Abstract
Objective
The objective of this study was to assess the ability to detect pancreatic metastasis of lung cancer and to clarify the degree of fluorodeoxyglucose (FDG) accumulation and computed tomography (CT) characteristics of pancreatic metastasis from lung cancer.
Methods
A total of 573 patients (415 men and 158 women) with lung cancer were retrospectively evaluated. All patients underwent FDG-positron emission tomography (PET)/CT with contrast-enhanced CT for first=stage (313 patients; initial study group) or follow-up study (260 patients; follow-up study group). A lesion was regarded as positive for metastasis on the basis of visual judgment of the degree of increased metabolism by two experienced and independent interpreters, supported by semiquantitative evaluation on the basis of calculation of the maximum standardized uptake value (SUVmax).
Results
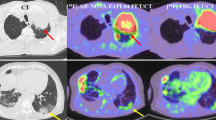
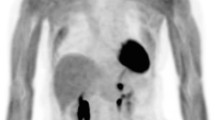
Abnormal accumulations in the pancreas were detected in 5 of 313 patients (1.60%) in the initial study group, and 6 of 260 patients (2.31%) in the follow-up study group. Seven of these patients had adenocarcinoma, three had small cell carcinoma, and the rest had large cell endocrine carcinoma. Tumor sizes (longitudinal diameter), measured by CT, of these 11 patients ranged from 6 mm to 52 mm (mean ± SD 8.3 mm ± 11.9 mm), and SUVmax for 1 h ranged from 3.37 to 11.1 (mean ± SD 6.12 ± 2.43). Three of these pancreatic lesions were difficult to determine by routine transaxial images, and detection was obvious only by thin-slice images or multiplanar reconstruction images. Contrast-enhanced CT showed gradual fill-in from the peripheral portion to the center. In addition, 10 of 11 cases did not show main pancreatic duct dilatation even if the tumor size was large.
Conclusions
Metastases to the pancreas in lung cancer patients are not so rare and radiologists first have an important role to detect the pancreatic mass and then suggest to metastasis as the likely diagnosis. For this purpose, FDG-PET/CT has an advantage in depicting unsuspected pancreatic metastasis from lung cancer, particularly that which is not detected by CT alone.
Similar content being viewed by others
References
Rumancik WM, Megihow AJ, Bosniak MA, Hilton S. Metastatic disease to the pancreas: evaluation by computed tomography. J Comput Assist Tomogr 1984;8:829–834.
Klein KA, Stephens DH, Welch TJ. CT characteristics of metastatic disease of the pancreas. Radiographics 1998;18:369–378.
Ferrozzi F, Bova D, Campodonico F, Chiara FD, Passari A, Bassi P. Pancreatic metastasis: CT assessment. Eur Radiol 1997;7:241–245.
Scatarige JC, Horton KM, Sheth S, Fishman EK. Pancreatic parenchymal metastases: observations on helical CT. AJR Am J Roentgenol 2001;176:695–699.
Merkle EM, Boaz T, Kolokythas O, Haaga JR, Lewin JS, Brambs HJ. Metastases to the pancreas. Br J Radiol 1998;71:1208–1214.
Maeno T, Satoh H, Ishikawa H, Yamashita YT, Naito T, Fujiwara M, et al. Patterns of pancreatic metastasis from lung cancer. Anticancer Res 1998;18:2881–2884.
García Viadl C, Carrillo E, Barreiro B. Solitary metastasis to the pancreas in a patient with lung cancer. Arch Bronconerumol 2003;39:601.
Liratzopoulos N, Efremidou EI, Papageorgiou MS, Romanidis K, Minopoulos GGJ, Manolas J. Extrahepatic biliary obstruction due to a solitary pancreatic metastasis of squamous cell lung carcinoma: case report. J Gastrointest Liver Dis 2006;15:73–75.
Johnson DH, Hainsworth JD, Greco FA. Extrahepatic biliary obstruction caused by small-cell lung cancer. Ann Intern Med 1985;102:487–490.
Chowhan NM, Madajewicz S. Management of metastasesinduced acute pancreatitis in small cell carcinoma of the lung. Cancer 1990;65:1445–1448.
Kim KH, Kim CD, Lee SJ, Lee G, Jean YT, Lee SC, et al. Metastasis-induced acute pancreatitis in a patient with small cell carcinoma of the lung. J Korean Med Sci 1999;14:107–109.
Lowe J, Naunheim S. Current role of positron emission tomography in thoracic oncology. Thorax 1998;53:703–712.
Gambihir S, Czernin J, Schwimmer J, Silverman DHS, Coleman RE, Phelps ME. A tabulated summary to the FDG PET literature. J Nucl Med 2001;42:1S–71S.
Towensend DW, Camey IPJ, Yap JT, Hall NC. PET/CT today and tomorrow. J Nucl Med; 45:4S–14S.
Schoder H, Gonen M. Screening for cancer with PET and PET/CT: potential and limitations. J Nucl Med 2007;48:4S–18S.
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, et al. Staging of non-small-cell lung cancer with integrated positron emission tomography and computed tomography. New Engl J Med 2003;348:2500–2507.
Antoch C, Stattaus J, Nemat AT, Mamitz AT, Beyer T, Kuehl H, et al. Non small cell lung cancer dual-modality PET/CT in preoperative staging. Radiology 2003;229:526–533.
Shim SS, Lee LS, Kim BT, Chung MJ, Lee EJ, Han J, et al. Non small cell lung cancer preoperative comparison of integrated FDG PET/CT and CT alone for preoperative staging. Radiology 2005;236:1101–1109.
Seki M, Tsuchita E, Hori M, Nakagawa K, Ohta H, Ueno M, et al. Pancreatic metastasis from a lung cancer. Int J Pancreatol 1998;24:55–59.
Muranaka T, Teshima K, Honda H, Nanjo T, Hanada K, Oshiumi Y. Computed tomography and histologic appearance of pancreatic metastases from distant source. Acta Radiol 1989;30:615–619.
Higashi T, Saga T, Nakamoto Y, Ishimori T, Fujimoto K, Doi R, et al. Diagnosis of pancreatic cancer using fluorine-18 fluorodeoxyglucose positron emission tomography (FDG PET): usefulness and limitations in “clinical reality”. Ann Nucl Med 2003;17:261–279.
Nakamoto Y, Higashi T, Sakahara H, Tamaki N, Itoh K, Iimura M, et al. Evaluation of pancreatic islet cell tumors by fluorine-18 fluorodeoxyglucose positron emission tomography; comparison with other modalities. Clin Nucl Med 2002;25:115–119.
Nakamoto Y, Saga T, Ishimori T, Higashi T, Mamede M, Okazaki K, et al. FDG-PET of autoimmune-related pancreatitis: preliminary results. Eur J Nucl Med 2000;27:1835–1838.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sato, M., Okumura, T., Kaito, K. et al. Usefulness of FDG-PET/CT in the detection of pancreatic metastases from lung cancer. Ann Nucl Med 23, 49–57 (2009). https://doi.org/10.1007/s12149-008-0205-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-008-0205-5