Abstract
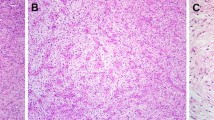
We present a 49 year old female with a diagnostically challenging myxoid solitary fibrous tumor arising in the soft tissue of the neck. The tumor was diffusely positive for CD34 and STAT6 on immunohistochemistry.


Similar content being viewed by others
References
Demicco EG, Park MS, Araujo DM, Fox PS, Bassett RL, Pollock RE, et al. Solitary fibrous tumor: a clinicopathological study of 110 cases and proposed risk assessment model. Mod Pathol. 2012;25(9):1298–306. https://doi.org/10.1038/modpathol.2012.83.
Bowe SN, Wakely PE Jr, Ozer E. Head and neck solitary fibrous tumors: diagnostic and therapeutic challenges. Laryngoscope. 2012;122(8):1748–55. https://doi.org/10.1002/lary.23350.
Stanisce L, Ahmad N, Levin K, Deckard N, Enriquez M, Brody J, et al. Solitary fibrous tumors in the head and neck: comprehensive review and analysis. Head Neck Pathol. 2019. https://doi.org/10.1007/s12105-019-01058-6.
Gold JS, Antonescu CR, Hajdu C, Ferrone CR, Hussain M, Lewis JJ, et al. Clinicopathologic correlates of solitary fibrous tumors. Cancer. 2002;94(4):1057–68.
England DM, Hochholzer L, McCarthy MJ. Localized benign and malignant fibrous tumors of the pleura. A clinicopathologic review of 223 cases. Am J Surg Pathol. 1989;13(8):640–58. https://doi.org/10.1097/00000478-198908000-00003.
Smith SC, Gooding WE, Elkins M, Patel RM, Harms PW, McDaniel AS, et al. Solitary fibrous tumors of the head and neck: a multi-institutional clinicopathologic study. Am J Surg Pathol. 2017;41(12):1642–56. https://doi.org/10.1097/pas.0000000000000940.
Cox DP, Daniels T, Jordan RC. Solitary fibrous tumor of the head and neck. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110(1):79–84. https://doi.org/10.1016/j.tripleo.2010.01.023.
Lau SK, Weiss LM, Chu PG. Myxoid solitary fibrous tumor: a clinicopathologic study of three cases. Virchows Arch Int J Pathol. 2009;454(2):189–94. https://doi.org/10.1007/s00428-008-0721-7.
Kim JH, Kim DC, Lee R, Shin CH, Han YS, Chung SH, et al. Myxoid solitary fibrous tumor on the scalp. Arch Craniofac Surg. 2017;18(4):269–72. https://doi.org/10.7181/acfs.2017.18.4.269.
Han H, Ahn S, Hwangbo W, Chae YS. Myxoid solitary fibrous tumor of the central nervous system. Korean J Pathol. 2013;47(6):505–6. https://doi.org/10.4132/koreanjpathol.2013.47.6.505.
Dantey K, Cooper K. Myxoid solitary fibrous tumor: a study of three cases. Int J Surg Pathol. 2013;21(4):358–62. https://doi.org/10.1177/1066896912470166.
Lee HJ, Lee SH, Roh MR. Cutaneous myxoid solitary fibrous tumour. Clin Exp Dermatol. 2012;37(2):197–9. https://doi.org/10.1111/j.1365-2230.2011.04165.x.
Wei YC, Li CF, Sung MT, Chen YT, Ko SF, Eng HL, et al. Primary myxoid solitary fibrous tumor involving the seminal vesicle. Pathol Int. 2006;56(10):642–4. https://doi.org/10.1111/j.1440-1827.2006.02022.x.
de Saint Aubain Somerhausen N, Rubin BP, Fletcher CD. Myxoid solitary fibrous tumor: a study of seven cases with emphasis on differential diagnosis. Mod Pathol. 1999;12(5):463–71.
Witkin GB, Rosai J. Solitary fibrous tumor of the upper respiratory tract. A report of six cases. Am J Surg Pathol. 1991;15(9):842–8. https://doi.org/10.1097/00000478-199109000-00004.
Park S-J, Lee YH, Lee KY, Oh KH, Kim Y. A solitary fibrous tumor of the subglottic larynx: case report and literature review. Balk Med J. 2016;33(6):698–700. https://doi.org/10.5152/balkanmedj.2016.151069.
Thompson LDR, Wei C, Rooper LM, Lau SK. Thyroid gland solitary fibrous tumor: report of 3 cases and a comprehensive review of the literature. Head Neck Pathol. 2019. https://doi.org/10.1007/s12105-019-01012-6.
Sousa AA, Souto GR, Sousa IA, Mesquita RA, Gomez RS, Jham BC. Solitary fibrous tumor of the parotid gland: case report. J Clin Exp Dent. 2013;5(4):e208–11. https://doi.org/10.4317/jced.51103.
Liu Y, Li K, Shi H, Tao X. Solitary fibrous tumours in the extracranial head and neck region: correlation of CT and MR features with pathologic findings. Radiol Med (Torino). 2014;119(12):910–9. https://doi.org/10.1007/s11547-014-0409-9.
Lee JY, Park SE, Shin SJ, Kim CW, Kim SS, Kim KH. Solitary fibrous tumor with myxoid stromal change. Am J Dermatopathol. 2015;37(7):570–3. https://doi.org/10.1097/dad.0000000000000154.
Robinson DR, Wu YM, Kalyana-Sundaram S, Cao X, Lonigro RJ, Sung YS, et al. Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat Genet. 2013;45(2):180–5. https://doi.org/10.1038/ng.2509.
Mohajeri A, Tayebwa J, Collin A, Nilsson J, Magnusson L, von Steyern FV, et al. Comprehensive genetic analysis identifies a pathognomonic NAB2/STAT6 fusion gene, nonrandom secondary genomic imbalances, and a characteristic gene expression profile in solitary fibrous tumor. Genes Chromosomes Cancer. 2013;52(10):873–86. https://doi.org/10.1002/gcc.22083.
Chmielecki J, Crago AM, Rosenberg M, O’Connor R, Walker SR, Ambrogio L, et al. Whole-exome sequencing identifies a recurrent NAB2-STAT6 fusion in solitary fibrous tumors. Nat Genet. 2013;45(2):131–2. https://doi.org/10.1038/ng.2522.
Barthelmess S, Geddert H, Boltze C, Moskalev EA, Bieg M, Sirbu H, et al. Solitary fibrous tumors/hemangiopericytomas with different variants of the NAB2-STAT6 gene fusion are characterized by specific histomorphology and distinct clinicopathological features. Am J Pathol. 2014;184(4):1209–18. https://doi.org/10.1016/j.ajpath.2013.12.016.
Demicco EG, Harms PW, Patel RM, Smith SC, Ingram D, Torres K, et al. Extensive survey of STAT6 expression in a large series of mesenchymal tumors. Am J Clin Pathol. 2015;143(5):672–82. https://doi.org/10.1309/ajcpn25njtounpnf.
Rekhi B, Shetty O, Tripathi P, Bapat P, Ramadwar M, Bajpai J, et al. Molecular characterization of a series of solitary fibrous tumors, including immunohistochemical expression of STAT6 and NATB2-STAT6 fusion transcripts, using Reverse Transcriptase (RT)-Polymerase chain reaction (PCR) technique: an Indian experience. Pathol Res Pract. 2017;213(11):1404–11. https://doi.org/10.1016/j.prp.2017.08.011.
Saeed O, Zhang S, Cheng L, Lin J, Alruwaii F, Chen S. STAT6 expression in solitary fibrous tumor and histologic mimics: a single institution experience. Appl Immunohistochem Mol Morphol. 2019. https://doi.org/10.1097/pai.0000000000000745.
Doyle LA, Vivero M, Fletcher CD, Mertens F, Hornick JL. Nuclear expression of STAT6 distinguishes solitary fibrous tumor from histologic mimics. Mod Pathol. 2014;27(3):390–5. https://doi.org/10.1038/modpathol.2013.164.
Doyle LA, Tao D, Marino-Enriquez A. STAT6 is amplified in a subset of dedifferentiated liposarcoma. Mod Pathol. 2014;27(9):1231–7. https://doi.org/10.1038/modpathol.2013.247.
Cheah AL, Billings SD, Goldblum JR, Carver P, Tanas MZ, Rubin BP. STAT6 rabbit monoclonal antibody is a robust diagnostic tool for the distinction of solitary fibrous tumour from its mimics. Pathology. 2014;46(5):389–95. https://doi.org/10.1097/pat.0000000000000122.
Creytens D, Libbrecht L, Ferdinande L. Nuclear expression of STAT6 in dedifferentiated liposarcomas with a solitary fibrous tumor-like morphology: a diagnostic pitfall. Appl Immunohistochem Mol Morphol. 2015;23(6):462–3. https://doi.org/10.1097/pai.0000000000000081.
Koelsche C, Schweizer L, Renner M, Warth A, Jones DT, Sahm F, et al. Nuclear relocation of STAT6 reliably predicts NAB2-STAT6 fusion for the diagnosis of solitary fibrous tumour. Histopathology. 2014;65(5):613–22. https://doi.org/10.1111/his.12431.
Yoshida A, Tsuta K, Ohno M, Yoshida M, Narita Y, Kawai A, et al. STAT6 immunohistochemistry is helpful in the diagnosis of solitary fibrous tumors. Am J Surg Pathol. 2014;38(4):552–9. https://doi.org/10.1097/pas.0000000000000137.
Matsuyama A, Hisaoka M, Shimajiri S, Hayashi T, Imamura T, Ishida T, et al. Molecular detection of FUS-CREB3L2 fusion transcripts in low-grade fibromyxoid sarcoma using formalin-fixed, paraffin-embedded tissue specimens. Am J Surg Pathol. 2006;30(9):1077–84. https://doi.org/10.1097/01.pas.0000209830.24230.1f.
Lee ATJ, Thway K, Huang PH, Jones RL. Clinical and molecular spectrum of liposarcoma. J Clin Oncol. 2018;36(2):151–9. https://doi.org/10.1200/jco.2017.74.9598.
Tornkvist M, Brodin B, Bartolazzi A, Larsson O. A novel type of SYT/SSX fusion: methodological and biological implications. Mod Pathol. 2002;15(6):679–85. https://doi.org/10.1038/modpathol.3880587.
Magro G, Spadola S, Motta F, Palazzo J, Catalano F, Vecchio GM, et al. STAT6 expression in spindle cell lesions of the breast: an immunohistochemical study of 48 cases. Pathol Res Pract. 2018;214(10):1544–9. https://doi.org/10.1016/j.prp.2018.07.011.
Funding
No funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical Approval
It is our institution’s policy not to require formal ethical approval for reports on up to two patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, B., Tay, S.Y. & Petersson, F. Cervical Myxoid Solitary Fibrous Tumor: Report of an Unusual Variant and a Brief Overview of the Literature. Head and Neck Pathol 14, 852–858 (2020). https://doi.org/10.1007/s12105-019-01107-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-019-01107-0