Abstract
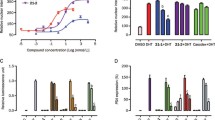
The aims of this study were to evaluate the positive inotropic effect of a new macrocyclic derivative (compound 11) and characterize the molecular mechanism involved in its biological activity. The first step was achieved by synthesis of a macrocyclic derivative involving a series of reactions for the preparation of several steroid derivatives such as (a) steroid-pyrimidinone (3 and 4), (b) steroid-amino (5), (c) steroid-imino (6), (d) ester-steroid (7 and 8), and (e) amido-steroid (9 and 10). Finally, 11 was prepared by removing the tert-butyldimethylsilane fragment of 10. The biological activity of compounds on perfusion pressure and vascular resistance was evaluated on isolated rat heart using the Langendorff model. The inotropic activity of 11 was evaluated in presence of prazosin, metoprolol, indomethacin, nifedipine, and flutamide to characterize its molecular mechanism. Theoretical experiments were carried out with a Docking model, to assess potential interactions of androgen receptor with 11. The results showed that only this macrocyclic derivative exerts changes on perfusion pressure and vascular resistance translated as the positive inotropic effect, and this effect was blocked with flutamide; these data indicate that the positive inotropic activity induced by this macrocyclic derivative was via androgen receptor activation. The theoretical results indicated that the interaction of the macrocyclic derivative with the androgen receptor involves several amino acid residues such as Leu704, Asn705, Met780, Cys784, Met749, Leu762, Phe764, Ser778, and Met787. In conclusion, all these data suggest that the positive inotropic activity of the macrocyclic derivative may depend on its chemical structure.












Similar content being viewed by others
Notes
Physiologic saline solution (Krebs-Henseleit solution; it was actively bubbled with a mixture of O2/CO2 (95:5/5%) and regulated at a pH of 7.4 and 37 °C) was composed of NaCl, 117.8 mmol; KCl, 6 mmol; CaCl2, 1.75 mmol; NaH2PO4, 1.2 mmol; MgSO4, 1.2 mmol; NaHCO3, 24.2 mmol; glucose, 5 mmol; and sodium pyruvate 5 mmol, and the coronary flow was adjusted with a variable speed peristaltic pump.
Krebs-Henseleit solution was actively bubbled with a mixture of O2/CO2 (95:5/5 %) and regulated at a pH of 7.4 and 37°C.
References
Kolbeck R, LaNeave C, Aguirre A, Nosek T, Pannel K (1992) Inotropic influence of macrocyclic polyethers on tracheal smooth muscle. Pharmacol Biochem Behavior 42:645–650
Trevisi L, Bova S, Cargnelli G, Danieli-Betto D, Floreani M, Germinario E, D’Auria M, Luciani S (2000) Callipeltin-A, a cyclic depsipeptide inhibitor of the cardiac sodium-calcium exchanger and positive inotropic agent. Biochem Biophys Res Comm 279:219–222
Watanabe H, Chiba S (1982) Cardiac stimulating effects of macrocyclic polyamines. Japan J Pharmacol 32:394–396
Idée J, Berthommier C, Goulas V, Corot C, Santus R, Hermine C, Schaefer M, Bonnemai B (1998) Haemodynamic effects of macrocyclic and linear gadolinium chelates in rats: role of calcium and transmetallation. Biometals 11:113–112
Bogatskii A, Luk’yanenko N, Savenko T, Vongai V, Nazarov E, Tsymbal I (1984) Effect of the macrocyclic polyester 15-crown-5 on ionic permeability of excitable membranes. Bull Exp Biol Med 98(98):1045–1048
Miyamoto S, Izumi M, Hori M, Kobayashi M, Ozaki H, Karaki H (2000) Xestospongin C, a selective and membrane-permeable inhibitor of IP3 receptor, attenuates the positive inotropic effect of α-adrenergic stimulation in guinea-pig papillary muscle. British J Pharmacol 130:650–654
Kolbeck R, Hendry L, Bransome E, Speir W (1980) Crown ethers which influence cardiac and respiratory muscle contractility. Experientia 40:727–731
Lunardi C, DaSilva R, Bendhack L (2009) New nitric oxide donors based on ruthenium complexes. Brazilian J Med Biol Res 42:87–93
Figueroa-Valverde L, Díaz-Cedillo F, García-Cervera E, Pool-Gómez E, López-Ramos M, Rosas-Nexticapa M, Hau-Heredia L, Sarabia-Alcocer B (2015) Synthesis and antibacterial activity evaluation of two androgen derivatives. Steroids 93:8–15
Bayne K (1996) Revised guide for the care and use of laboratory animals available. Am Physiol Soc 39:208–211
Garcia-Cervera E, Figueroa-Valverde L, Díaz-Cedillo F, López-Ramos M, Rosas-Nexticapa M, Pool-Gómez E, Jarquin-Barberena H, Rodriguez-Hurtado M, Chan-Salvador M (2011) Design and synthesis of a new pirrol-indol derivative with positive inotropic activity. Oriental J Chem 31:31–41
Sarabia-Alcocer B, Figueroa-Valverde L, Díaz-Cedillo F, Hau-Heredia L, Rosas-Nexticapa M, Garcia-Cervera E, Pool-Gómez E, Garcia-Martinez R (2014) Activity induced by a naphthalene-prazosin derivative on ischemia/reperfusion injury in rats. Pharmacol Pharm 5:1130–1142
Hocht C, Opezzo J, Gorzalczany S, Bramuglia G, Tiara C (1997) Una aproximación cinética y dinámica de metildopa en ratas con coartación aórtica mediante microdiálisis. Rev Argentina Cardiol 67:769–773
Bikadi Z, Hazai E (2009) Application of the PM6 semiempirical method to modeling proteins enhances docking accuracy of AutoDock. J Cheminform 1–16
Halgren T (1999) MMFF VI. MMFF94s option for energy minimization studies. J Comput Chem 20:720–729
Morris M, Goodsell D, Hallyday R, Huey R, Hart W, Belew R, Olson A (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 19:1639–1662
Solis F (1981) Minimization by random search techniques. Mathem Meth Oper Res 19–30.
Liebscher J, Hartmann H (1982) Synthese und Abwandlung N-substituierter 2(1 H)-Pyrimidin-thione-Ein einfacher Zugang zu Pyrimidino[2,3-b]1,3,4-thiadiazoliumsalzen. Adv Syn Catal 324:942–946
Önal Z, Korkusuz E, İlhan I (2010) Cyclization reactions of l-pyrimidinyl-3-arylthiourea derivatives with oxalyl dichloride. Heter Comm 16:79–84
Yu A, Sharanin V, Shestopalov V, Nesterov V, Litvinov P, Mortikov Y, Promonenkov V, Shklover V, Struchkov Y (1987) Cyclization reactions of nitriles. 26. Synthesis, structure, and properties of 2-amino-4-methylthio-5-cyano-6(1H)-pyrimidinethione. Chem Heter Comp 23:1105–1112
Zigeuner G, Frank A, Adam A (1970) Reaction of dihydro-6-methyl-4-phenyl-2(1H)-pyrimidinthione with formaldehyde and amines. (heterocyclic compounds, XXVIII). Monats für Chem 101:1788–1793
Parish A, Gilliom R, Purcell W, Browne R, Spirk R, White H (1982) Syntheses and diuretic activity of 1,2-dihydro-2-(3-pyridyl)-3H-pyrido[2,3-d]pyrimidin-4-one and related compounds. J Med Chem 25:98–102
Shawali A, Abdallah M, Mosslhe A, Farghaly T (2000) A facile one-pot regioselective synthesis of [1,2,4]triazolo[4,3-a]5(1H)-pyrimidinones via tandem Japp-Klingemann, Smiles rearrangement, and cyclization reactions. Heteroatom Chem 13:136–140
Shirayev A, Moiseev I, Karpeev S (2005) Synthesis and cis/trans isomerism of N-alkyl-1,3-oxathiolane-2-imines. Arkivok iv: 199–207
Uppiah D, Bhowon D, Jhaumeer M (2009) Synthesis of imines derived from diphenyldisulphide diamine or p-vanillin. E-J. Chem l: S195–200, 2009
Figueroa-Valverde L, Díaz-Cedillo F, García-Cervera E, Rosas-Nexticapan M, Ramos-López M (2013) Design and synthesis of naphthol derivative. Asian J Chem 25:6724–6726
Clark R, Graham W, Winter A (1925) The catalytic preparation of ether from alcohol by means of aluminum oxide. J Am Chem Soc 47:2748–2754
Ravi E, Byun H, Wang S, Bittman R (1994) Preparation of ether-linked 2-acetamido-2-deoxy β-glycolipids via zinc chloride promoted coupling of Ac4GlcNAcCl with lipid hydroxy groups. Tetrahedron Lett 35:505–508
Takekoshi T (1987) Synthesis of high performance aromatic polymers via nucleophilic nitro displacement reaction. Polymer J 19:191–202
Figueroa-Valverde L, Díaz-Cedillo F, Rosas-Nexticapa M, García-Cervera E, Pool-Gomez E, Barberena H, Lopez-Ramos M, Rodriguez-Hurtado F, Chan-Salvador M (2015) Design and synthesis of some carbamazepine derivatives using several strategies. Lett Org Chem 12:394–401
Saxon E, Armstrong E, Bertozzi J (2000) Traceless Staudinger ligation for the chemoselective synthesis of amide bonds. Org Lett 2:2141–2143
Masala S, Taddei M Solid-supported chloro[1,3,5]triazine. A versatile new synthetic auxiliary for the synthesis of amide libraries. Org Lett 1: 1355–1357
Figueroa-Valverde L, Díaz-Cedillo F, Ceballos-Reyes G (2006) Synthesis of pregnenolone-pregnenolone dimer via ring A-ring a connection. J Mex Chem Soc 50:42–45
Ogawa Y, Shibasaki M (1984) Selective removal of tetrahydropyranyl ethers in the presence of t-butyldimethylsilyl ethers. Tetrahedron Lett 25:663–664
Wilson N, Keay B (1996) A mild palladium(II) catalyzed desilylation of phenolic t-butyldimethylsilyl ethers. Tetrahedron Lett 37:153–156
Newton R, Reynolds D, Finch M, Kelly D, Roberts D (1979) An excellent reagent for the removal of the t-butyldimethylsilyl protecting group. Tetrahedron Lett 20:398–3982
Idée J, Berthommier C, Goulas V, Corot C, Santus R, Hermine C, Schaefer M, Bonnemain B (1998) Haemodynamic effects of macrocyclic and linear gadolinium chelates in rats: role of calcium and transmetallation. Biometals 11:113–123
Parker J, Waite M, Pettit G, Daniel L (1988) Stimulation of arachidonic acid release and prostaglandin synthesis by bryostatin. Carcinogenesis 9:1471–1474
Watanabe H, Chiba S (1982) Cardiac stimulating effects of macrocyclic polyamines. Japanese J Pharmacol 32:394–396
Hu Z, Zhang D, Wang D, Sun B, Safoor A, Young C, Lou H, Yuan H (2015) Bisbibenzyls, novel proteasome inhibitors, suppress androgen receptor transcriptional activity and expression accompanied by activation of autophagy in prostate cancer LNCaP cells. Pharm Biol 54:364–374
Thiemermann C, Bowes J, Myint F, Vane J (2012) Inhibition of the activity of poly(ADP ribose) synthetase reduces ischemia–reperfusion injury in the heart and skeletal muscle. Proc Natl Acad Sci 94:679–683
Levine P, Imberg K, Garabedian M, Kirshenbaum K (2012) Multivalent peptidomimetic conjugates: a versatile platform for modulating androgen receptor activity. J Am Chem Soc 134:6912–6915
Calleja C, Pascussi J, Mani J, Maurel P, Vilarem M (1998) The antibiotic rifampicin is a nonsteroidal ligand and activator of the human glucocorticoid receptor. Nat Med 4:92–96
Liu R, Perez J, Liang D, Saven J (2012) Binding site and affinity prediction of general anesthetics to protein targets using docking. Anesth Analg 114:947–955
Rosales M, Correa J (2015) The importance of employing computational resources for the automation of drug discovery. Expert Opinion Drug Dis 10:213–219
Askew E, Gampe R, Stanley T, Faggart J, Wilson E (2007) Modulation of androgen receptor activation function 2 by testosterone and dihydrotestosterone. J Biol Chem 282:25801–25816
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experimental methods used in this investigation were reviewed and approved by the Animal Care and Use Committee of University Autonomous of Campeche (no. PI-420/12) and were in accordance with the guide for the care and use of laboratory animals [10]. Male Wistar rats, weighing 200–250 g, were obtained from University Autonomous of Campeche.
Rights and permissions
About this article
Cite this article
López-Ramos, M., Figueroa-Valverde, L., Herrera-Meza, S. et al. Design and synthesis of a new steroid-macrocyclic derivative with biological activity. J Chem Biol 10, 69–84 (2017). https://doi.org/10.1007/s12154-017-0165-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12154-017-0165-0