulates cold-induced sweetening of tubers. Molecular Genetics and Genomics 286: 109–118.
Liu, X., W. Shi, W. Yin, and J. Wang. 2017. Distinct cold responsiveness of a StInvInh2 gene promoter in transgenic potato tubers with contrasting resistance to cold-induced sweetening. Journal of Plant Physiology and Biochemistry 111: 77–84.
Lulai, E.C., and P.H. Orr. 1990. Quality-testing facilities for grower use at the potato research laboratory. American Journal of Potato Research 57: 622–628.
Manchencko, G.P. 1994. Method of detection of specific enzymes. In: GP Manchencko (ed), Handbook of Detection of Enzymes on Electrophoretic Gels. CRC Press, Boca Raton, FL. p 20.
Mares, D.J., J.R. Sowokinos, and J.S. Hawker. 1985. Carbohydrate metabolism in developing potato tubers. In Potato physiology, ed. P.H. Li, 279–327. New York: Academic Press.
Matsuura-Endo, C., A. Kobayashi, T. Noda, S. Takigawa, H. Yamacuchi, and M. Mori. 2004. Changes in sugar content and activity of vacuolar acid invertase during low-temperature storage of potato tubers from six Japanese cultivars. Journal of Plant Research 117: 131–137.
McKenzie, M.J., J.R. Sowokinos, I.M. Shea, S.K. Gupta, R.R. Linlauf, and J.A.D. Anderson. 2005. Investigations on the role of acid invertase and UDP-glucose pyrophosphorylase in potato clones with varying resistance to cold induced sweetening. American Journal of Potato Research 82: 231–239.
McKenzie, M.J., R.K.Y. Chen, J.C. Harris, M.J. Ashworth, and D.A. Brummell. 2013. Post-translational regulation of acid invertase activity by vacuolar invertase inhibitor affects resistance to cold-induced sweetening of potato tubers. Plant, Cell and Environment 36: 176–185.
Reimholz, R., P. Geigenberger, and M. Stitt. 1994. Sucrose-phosphate synthase is regulated via metabolites and protein phosphorylation in potato tubers, in a manner analogous to the enzyme in leaves. Planta 192: 480–488.
Reimholz, R., M. Geiger, V. Haake, U. Deiting, K.P. Krause, U. Sonnewald, and M. Stitt. 1997. Potato plants contain multiple forms of sucrose phosphate synthase, which differ in their tissue distributions, their levels during development, and their responses to low temperature. Plant, Cell and Environment 20: 291–305.
Richardson, D.L., H.V. Davis, H.A. Ross, and G.R. MacKay. 1990. Invertase activity and its relation to hexose accumulation in potato tubers. Journal of Experimental Botany 41: 95–99.
Sicher, R.C. 1986. Sucrose biosynthesis in photosynthetic tissue rate-controlling factors and metabolic pathway. Physiological Plantaterium 67: 118–121.
Sowokinos, J.R. 1994. Post-harvest regulation of sucrose accumulation in transgenic potatoes: Role and properties of potato tuber UDP-glucose pyrophosphorylase. In The Molecular and Cellular Biology of the Potato, Ed 2, ed. W.R. Belknap, M.E. Vayda, and W.D. Park, 81–106. Wallingford, UK: CAB International.
Sowokinos, J.R. 2001a. Biochemical and molecular control of cold-induced sweetening in potatoes: Invited review. American Journal of Potato Research 78: 221–236.
Sowokinos, J.R. 2001b. Allele and isozyme patterns of UDP-glucose pyrophosphorylase as a marker for cold-sweetening resistance in potatoes. American Journal of Potato Research 78: 57–64.
Sowokinos, J.R., J.P. Spychalla, and S.L. Desborough. 1993. Pyrophosphorylases in Solanum tuberosum IV. Purification, tissue localization, and physicochemical properties of UGP-glucose pyrophosphorylase. Plant Physiology 101: 1073–1080.
Sowokinos, J.R., C. Thomas, and M.M. Burrell. 1997. Pyrophosphorylases in potato. V. Allelic polymorphism of UDP-glucose pyrophosphorylase in potato cultivars and its association with tuber resistance to sweetening in the cold. Plant Physiology 113: 511–517.
Sowokinos, J.R., C.C. Shock, T.D. Stieber, and E.P. Eldredge. 2000. Compositional and enzymatic changes associated with the sugar-end defect in russet Burbank potatoes. American Journal of Potato Research 77: 47–56.
Sowokinos, J.R., V. Vigdorovich, and M. Abrahamsen. 2004. Molecular cloning and sequence variation of UDP-glucose pyrophosphorylase cDNAs from potatoes sensitive and resistant to cold sweetening. Journal of Plant Physiology 161: 947–955.
Townsend, L.R., and G.W. Hope. 1960. Factors influencing the color of potato chips. Canadian Journal of Plant Science 40: 58–64.
Xu, C., W.K. Coleman, F.R. Meng, M. Bonierbale, and X.Q. Li. 2009. Relationship between glucose accumulation and activities of acid invertase and its inhibitors in potatoes under simulated commercial conditions. Potato Journal 36 (1): 35–44.
Zrenner, R., K. Schuler, and U. Sonnewald. 1996. Soluble acid invertase determines the hexose-to-sucrose ratio in cold-stored potato tubers. Planta 198: 246–252.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sowokinos, J.R., Hayes, R.J. & Thill, C.A. Coordinated Regulation of Cold Induced Sweetening in Tetraploid Potato Families by Isozymes of UDP-Glucose Pyrophosphorylase and Vacuolar Acid Invertase. Am. J. Potato Res. 95, 487–494 (2018). https://doi.org/10.1007/s12230-018-9653-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-018-9653-1