Abstract
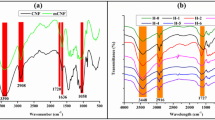
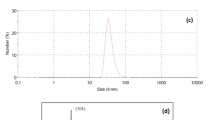
In this article, the synthesis of pectin nano hydrogel, it’s having high absorption ability and its related applications in the treatment of colored wastewater have been investigated. Pectin has been extracted from orange peel as a good source of pectin and the degree of esterification (DE) of pectin as a main parameter on the gel formation were determined. Pectin nano hydrogel was synthesized using Calcium Chloride as a cross-linking agent. The nanometer size and dye adsorption ability of hydrogel particles from colored wastewater have been confirmed by the SEM and UV-Vis spectroscopy methods, respectively. At the next step, the dye adsorption ability of the treated polyester fabrics with pectin nano hydrogel was assessed and results indicated the high adsorption capacity of synthesized nano hydrogel. More specifically the spectrophotometry method indicated that treated polyester fabrics with chitosan and pectin nano hydrogel showed more dye absorption in compare to pectin and cationic surface agent treated fabrics.
Similar content being viewed by others
References
B. Sharma, L. Naresh, N. Dhuldhoya, S. Merchant, and U. Merchant, Times Food Processing J., 23, 44 (2006).
R. Das, A. Panda, and S. J. C. Pal, Cellulose, 19, 933 (2012).
V. Van Tran, D. Park, Y.-C. J. E. S. Lee, and P. Research, Environ. Sci. Pollution Res., 25, 24569 (2018).
N. P. Birch, L. E. Barney, E. Pandres, S. R. Peyton, and J. D. J. B. Schiffinan, Biomacromolecules, 16, 1837 (2015).
L. S. Liu, J. Kost, F. Yan, and R. C. J. P. Spiro, Polymers, 4, 997 (2012).
M. A. Atmodjo, Z. Hao, and D. Mohnen, Annu. Rev. Plant. Biol., 64, 747 (2013).
P. Sriamornsak, Silpakorn Univ. Int. J., 3, 206 (2003).
C. Voiniciuc, K. A. Engle, M. Günl, S. Dieluweit, M. H.-W. Schmidt, J.-Y. Yang, K. W. Moremen, D. Mohnen, and B. J. P. P. Usadel, Plant Physiology, 178, 1045 (2018).
F. Munarin, M. C. Tanzi, and P. Petrini, Biological Macromolecules, 51, 681 (2012).
S. Thakur, J. Chaudhary, V. Kumar, and V. K. Thakur, Environ. Manag, 238, 210 (2019).
R. Wang, X. Cai, and F. Shen, Appl. Surface Sci., 305, 352 (2014).
D.-Q. Li, J. Wang, Z.-G. Guo, J. Li, and J. Shuai, Molecular Liquids, 238, 36 (2017).
A. C. Khorasani and S. A. Shojaosadati, Environ. Chem. Eng., 7, 103062 (2019).
R. Kumar, R. K. Sharma, and A. P. Singh, Environ. Chem. Eng., 6, 6037 (2018).
H. Mittal, A. Maity, and S. S. Ray, Appl. Surface Sci., 364, 917 (2016).
V. K. Gupta, T. A. Saleh, D. Pathania, B. S. Rathore, and G. J. I. Sharma, Ionics, 21, 1787 (2015).
S. Lapwanit, T. Sooksimuang, and T. Trakulsujaritchok, Environ. Chem. Eng., 6, 6221 (2018).
A. Karim, B. Achiou, A. Bouazizi, A. Aaddane, M. Ouammou, M. Bouziane, J. Bennazha, and S. A. Younssi, Environ. Chem. Eng., 6, 1475 (2018).
A. F. S. Costa, C. D. C. Albuquerque, A. A. Salgueiro, L. A. J. P. S. Sarubbo, and E. Protection, Process Saf. Environm. Prot., 118, 203 (2018).
R. Goswami, M. Gogoi, H. J. Borah, P. G. Ingole, and S. Hazarika, Environ. Chem. Eng., 6, 6139 (2018).
X. C. Ruan, M. Y. Liu, Q. F. Zeng, and Y. H. Ding, Sep. Purif. Technol., 74, 195 (2010).
J. Mao, S. W. Won, J. Min, and Y. S. Yun, Korean J. Chem. Eng., 25, 1060 (2008).
P. Nidheesh, M. Zhou, and M. A. Oturan, Chemosphere, 197, 210 (2018).
M. R. Awual, Y. Miyazaki, T. Taguchi, H. Shiwaku, and T. Yaita, Chem. Eng., 291, 128 (2016).
S. Schiewer and M. Iqbal, Hazardous Materials, 177, 899 (2010).
I. Braccini and S. J. B. Pérez, Biomacromolecules, 2, 1089 (2001).
N. T. X. Sam, Sci. Technol., 55, 195 (2017).
A. Bashari, N. Hemmatinejad, and A. Pourjavadi, Polym. Adv. Technol, 24, 797 (2013).
P. Agrawal, M. Parvinzadeh Gashti, and J. Willoughby, “Surface and Bulk Modification of Synthetic Textiles to Improve Dye Ability”, p.261, Saxion University of Applied Sciences, 2011.
Z. Košt’álová, Z. Hromádková, A. Ebringerová, M. Polovka, T. E. Michaelsen, and B. S. Paulsen, Ind. Crops Prod., 41, 127 (2013).
A. T. Paulino, M. R. Guilherme, A. V. Reis, G. M. Campese, E. C. Muniz, and J. Nozaki, Colloid and Interface Sci., 301, 55 (2006).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Shakibi, S., Hemmatinejad, N. & Bashari, A. Sorbent Textiles for Colored Wastewater Made from Orange Based Pectin Nano-hydrogel. Fibers Polym 21, 1275–1282 (2020). https://doi.org/10.1007/s12221-020-9907-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12221-020-9907-7