Abstract
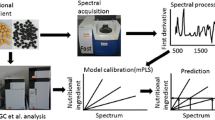
Direct analytical methods assessing lutein content (e.g., high-performance liquid chromatography [HPLC]) are expensive and time-consuming. Their utilization in broad mapping of genetic resources or selecting among hundreds of breeding lines is limited. Therefore, the development of reliable predictive models based on reflectance spectrometry for assessing lutein content would be very useful. This study aims at finding a rapid and accurate lutein quantification method for wheat breeding purposes. Reference samples were represented by hexaploid, tetraploid, and diploid wheat species (119 accessions) cultivated in 2 years. The species Triticum aestivum L. predominated, with 81 accessions. Two near-infrared (NIR) spectroscopy devices were used to collect visible (VIS) and/or NIR spectra: the model FOSS 6500 with dispersive element and wavelength range 400–2500 nm and the Nicolet Antaris II Fourier transform spectrophotometer with wavelength range 1000–2500 nm. HPLC was used as the reference method for assessing lutein content in wheat grain. Sample grinding and proper NIR and VIS spectral combinations were essential for obtaining sufficient predictive parameters in calibration models. The FOSS 6500 calibration models were the most promising (standard error of prediction 0.46–0.78; ratio of performance to deviation 2.36–2.83 mg/kg). NIR spectral instability of ground samples during 12 days in storage was detected and evoked a significant relative increase in lutein content of about 35% using the FOSS 6500 calibration model. After excluding five time-instable spectral regions, prediction differences of lutein content were markedly eliminated and did not exceed 7% in relative terms during 12 days of storage.






Similar content being viewed by others
References
Ahmad FT, Asenstorfer RE, Soriano IR, Mares DJ (2013) Effect of temperature on lutein esterification and lutein stability in wheat grain. J Cereal Sci 58:408–413
Arts ICW, Hollman PCH (2005) Polyphenols and disease risk in epidemiologic studies. Am J Clin Nutr 81:317–325
Baptista MS, Tran CD, Gao G-H (1996) Near-infrared detection of flow injection analysis by acoustooptic tunable filter-based spectrophotometry. Anal Chem 68(6):971–976
Beleggia R, Platani C, Nigro F, Papa R (2011) Yellow pigment determination for single kernels of durum wheat (Triticum durum Desf.) Cereal Chem 88(5):504–508
Bonierbale M, Grüneberg W, Amoros W, Burgos G, Salas E, Porras E, Felde T (2009) Total and individual carotenoid profiles in Solanum phureja cultivated potatoes: II. Development and application of near-infrared reflectance spectroscopy (NIRS) calibrations for germplasm characterization. J Food Compos Anal 22:509–516
Cantor SL, Hoag SW, Ellison CD, Khan MA, Lyon RC (2011) NIR spectroscopy applications in the development of a compacted multiparticulate system for modified release. AAPS PharmSciTech 12(1):262–268
Chen X, Wu J, Zhou S, Yang Y, Ni X, Yang J, Zhu Z, Shi C (2009) Application of near-infrared reflectance spectroscopy to evaluate the lutein and b-carotene in Chinese kale. J Food Compos Anal 22:148–153
Duckworth J (2004) Mathematical data processing. In: (ed.) L. Al-Amoodi, Near infrared spectroscopy in agriculture, agronomy monograph 44, Medison Wisconsin, USA, pp 125–127
Geleta N, Grausgruber H, Vollmann J (2014) Near-infrared spectroscopy of yellow endosperm pigments in wheat. Int J Agr Forest 4(6):446–450
Gonzalez AG, Herrador MA (2007) A practical guide to analytical method validation, including measurement uncertainty and accuracy profiles. Trends Anal Chem 26(3):227–238
Granado F, Olmedilla B, Blanco I (2003) Nutritional and clinical relevance of lutein in human health. Br J Nutr 90(3):487–502
Hansen L, Rose M (1996) Sensory acceptability is inversely related to development of fat rancidity in bread made from stored flour. J Am Diet Assoc 96:792–793
Hidalgo A, Brandolini A, Pompei C, Piscozzi R (2006) Carotenoids and tocols of einkorn wheat (Triticum monococcum ssp. monococcum L.) J Cereal Sci 44:182–193
Hidalgo A, Brandolini A (2008) Kinetics of carotenoids degradationduring the storage of einkorn (Triticum monococum L. spp. monococcum) and bread wheat (Triticum aestivum L. spp. aestivum) flours. J Agric Food Chem 56(23):11300–11305
Jirsa O, Hrušková M, Švec I (2008) Near-infrared prediction of milling and baking parameters of wheat varieties. J Food Eng 87:21–25
Landrum JT, Bone RA (2004) Dietary lutein and zeaxanthin: reducing the risk for macular degeneration. Agro Food Industry Hi-Tech 15:22–25
Leenhardt F, Lyan B, Rock E, Boussard A, Potus J, Chanliaud E, Remesy C (2006) Genetic variability of carotenoid concentration, and lipoxygenase and peroxidase activities among cultivated wheat species and bread wheat varieties. Europ J Agronomy 25:170–176
Mellado-Ortega E, Atienza SG, Hornero-Méndez D (2015) Carotenoid evolution during postharvest storage of durum wheat (Triticum turgidum conv. durum) and tritordeum (Tritordeum Ascherson et Graebner) grains. J Cereal Sci 62:134–142
Mellado-Ortega E, Hornero-Méndez D (2015) Carotenoids in cereals: an ancient resource with present and future applications. Phytochem Rev 14:873–890
Meydani M (2002) Antioxidants in the prevention of chronic diseases. Nutr Clin Care 5:47–49
Miedaner T, Han S, Kessel B, Ouzunova M, Schrag T, Utz FH, Melchinger AE (2015) Prediction of deoxynivalenol and zearalenone concentrations in Fusarium graminearum inoculated backcross populations of maize by symptom rating and near-infrared spectroscopy. Plant Breed 134:529–534
Moncada GW, Martín MIG, Escuredo O, Fischer S, Míguez M (2013) Multivariate calibration by near infrared spectroscopy for the determination of the vitamin E and the antioxidant properties of quinoa. Talanta 116:65–70
Moore J, Hao Z, Zhou K, Luther M, Costa J, Yu L (2005) Carotenoid, tocopherol, phenolic acid, and antioxidant properties of Maryland-grown soft wheat. J Agric Food Chem 53:6649–6657
Pasquini C (2003) Near infrared spectroscopy: fundamentals, practical aspects and analytical applications. J Braz Chem Soc 14(2):198–219
Sujka K, Koczoń P, Ceglińska A, Reder M, Ciemniewska-Zytkiewicz H (2017) The application of FT-IR spectroscopy for quality control of flours obtained from polish producers. J Anal Met Chem: online 2017:1–9. https://doi.org/10.1155/2017/4315678
Tsuchikawa S, Kobori H (2015) A review of recent application of near infrared spectroscopy to wood science and technology. J Wood Sci 61:213–220
Wetzel DL, Sweat JA (1997) FT-NIR comparative quantitative performance for complex samples. In: Progress in Fourier transform spectroscopy. Springer, Vienna, pp 325–327
Weyer L, Lo S-C (2002) Spectra-structure correlations in the near-infrared. In: Griffiths P, Chalmers JM (eds) Handbook of vibrational spectroscopy, vol 4. Wiley, U.K., pp 1817–1837
Ziegler JU, Schweiggert RM, Würschum T, Longin CFH, Carle R (2016) Lipophilic antioxidants in wheat (Triticum spp.): a target for breeding new varieties for future functional cereal products. J Funct Foods 20:594–605
Funding
This work was funded by the Ministry of Agriculture of the Czech Republic under Project Nos. MZE RO 0417 and QJ1510163.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Václav Dvořáček declares that he has no conflict of interest. Lenka Štěrbová declares that she has no conflict of interest. Eva Matějová declares that she has no conflict of interest. Jana Bradová declares that she has no conflict of interest. Jiří Hermuth declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Electronic Supplementary Material
ESM 1
(XLSX 17 kb)
Rights and permissions
About this article
Cite this article
Dvořáček, V., Štěrbová, L., Matějová, E. et al. Reflectance Spectrometry as a Screening Tool for Prediction of Lutein Content in Diverse Wheat Species (Triticum spp.). Food Anal. Methods 11, 2579–2589 (2018). https://doi.org/10.1007/s12161-018-1215-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1215-0