Abstract
Immune thrombocytopenic purpura (ITP) is a common hematological disease treated primarily by corticosteroids. The aim of the present study was to compare response rate between patients, underwent splenectomy vs. rituximab as second-line therapy. Adult patients diagnosed with ITP who did not respond to corticosteroids or relapsed during the period 1990–2014 were included in a quasi-experimental study. Categorical variables were compared using Fisher exact test. Response to treatment was compared using logistic regression. Data were analyzed using SAS V9.2. One-hundred and forty-three patients with ITP were identified through medical records. Of 62 patients treated, 30 (48.38%) required second-line therapy. 19 (63%) patients received rituximab, and 11 (37%) underwent splenectomy. Platelets at diagnosis were not different between study groups (p = 0.062). Splenectomy group patients were younger (p = 0.011). Response to second-line therapy showed no significant difference between two groups (OR 2.03, 95% CI (0.21–22.09), p = 0.549). Results did not show a statistically significant difference in platelet counts over time between treatment groups (p = 0.101). When used exclusively as a second-line therapy for steroid-refractory ITP, the response rate was not statistically different between rituximab and splenectomy. However, further large studies are needed to assess the response rates for these treatment modalities as a second-line therapy.
Similar content being viewed by others
Introduction
Immune thrombocytopenic purpura (ITP) is a common hematological disease that is characterized by autoimmune-mediated platelet destruction and impairment of thrombopoiesis [1]. The term ITP was referred to “idiopathic” or “immune” thrombocytopenic purpura. However, ITP can no longer be stated as idiopathic since not all cases have idiopathic causes [1]. ITP is defined as “isolated thrombocytopenia with no clinically apparent associated conditions or other causes of thrombocytopenia” [1]. The platelets count cut-off to identify ITP cases has shifted down from 150 × 109/L to 100 × 109/L [1]. The ITP prevalence ranges between 9.5 and 23.6 per 100,000 persons, while incidence among adults ranges between 1.6 and 3.9 per 100,000 persons per year [2]. The incidence is higher among women than men (4.4 vs. 3.4 per 100,000 per year) [3].
ITP is divided into two major diagnostic categories either as primary or secondary type. The majority of cases are primary ITP which is described as the absence of other conditions associated with immune thrombocytopenia [1]. ITP is diagnosed based on isolated thrombocytopenia, unremarkable peripheral blood smear, and physical examination revealing bleeding signs consistent with low platelets in, otherwise, healthy person [4]. ITP treatments depend on the level of platelet count, history of bleeding or if the patient is going for an invasive procedure mandating bleeding prevention [5]. As per the ITP management guidelines, ITP treatment is not required when platelet counts exceeds 20–30 × 109 [4, 5]. The recommended platelet level for initiating therapy is less than 20 × 109/L [4]. The first-line of treatment for ITP is Anti-D, oral corticosteroids and intravenous immunoglobulin (IVIg) [4, 6,7,8]. The sustained response to dexamethasone is 50–80%. Response to IVIg is usually transient and platelets return to baseline level in 2–4 weeks [6]. IVIg is mostly used as an effect modifier in addition to first-line therapy during presence of moderate to severe bleeding.
Splenectomy is often cited as second-line therapy post failure of steroid therapy [5, 6]. Patients who fail to respond to treatment with corticosteroids or require unacceptably high doses of corticosteroid to maintain a safe platelet count should be considered for splenectomy [5, 6]. Two-thirds of patients with ITP who undergo splenectomy will achieve a normal platelet count, which is often sustained with no additional therapy [9].
Rituximab is a monoclonal antibody against CD20 B lymphocytes used mainly in the treatment of lymphoproliferative disorders of B cell origin [10]. Rituximab at the dose of 375 mg/m2 weekly has 40% complete response rate among patients with ITP [6, 10]. Studies have shown complete response to rituximab therapy ranges between 28 and 80% [11, 12].
According to our previously published study, the sustained response at 1 year was achieved in 24% of patients who received rituximab regardless of the order of line of therapy, while patients with prior splenectomy had a poorer response to rituximab [13]. Other therapeutic options as a second-line therapy include azathioprine, cyclosporin, cyclophosphamide, dapsone, mycophenolate mofetil, rituximab, thrombopoietin receptor agonist, and Vinca alkaloid regimens [5, 6, 8].
In this single center study, we aim to compare chronic ITP response rate at 2 years between patients who had splenectomy vs. rituximab as second-line therapy.
Methods
A quasi-experimental study was conducted in November 2014 in Hematology department after receiving institutional review board approval. All patients above the age of 14 years diagnosed with ITP who had relapsed or were refractory to first-line therapy between 1990 and 2014 were screened. ITP diagnosis was confirmed using IWC (International Working Group) criteria which are based on platelet count less than 100 × 109/L in the absence of secondary causes of thrombocytopenia [5].
The index date was start of first-line therapy. The first-line therapy was described as oral prednisolone or dexamethasone. Splenectomy or rituximab was prescribed as a second-line therapy. The indication for second-line therapy was either no response to corticosteroids or inability to wean off corticosteroids. Patients who had failed response to corticosteroids and received second-line therapy rituximab or splenectomy for treating chronic ITP were included in the study. Chronic ITP was considered if ITP was lasting for more than 12 months [5]. Patients diagnosed with secondary ITP (induced by systemic lupus erythematosus, rheumatoid arthritis, HIV and malignancy) were excluded. Rituximab dose was prescribed as 375 mg/m2 intravenously weekly for 4 weeks. The primary outcome of the study was the comparison of response rate for second-line therapy (splenectomy vs. rituximab). Patients were followed up for their response to treatment by measuring platelet counts every 3 months for 2 years.
Response to second-line therapy was measured at three-month intervals, based on platelet count (1) no response (sustained no response) (<30 × 109/L), (2) partial response (based on one reading out of 8 intervals) (30–100,000 × 109/L), (3) complete response (sustained response for 8 intervals) (>100 × 109/L) [9].
The primary data for the study were extracted from patients’ medical charts and hospital health information system. The demographic and clinical characteristics were captured at the time of ITP diagnosis. Platelet counts were collected during patients’ follow-up in the clinic.
Data were analyzed using SAS version 9.2 (SAS Institute Inc., Cary, NC, USA). Gender, bone marrow trephine, cytogenetic results were compared using Fisher Exact test. Age, Hb, WBC, and platelets at diagnosis were compared using Wilcoxon Rank Sum test. The primary outcome of the study response rate comparison between study groups was analyzed using Logistic Regression which was based on probability of having response to treatment, using ‘Splenectomy’ as a reference group. Propensity score was calculated for each patient to make study groups comparable. Change in platelets count at every 3-month intervals for 2 years was analyzed using generalized estimating equation (GEE). The platelet count was assumed as a response variable whereas the treatment groups (rituximab/splenectomy) were considered as an independent variable. The follow-up platelet counts were found missing, once the patient’s platelet count got stabilized in the clinic. Based on that the missing data were imputed using last observation carried forward (LOCF). Significance was declared at alpha less than 0.05.
Results
During the study period (1990–2014), a total of 143 patients with ITP were identified through outpatient medical records. 81(57%) patients required no treatment but observation. Corticosteroids (oral prednisone/dexamethasone) were primarily first-line therapy received by 62 (43%) patients with supplemental IVIg which was given to 23 (26.4%) patients only.
Among those who received corticosteroids, 30 (48.38%) patients were shifted to second-line therapy. 19 (63%) patients received rituximab, while 11 (37%) patients underwent splenectomy (Table 1). Supplemental IVIg was received by 5 (26.3%) of rituximab group and 3 (27.2%) among splenectomy patients.
Among patients who received second-line therapy, more than half of patients were females 19 (63.3%). Patients who had splenectomy were younger compared to patients who had received rituximab (p = 0.011). The study groups were similar in terms of gender, WBC, hemoglobin and baseline platelets count at diagnosis (p = 1.00, 0.961, 0.157 and 0.062, respectively) (Table 1).
No response vs. complete response to second-line therapy showed no significant difference between rituximab and splenectomy groups OR 2.03, 95% CI (0.21–22.90), p = 0.549. Among 19 patients who received rituximab, only 3 (15.8%) patients were switched to third-line therapy (including splenectomy and prednisolone). Among 11 patients who underwent splenectomy, also 3 (27.27%) patients were switched to third-line therapy.
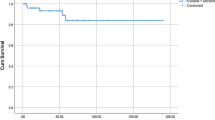
Rituximab group had platelet count 29 at diagnosis, while patients who underwent splenectomy had a platelet count 9 at diagnosis. The baseline platelet prior to initiating second-line therapy was 127 among rituximab group, which dropped to 104 at first (3-month) interval; however, platelets count plateaued across all intervals. On the other hand, the baseline platelet count was 38 among splenectomy group, which increased to 234 during first (3-month) interval. The platelet count dropped at fourth (3-month) interval among splenectomy group; however, the platelet count had jumped again at sixth (3-month) interval. The average platelet count stayed above 100 among both groups across all intervals. Moreover, platelet count was not statistically different between two groups (p = 0.101) (Fig. 1).
Change in platelet count overtime between study groups. All patients have received steroid from 1st line 3-month to 1st line 24-month follow-up. Patients were switched to second-line therapy from 2nd line 3 months to 2nd line 24 months. The red line indicates rituximab group, blue line indicates splenectomy group. N denotes the number of patients in each group at every 3-month interval. There was a steep increase in platelet count among splenectomy, with a drop in platelet at 12-month follow-up; however, platelet count kept plateaued among rituximab group
Discussion
During study period, 48% patients were identified to be refractory or relapsed ITP following first-line treatment with corticosteroids. This is concordant with a study done by Mazzucconi et al. [14] stated that more than 50% of patients who failed to respond to corticosteroids shifted to other types of treatments as a second-line. [14].
The patient selection criteria for our study were based on patients diagnosed with chronic ITP who had received corticosteroids as first-line therapy, and failed to respond or were corticosteroids dependent, compared to other studies which had reported different treatment modalities (e.g., IVIg, cyclophosphamide, cyclosporine, anti-D immune globulin or splenectomy) as first-line [15]. The studies have reported response to rituximab alone with no comparative group. [16].
Two randomized placebo controlled trials have compared treatment failure between rituximab and placebo [17, 18]. However, the patients were newly diagnosed as well as relapsed with prior corticosteroid, IVIg, romiplostim [17].
To our knowledge only one direct comparison was made between rituximab and splenectomy but not exclusively as a second-line therapy and more frequently as third or fourth line therapy [19]. Our study reports direct comparison between rituximab and splenectomy as mutually exclusive groups where all patients had received standardized first-line therapy; corticosteroids.
The response to rituximab has been evaluated by Stasi et al., where 20% of the patients achieved complete remission and another 20% achieved partial remission. Most of these responses continued stable for more than 6 months [20]. In our study, sustained response to second-line treatment showed no significant difference between rituximab and splenectomy groups (OR 2.03, 95% CI 0.21–22.90, p = 0.549). The propensity score was estimated to account for differences between the study groups and to provide the accurate estimate of the effect size. The propensity score variable was calculated using predicted probability from logistic regression; modeling the study group as the dependent variable and all relevant potential confounding variables as the independent variables. C-statistics (0.604) shows that logistic model is weak in strength; this probably is attributable to small sample size. However, firth’s criteria were used to adjust for small sample size.
Because of the retrospective nature of the data, missing values were observed for the platelets follow-up. Last observation carried forward (LOCF) approach was used to deal with missing platelet count for the follow-up visits or complete drop out from the clinic. LOCF is implemented since we assumed that the patients were discharged from the clinic after they were stabilized. The average platelet count 38 vs. 127 prior to starting second-line therapy is supported by the corticosteroids or/and IVIg (Fig. 1) The change in platelet count at first (3-month) interval, showed steep increase in platelet count among patients who underwent splenectomy; however, the rise in platelet count dropped at fourth (3-month) interval and was raised up at sixth (3-month) interval. Comparatively the patients who had received rituximab the average platelet count stayed above 100 and kept plateaued. However, platelet count was not statistically different between treatment groups (p = 0.101) (Fig. 1). The average platelet count for splenectomy group is in concordance with a study done by Kojouri and his colleagues where they found that two-thirds of ITP patients who underwent splenectomy achieved normal platelets counts and sustained without additional treatments [9].
The patients undergoing splenectomy were younger in age (p = 0.011), which may suggest that clinician tend to lean toward splenectomy in young patients or older patients prefer rituximab to avoid the invasive surgical removal of the spleen, hospitalization and surgical complications. However, the surgical complications found to be much less with laparoscopic splenectomy than open splenectomy [17] with lifelong risk for infection [6]. The relative risk of developing infection in first year post splenectomy is 1.49 (95% CI 1.0–2.0) with 50% fatality [6]. On the other hand, one of the major but extremely rare side effects of rituximab is multifocal leukoencephalopathy [13]. One of the cons of the rituximab therapy is that so far it is not approved as a treatment for ITP by US Food and Drug Administration (FDA) [21].
In our study, the administration of rituximab as a second-line has been associated with positive outcomes as platelet count was more stabilized without the side effect of surgery. Among patients who had received rituximab, only 3 (15.8%) switched to third-line therapy that includes splenectomy and prednisolone. As well as 3 (27.27%) patients were switched to third-line therapy among patients who had splenectomy.
Studies have shown the ability of rituximab to induce initial response 40–60% of cases with a sustained response in 35–67% of patients [12, 13, 15, 22]. Godeau and his colleagues had reported the usage of rituximab as a splenectomy sparing agent can yield sustained responses in 33% patients [23]. Aleem et al. have shown that rituximab can induce complete response in 45% patients, while had poorer response in patients who underwent splenectomy [13]. The response was assessed for rituximab in general regardless if it was given as a second-line therapy or beyond. 11 patients out of 24 received rituximab as a third line or beyond after splenectomy. In the current study, response was assessed for rituximab vs. splenectomy when it was given as a second-line therapy exclusively [13]. Cindy et al. described refractory ITP as those who failed splenectomy proposing that splenectomy had to be performed as a second-line therapy given its higher yield in the literature. They also concluded that splenectomy is the only treatment that provides sustained remission at 1 year and beyond [5]. Based on our findings, it appears that the higher yield reported with splenectomy is due to large early use of splenectomy in the order of therapeutic options more than the modality specific. Rituximab yield assessment as a second line shows that yield became equivalent to splenectomy with even better sustainability. This finding should negate the notion that necessitates use of splenectomy as a second-line therapy for ITP after failure of corticosteroid. To our knowledge, this is the first time a comparison is done between two treatment modalities as a second-line treatment for corticosteroid-non-responsive ITP in Saudi Arabia.
We acknowledge that this study being retrospective imposes some limitation. The small sample size may limit the generalization of the study conclusion. However, it certainly raises a valid question and challenges the old notion about proper sequence of therapy when it comes to ITP with poor response to corticosteroids.
Conclusion
In steroid-refractory ITP, the response rate was not statistically different between rituximab and splenectomy when used as a second-line therapy. Furthermore, rituximab resulted in a more sustainable rise in platelets count overtime. However, further large studies are needed to assess the response rate directly comparing the two treatment modalities as a second-line therapy.
References
McCrae K. Immune thrombocytopenia: no longer ‘idiopathic’. Cleve Clin J Med. 2011;78(6):358–73. doi:10.3949/ccjm.78gr.10005.
Abrahamson PE, Hall SA, Feudjo-Tepie M, Mitrani-Gold FS, Logie J. The incidence of idiopathic thrombocytopenic purpura among adults: a population-based study and literature review. Eur J Haematol. 2009;83(2):83–9. doi:10.1111/j.1600-0609.2009.01247.x.
Schoonen WM, Kucera G, Coalson J, Li L, Rutstein M, Mowat F, et al. Epidemiology of immune thrombocytopenic purpura in the General Practice Research Database. Br J Haematol. 2009;145(2):235–44. doi:10.1111/j.1365-2141.2009.07615.x.
Cines DB, Bussel JB. How I treat idiopathic thrombocytopenic purpura (ITP). Blood. 2005;106(7):2244–51.
Neunert C, Lim W, Crowther M, Cohen A, Solberg L Jr, Crowther MA. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011;117(16):4190–207.
Provan D, Stasi R, Newland A, Blanchette VS, Bolton-Maggs P, Bussel JB, et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood 2010;115:168–86. doi:10.1182/blood-2009-06-225565 (published ahead of print October 21, 2009).
The ASH 2011 Clinical Practice Guideline on the Evaluation and Management of Immune Thrombocytopenia; 2011. American Society of Hematology. http://www.hematology.org/Practice/Guidelines/6584.aspx. Cited 2013 June 2011.
Michel M. Immune thrombocytopenia nomenclature, consensus reports, and guidelines: what are the consequences for daily practice and clinical research? Semin Hematol. 2013;50(Suppl 1):S50–4. doi:10.1053/j.seminhematol.2013.03.008.
Kojouri K, Vesely SK, Terrell DR, George JN. Splenectomy for adult patients with idiopathic thrombocytopenic purpura: a systematic review to assess long-term platelet count responses, prediction of response, and surgical complications. Blood. 2004;104(9):2623–34.
Zaja F, Volpetti S, Chiozzotto M, Puglisi S, Isola M, Buttignol S, et al. Long-term follow-up analysis after rituximab salvage therapy in adult patients with immune thrombocytopenia. Am J Hematology. 2012;87:886–9.
Patel VL, Mahevas M, Lee SY, Stasi R, Cunningham-Rundles S, Godeau B, et al. Outcomes 5 years after response to rituximab therapy in children and adults with ITP. Blood. 2012;119(25):5989–95.
Garcia-Chavez J, Majluf-Cruz A, Montiel-Cervantes L, Esparza MG, Vela-Ojeda J, Mexican Hematology Study Group. Rituximab therapy for chronic and refractory immune thrombocytopenic purpura: a long-term follow-up analysis. Ann Hematol. 2007;86(12):871–7.
Aleem A, Alaskar AS, Algahtani F, Rather M, Almahayni MH, Al-Momen A. Rituximab in immune thrombocytopenia: transient responses, low rate of sustained remissions and poor response to further therapy in refractory patients. Int J Hematol. 2010;92(2):283–8. doi:10.1007/s12185-010-0635-4.
Mazzucconi MG, Fazi P, Bernasconi S, De Rossiet Giulio, Leone Giuseppe, Gugliotta Luigi, et al. Therapy with high-dose dexamethasone (HD-DXM) in previously untreated patients affected by idiopathic thrombocytopenic purpura: a GIMEMA experience. Blood. 2007;109(4):1401–7.
Peñalver FJ, Jiménez-Yuste V, Almagro M, Alvarez-Larran A, Rodriguez L, Casado M, et al. Multi-institutional Retrospective Spanish Study Group on the use of rituximab in refractory ITP. Rituximab in the management of chronic immune thrombocytopenic purpura: an effective and safe therapeutic alternative in refractory patients. Ann Hematol. 2006;85(6):400–6.
Dabak V, Hanbali A, Kuriakose P, et al. Can rituximab replace splenectomy in immune thrombocytopenic purpura? Indian J Hematol Blood Transfus. 2009;25(1):6–9.
Arnold DM, Heddle NM, Carruthers J, Cook DJ, Crowther MA, Meyer RM, et al. A pilot randomized trial of adjuvant rituximab or placebo for non-splenectomized patients with immune thrombocytopenia. Blood. 2012;119(6):1356–62.
Ghanima W, Khelif A, Waage A, Michel M, Tjønnfjord GE, Romdhan NB, et al. Rituximab as second-line treatment for adult immune thrombocytopenia (the RITP trial): a multicentre, randomized, double-blind, placebo-controlled trial. Lancet. 2015;385:1653–61.
Moulis G, Sailler L, Sommet A, Lapeyre-Mestre M, Derumeaux H, Adoue D. Rituximab versus splenectomy in persistent or chronic adult primary immune thrombocytopenia: an adjusted comparison of mortality and morbidity. Am J Hematol. 2014;89:41–6.
Stasi R, Pagano A, Stipa E, Amadori S. Rituximab chimeric anti-CD20 monoclonal antibody treatment for adults with chronic idiopathic thrombocytopenic purpura. Blood. 2001;98(4):952–7.
Fda.gov. Hematology/oncology (cancer) approvals and safety notifications: previous news items; 2015. http://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm279177.htm. Cited 6 July 2015.
Arnold DM, Dentali F, Crowther MA, Meyer RM, Cook RJ, Sigouin C, et al. Systematic review: efficacy and safety of rituximab for adults with idiopathic thrombocytopenic purpura. Ann Intern Med. 2007;146(1):25–33.
Godeau B, Porcher R, Fain O, Francois L, Pierre F, Stephane C, et al. Rituximab efficacy and safety in adult splenectomy candidates with chronic immune thrombocytopenic purpura: results of a prospective multicenter phase 2 study. Blood. 2008;112:999–1004.
Acknowledgements
The authors would like to thank Department of Biostatistics and Bioinformatics at King Abdullah International Medical Research Center (KAIMRC), Riyadh for conducting the data analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
About this article
Cite this article
Al Askar, A.S., Shaheen, N.A., Al Zahrani, M. et al. Splenectomy vs. rituximab as a second-line therapy in immune thrombocytopenic purpura: a single center experience. Int J Hematol 107, 69–74 (2018). https://doi.org/10.1007/s12185-017-2325-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-017-2325-y