Abstract
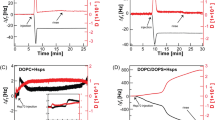
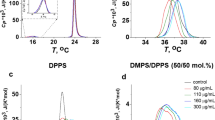
Hsp70-1A—the major stress-inducible member of the HSP70 chaperone family—is being implicated in cancer diseases with the development of resistances to standard therapies. In normal cells, the protein is purely cytosolic, but in a growing number of tumor cells, a significant fraction can be identified on to the cell surface. The anchoring mechanism is still under debate, as Hsp70-1A lacks conventional signaling sequences for translocation from the cytosol to exoplasmic leaflet of the plasma membrane and common membrane binding domains. Recent reports propose a lipid-mediated anchoring mechanism based on a specific interaction with charged, saturated lipids such as dipalmitoyl phosphatidylserine (DPPS). Here, we prepared planar supported lipid bilayers (SLBs) to visualize the association of Hsp70-1A directly and on the single molecule level by atomic force microscopy (AFM). The single molecule sensitivity of our approach allowed us to explore the low concentration range of 0.05 to 1.0 μg/ml of Hsp70-1A which was not studied before. We compared the binding of the protein to bilayers with 20% DPPS lipid content both in the absence and presence of cholesterol. Hsp70-1A inserted exclusively into DPPS domains and assembled in clusters with increasing protein density. A critical density was reached for incubation with 0.5 μg/ml (7 nM); at higher concentrations, membrane defects were observed that originated from cluster centers. In the presence of cholesterol, this critical concentration leads to the formation of membrane blebs, which burst at higher concentrations supporting a previously proposed non-classical pathway for the export of Hsp70-1A by tumor cells. In the discussion of our data, we attempt to link the lipid-mediated plasma membrane localization of Hsp70-1A to its potential involvement in the development of resistances to radiation and chemotherapy based on our own findings and the current literature.





Similar content being viewed by others
Abbreviations
- AFM :
-
atomic force microscopy
- chol :
-
cholesterol
- DOPC :
-
1,2-dioleoyl-sn-glycero-3-phosphocholine
- DOPS :
-
1,2-dioleoyl-sn-glycero-3-phospho-L-serine
- DPPC :
-
1,2-dipalmitoylphosphatidylcholine
- DPPS :
-
1,2-dioleoyl-sn-glycero-3-phospho-L-serine
- eSM :
-
sphingomyelin from chicken egg yolk
- GUV :
-
giant unilamellar vesicle
- Hsp70-1A :
-
heat shock protein 70-1A
- Lα :
-
liquid crystalline phase
- Lβ :
-
solid or gel phase
- Ld :
-
liquid disordered phase
- Lo :
-
liquid ordered phase
- PC :
-
phosphatidylcholine
- SLB :
-
supported lipid bilayer
- SUV :
-
small unilamellar vesicle
References
Aprile FA, Dhulesia A, Stengel F, Roodveldt C, Benesch JLP, Tortora P, Robinson CV, Salvatella X, Dobson CM, Cremades N (2013) Hsp70 oligomerization is mediated by an interaction between the Interdomain linker and the substrate-binding domain. PLoS One 8(6):e67961. https://doi.org/10.1371/journal.pone.0067961
Arispe N, De Maio A (2000) ATP and ADP modulate a cation channel formed by Hsc70 in acidic phospholipid membranes. J Biol Chem 275(40):30839–30843. https://doi.org/10.1074/jbc.M005226200
Arispe N, Doh M, De Maio A (2002) Lipid interaction differentiates the constitutive and stress-induced heat shock proteins Hsc70 and Hsp70. Cell Stress Chaperones 7(4):330–338. https://doi.org/10.1379/1466-1268(2002)007<0330:LIDTCA>2.0.CO;2
Arispe N, Doh M, Simakova O, Kurganov B, De Maio A (2004) Hsc70 and Hsp70 interact with phosphatidylserine on the surface of PC12 cells resulting in a decrease of viability. FASEB J 18(14):1636–1645. https://doi.org/10.1096/fj.04-2088com
Armijo G, Okerblom J, Cauvi DM, Lopez V, Schlamadinger DE, Kim J, Arispe N, de Maio A (2014) Interaction of heat shock protein 70 with membranes depends on the lipid environment. Cell Stress Chaperones 19(6):877–886. https://doi.org/10.1007/s12192-014-0511-x
Asano K, Miwa M, Miwa K, Hanayama R, Nagase H, Nagata S, Tanaka M (2004) Masking of phosphatidylserine inhibits apoptotic cell engulfment and induces autoantibody production in mice. J Exp Med 200(4):459–467. https://doi.org/10.1084/jem.20040342
Bach D, Wachtel E (2003) Phospholipid/cholesterol model membranes: formation of cholesterol crystallites. Biochim Biophys Acta Biomembr 1610(2):187–197. https://doi.org/10.1016/S0005-2736(03)00017-8
Beloribi-Djefaflia S, Vasseur S, Guillaumond F (2016) Lipid metabolic reprogramming in cancer cells. Oncogenesis 5(1):e189. https://doi.org/10.1038/oncsis.2015.49
Farkas B et al (2003) Heat shock protein 70 membrane expression and melanoma-associated marker phenotype in primary and metastatic melanoma. Melanoma Res 13:147–152. https://doi.org/10.1097/01.cmr.0000056221.78713.57
Fritzsching KJ, Kim J, Holland GP (2013) Probing lipid-cholesterol interactions in DOPC/eSM/Chol and DOPC/DPPC/Chol model lipid rafts with DSC and C-13 solid-state NMR. Bba-Biomembranes 1828(8):1889–1898. https://doi.org/10.1016/j.bbamem.2013.03.028
Garcia-Saez AJ, Chiantia S, Schwille P (2007) Effect of line tension on the lateral organization of lipid membranes. J Biol Chem 282(46):33537–33544. https://doi.org/10.1074/jbc.M706162200
Gehrmann M, Marienhagen J, Eichholtz-Wirth H, Fritz E, Ellwart J, Jäättelä M, Zilch T, Multhoff G (2005) Dual function of membrane-bound heat shock protein 70 (Hsp70), Bag-4, and Hsp40: protection against radiation-induced effects and target structure for natural killer cells. Cell Death Differ 12(1):38–51. https://doi.org/10.1038/sj.cdd.4401510
Hantschel M, Pfister K, Jordan A, Scholz R, Andreesen R, Schmitz G, Schmetzer H, Hiddemann W, Multhoff G (2000) Hsp70 plasma membrane expression on primary tumor biopsy material and bone marrow of leukemic patients. Cell Stress Chaperones 5(5):438–442. https://doi.org/10.1379/1466-1268(2000)005<0438:HPMEOP>2.0.CO;2
Ira ZS, Ramirez DMC, Vanderlip S, Ogilvie W, Jakubek ZJ, Johnston LJ (2009) Enzymatic generation of ceramide induces membrane restructuring: correlated AFM and fluorescence imaging of supported bilayers. J Struct Biol 168(1):78–89. https://doi.org/10.1016/j.jsb.2009.03.014
Juhasz K, Lipp AM, Nimmervoll B, Sonnleitner A, Hesse J, Haselgruebler T, Balogi Z (2013) The complex function of hsp70 in metastatic cancer. Cancers (Basel) 6(1):42–66. https://doi.org/10.3390/cancers6010042
Kiessling V, Wan C, Tamm LK (2009) Domain coupling in asymmetric lipid bilayers. Biochim Biophys Acta 1788(1):64–71. https://doi.org/10.1016/j.bbamem.2008.09.003
Mahalka AK, Kirkegaard T, Jukola LT, Jaattela M, Kinnunen PK (2014) Human heat shock protein 70 (Hsp70) as a peripheral membrane protein. Biochim Biophys Acta 1838(5):1344–1361. https://doi.org/10.1016/j.bbamem.2014.01.022
Marquardt D, Kucerka N, Wassall SR, Harroun TA, Katsaras J (2016) Cholesterol's location in lipid bilayers. Chem Phys Lipids 199:17–25. https://doi.org/10.1016/j.chemphyslip.2016.04.001
McCallister C, Kdeiss B, Nikolaidis N (2016) Biochemical characterization of the interaction between HspA1A and phospholipids. Cell Stress Chaperones 21(1):41–53. https://doi.org/10.1007/s12192-015-0636-6
Multhoff G (2007) Heat shock protein 70 (Hsp70): membrane location, export and immunological relevance. Methods 43(3):229–237. https://doi.org/10.1016/j.ymeth.2007.06.006
Multhoff G, Hightower LE (2011) Distinguishing integral and receptor-bound heat shock protein 70 (Hsp70) on the cell surface by Hsp70-specific antibodies. Cell Stress Chaperones 16(3):251–255. https://doi.org/10.1007/s12192-010-0247-1
Multhoff G, Botzler C, Wiesnet M, Muller E, Meier T, Wilmanns W, Issels RD (1995) A stress-inducible 72-kDa heat-shock protein (HSP72) is expressed on the surface of human tumor cells, but not on normal cells. Int J Cancer 61:272–279
Murakami N, Kühnel A, Schmid TE, Ilicic K, Stangl S, Braun IS, Gehrmann M, Molls M, Itami J, Multhoff G (2015) Role of membrane Hsp70 in radiation sensitivity of tumor cells. Radiat Oncol (Lond, Engl) 10(1):149. https://doi.org/10.1186/s13014-015-0461-1
Nečas D, Klapetek P (2012) Gwyddion: an open-source software for SPM data analysis. Cent Eur J Phys 10(1):181–188. https://doi.org/10.2478/s11534-011-0096-2
Nimmervoll B, Chtcheglova LA, Juhasz K, Cremades N, Aprile FA, Sonnleitner A, Hinterdorfer P, Vigh L, Preiner J, Balogi Z (2015) Cell surface localised Hsp70 is a cancer specific regulator of clathrin-independent endocytosis. FEBS Lett 589(19PartB):2747–2753. https://doi.org/10.1016/j.febslet.2015.07.037
Pfister K, Radons J, Busch R, Tidball JG, Pfeifer M, Freitag L, Feldmann HJ, Milani V, Issels R, Multhoff G (2007) Patient survival by Hsp70 membrane phenotype: association with different routes of metastasis. Cancer 110(4):926–935. https://doi.org/10.1002/cncr.22864
Radons J (2016) The human HSP70 family of chaperones: where do we stand? Cell Stress Chaperones 21(3):379–404. https://doi.org/10.1007/s12192-016-0676-6
Ramstedt B, Leppimäki P, Axberg M, Slotte JP (1999) Analysis of natural and synthetic sphingomyelins using high-performance thin-layer chromatography. Eur J Biochem 266(3):997–1002. https://doi.org/10.1046/j.1432-1327.1999.00938.x
Resh MD (2016) Fatty acylation of proteins: the long and the short of it. Prog Lipid Res 63:120–131. https://doi.org/10.1016/j.plipres.2016.05.002
Rohde M, Daugaard M, Jensen MH, Helin K, Nylandsted J, Jaattela M (2005) Members of the heat-shock protein 70 family promote cancer cell growth by distinct mechanisms. Genes Dev 19(5):570–582. https://doi.org/10.1101/gad.305405
Schilling D, Gehrmann M, Steinem C, de Maio A, Pockley AG, Abend M, Molls M, Multhoff G (2009) Binding of heat shock protein 70 to extracellular phosphatidylserine promotes killing of normoxic and hypoxic tumor cells. FASEB J 23(8):2467–2477. https://doi.org/10.1096/fj.08-125229
Schindelin J, Rueden CT, Hiner MC, Eliceiri KW (2015) The ImageJ ecosystem: an open platform for biomedical image analysis. Mol Reprod Dev 82(7-8):518–529. https://doi.org/10.1002/mrd.22489
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Meth 9(7):671–675. https://doi.org/10.1038/nmeth.2089
Sergelius C, Yamaguchi S, Yamamoto T, Engberg O, Katsumura S, Slotte JP (2013) Cholesterol's interactions with serine phospholipids — a comparison of N-palmitoyl ceramide phosphoserine with dipalmitoyl phosphatidylserine. Biochim Biophys Acta Biomembr 1828(2):785–791. https://doi.org/10.1016/j.bbamem.2012.11.009
Simons K, Vaz WLC (2004) Model systems, lipid rafts, and cell membranes. Annu Rev Bioph Biom 33:269–295. https://doi.org/10.1146/annurev.biophys.32.110601.141803
Sovik A, Malinen E, Skogmo HK, Bentzen SM, Bruland OS, Olsen DR (2007) Radiotherapy adapted to spatial and temporal variability in tumor hypoxia. Int J Radiat Oncol Biol Phys 68(5):1496–1504. https://doi.org/10.1016/j.ijrobp.2007.04.027
Sullan RMA, Li JK, Hao C, Walker GC, Zou S (2010) Cholesterol-dependent Nanomechanical stability of phase-segregated multicomponent lipid bilayers. Biophys J 99(2):507–516. https://doi.org/10.1016/j.bpj.2010.04.044
van Duyl BY, Ganchev D, Chupin V, de Kruijff B, Killian JA (2003) Sphingomyelin is much more effective than saturated phosphatidylcholine in excluding unsaturated phosphatidylcholine from domains formed with cholesterol. FEBS Lett 547(1-3):101–106. https://doi.org/10.1016/S0014-5793(03)00678-1
van Meer G, Voelker DR, Feigenson GW (2008) Membrane lipids: where they are and how they behave. Nat Rev Mol Cell Biol 9(2):112–124. https://doi.org/10.1038/nrm2330
Verhoven B, Schlegel RA, Williamson P (1995) Mechanisms of phosphatidylserine exposure, a phagocyte recognition signal, on apoptotic T lymphocytes. J Exp Med 182(5):1597–1601. https://doi.org/10.1084/jem.182.5.1597
Windschiegl B, Orth A, Römer W, Berland L, Stechmann B, Bassereau P, Johannes L, Steinem C (2009) Lipid reorganization induced by Shiga toxin clustering on planar membranes. PLoS One 4(7):e6238. https://doi.org/10.1371/journal.pone.0006238
Zorzi E, Bonvini P (2011) Inducible hsp70 in the regulation of cancer cell survival: analysis of chaperone induction, expression and activity. Cancers (Basel) 3(4):3921–3956. https://doi.org/10.3390/cancers3043921
Acknowledgments
This project received funding from the European Union’S Framework Programme for Research and Innovation Horizon 2020 (2014-2020) under the Marie Sklodowska-Curie Grant Agreement No. 656842 and the Carl-Zeiss-Stiftung (Carl Zeiss Foundation) (Az. 0563-2.8/685/4). The work has been supported in part by the German Federal Ministry of Education and Research (BMBF) in the framework of the EU ERASynBio project SynGlycTis (031A464), by the Ministry of Science, Research and the Arts of Baden-Württemberg (Az: 33-7532.20) and by the Excellence Initiative of the German Research Foundation (EXC 294).
Author information
Authors and Affiliations
Contributions
CL and MG designed the experiments. CL and JM conducted the experiments. CL analyzed the data. AE, WR, GM, and MG helped to conceive the project and discuss results. The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Rights and permissions
About this article
Cite this article
Lamprecht, C., Gehrmann, M., Madl, J. et al. Molecular AFM imaging of Hsp70-1A association with dipalmitoyl phosphatidylserine reveals membrane blebbing in the presence of cholesterol. Cell Stress and Chaperones 23, 673–683 (2018). https://doi.org/10.1007/s12192-018-0879-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-018-0879-0