Abstract
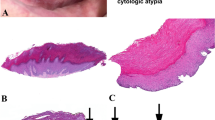
Proliferative verrucous leukoplakia (PVL) is defined as an aggressive, relentless and recalcitrant form of leukoplakia that has a high propensity for malignant transformation. The aim of this study was to evaluate the malignant potential of PVL and determine its possible association with high-risk human papillomavirus (HPV). Twenty cases with a clinical and biopsy proven diagnosis of PVL were collected from the University of Florida Oral Medicine clinic database. Immunohistochemistry was performed to evaluate the expression of p16INK4A and p53 genes in the PVL lesions. The lesions were also tested for high-risk HPV by DNA in-situ hybridization. The average age of the patients at the time of first biopsy was 62.7 years. Most patients had multiple sites of involvement, gingiva being the most common location. The lesions progressed to malignancy in approximately 50% of patients. The expression of p16INK4A gene was considered negative, with at least a 50–65% immunoreactivity observed in only three cases that progressed to malignancy. No expression of high-risk HPV was detected, whereas p53 staining was positive in less than 25% of the cells demonstrating gene expression. No definite association between PVL and high-risk HPV infection could be established. Due to the high transformation potential of PVL, early recognition with aggressive treatment, including multiple biopsies, and continued close clinical follow-up, remain the mainstay of favorable management of this condition.



Similar content being viewed by others
References
Hansen LS, Olson JA, Silverman S Jr. Proliferative verrucous leukoplakia. A long-term study of thirty patients. Oral Surg Oral Med Oral Pathol. 1985;60:285–98.
El-Naggar AK, Chan JKC, Grandis JR, Takata T, Slootweg PJ. World Health Organization classification of head and neck tumors. 4th ed. Lyon: IARC; 2017.
Palefsky JM, Silverman S Jr, Abdel-Salaam M, Daniels TE, Greenspan JS. Association between proliferative verrucous leukoplakia and infection with human papillomavirus type 16. J Oral Pathol Med. 1995;24:193–7.
Shear M, Pindborg JJ. Verrucous hyperplasia of the oral mucosa. Cancer. 1980;46:1855–62.
Feller L, Wood NH, Raubenheimer EJ. Proliferative verrucous leukoplakia and field cancerization: report of a case. J Int Acad Periodontol. 2006;8:67–70.
Bagan JV, Murillo J, Poveda R, Gavalda C, Jimenez Y, Scully C. Proliferative verrucous leukoplakia: unusual locations of oral squamous cell carcinomas, and field cancerization as shown by the appearance of multiple OSCCs. Oral Oncol. 2004;40:440–3.
Eversole LR. Papillary lesions of the oral cavity: relationship to human papillomaviruses. J Calif Dent Assoc. 2000;28:922–7.
Campisi G, Giovannelli L, Ammatuna P, Capra G, Colella G, Di Liberto C, et al. Proliferative verrucous vs conventional leukoplakia: no significantly increased risk of HPV infection. Oral Oncol. 2004;40:835–40.
Fettig A, Pogrel MA, Silverman S Jr, Bramanti TE, Da Costa M, Regezi JA. Proliferative verrucous leukoplakia of the gingiva. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90:723–30.
Bagan JV, Jimenez Y, Murillo J, Gavalda C, Poveda R, Scully C, et al. Lack of association between proliferative verrucous leukoplakia and human papillomavirus infection. J Oral Maxillofac Surg. 2007;65:46–9.
Hay CM, Lachance JA, Lucas FL, Smith KA, Jones MA. Biomarkers p16, human papillomavirus and p53 predict recurrence and survival in early stage squamous cell carcinoma of the vulva. J Low Genit Tract Dis. 2016;20:252–6.
Scheffner M, Munger K, Byrne JC, Howley PM. The state of the p53 and retinoblastoma genes in human cervical carcinoma cell lines. Proc Natl Acad USA. 1991;88:5523–7.
Adegboyega PA, Boromound N, Freeman DH. Diagnostic utility of cell cycle and apoptosis regulatory proteins in verrucous squamous carcinoma. Appl Immunohistochem Mol Morphol. 2005;13:171–7.
Gopalakrishnan R, Weghorst CM, Lehman TA, Calvert RJ, Bijur G, Sabourin CL, et al. Mutated and wild-type p53 expression and HPV integration in proliferative verrucous leukoplakia and oral squamous cell carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997;83:471–7.
Lewis JS Jr, Chernock RD, Ma XJ, Flanagan JJ, Luo Y, Gao G, et al. Partial p16 staining in oropharyngeal squamous cell carcinoma: extent and pattern correlate with human papillomavirus RNA status. Mod Pathol. 2012;25:1212–20.
Cerero-Lapiedra R, Balade-Martinez D, Moreno-Lopez LA, Esparza-Gomez G, Bagan JV. Proliferative verrucous leukoplakia: a proposal for diagnostic criteria. Med Oral Patol Oral Cir Bucal. 2010;15:e839–45.
Carrard VC, Brouns ER, van der Waal I. Proliferative verrucous leukoplakia; a critical appraisal of the diagnostic criteria. Med Oral Patol Oral Cir Bucal. 2013;18:e411–3.
Batsakis JG, Suarez P, el-Naggar AK. Proliferative verrucous leukoplakia and its related lesions. Oral Oncol. 1999;35:354–9.
Slootweg PJ, Muller H. Verrucous hyperplasia or verrucous carcinoma. An analysis of 27 patients. J Maxillofac Surg. 1983;11:13–9.
Murrah VA, Batsakis JG. Proliferative verrucous leukoplakia and verrucous hyperplasia. Ann Otol Rhinol Laryngol. 1994;103:660–3.
Silverman S Jr, Gorsky M. Proliferative verrucous leukoplakia: a follow-up study of 54 cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997;84:154–7.
Bagan JV, Jimenez Y, Sanchis JM, Poveda R, Milian MA, Murillo J, et al. Proliferative verrucous leukoplakia: high incidence of gingival squamous cell carcinoma. J Oral Pathol Med. 2003;32:379–82.
Bagan JV, Jimenez-Soriano Y, Diaz-Fernandez JM, Murillo-Cortes J, Sanchis-Bielsa JM, Poveda-Roda R, et al. Malignant transformation of proliferative verrucous leukoplakia to oral squamous cell carcinoma: a series of 55 cases. Oral Oncol. 2011;47:732–5.
Reuschenbach M, Waterboer T, Wallin KL, Einenkel J, Dillner J, Hamsikova E, et al. Characterization of humoral immune responses against p16, p53, HPV16 E6 and HPV16 E7 in patients with HPV-associated cancers. Int J Cancer. 2008;123:2626–31.
Munger K, Werness BA, Dyson N, Phelps WC, Harlow E, Howley PM. Complex formation of human papillomavirus E7 proteins with the retinoblastoma tumor suppressor gene product. EMBO J. 1989;8:4099–105.
Benson E, Li R, Eisele D, Fakhry C. The clinical impact of HPV tumor status upon head and neck squamous cell carcinomas. Oral Oncol. 2014;50:565–74.
Akrish S, Ben-Izhak O, Sabo E, Rachmiel A. Oral squamous cell carcinoma associated with proliferative verrucous leukoplakia compared with conventional squamous cell carcinoma-a clinical, histologic and immunohistochemical study. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;119:318–25.
Thennavan A, Byatnal AA, Solomon MC, Radhakrishnan RA. The role of Ki-67, p16, CD34, Bcl-2, cyclooxygenase-2 in the pathogenesis of proliferative verrucous leukoplakia. Indian J Cancer. 2015;52:498–502.
Femiano F, Gombos F, Scully C. Oral proliferative verrucous leukoplakia (PVL); open trial of surgery compared with combined therapy using surgery and methisoprinol in papillomavirus-related PVL. Int J Oral Maxillofac Surg. 2001;30:318–22.
Wang SS, Hildesheim A. Chapter 5: Viral and host factors in human papillomavirus persistence and progression. J Natl Cancer Inst Monogr. 2003;2003(31):35–40.
Santos M, Montagut C, Mellado B, Garcia A, Ramon y Cajal S, Cardesa A, et al. Immunohistochemical staining for p16 and p53 in premalignant and malignant epithelial lesions of the vulva. Int J Gynecol Pathol. 2004;23:206–14.
Lee JJ, Hong WK, Hittelman WN, Mao L, Lotan R, Shin DM, et al. Predicting cancer development in oral leukoplakia: ten years of translational research. Clin Cancer Res. 2000;6:1702–10.
Acin S, Li Z, Mejia O, Roop DR, El-Naggar AK, Caulin C. Gain-of-function mutant p53 but not p53 deletion promotes head and neck cancer progression in response to oncogenic K-ras. J Pathol. 2011;225:479–89.
Shin DM, Lee JS, Lippman SM, Lee JJ, Tu ZN, Choi G, et al. p53 expressions: predicting recurrence and second primary tumors in head and neck squamous cell carcinoma. J Natl Cancer Inst. 1996;88:519–29.
Zakrzewska JM, Lopes V, Speight P, Hopper C. Proliferative verrucous leukoplakia: a report of ten cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;82:396–401.
Poveda-Roda R, Bagan JV, Jimenez-Soriano Y, Diaz-Fernandez JM, Gavalda-Esteve C. Retinoids and proliferative verrucous leukoplakia (PVL). A preliminary study. Med Oral Patol Oral Cir Bucal. 2010;15:e3–9.
Acknowledgements
The authors thank Dr. Elizabeth Bilodeau, Associate Professor, Oral & Maxillofacial Pathology, University of Pittsburgh, for providing assistance with p53 immunohistochemistry and high-risk HPV in-situ hybridization techniques.
Funding
No funding was required for completion of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest or other disclosures.
Ethical Approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
The study was conducted after obtaining an approval from the University of Florida Institutional Review Board.
Rights and permissions
About this article
Cite this article
Upadhyaya, J.D., Fitzpatrick, S.G., Islam, M.N. et al. A Retrospective 20-Year Analysis of Proliferative Verrucous Leukoplakia and Its Progression to Malignancy and Association with High-risk Human Papillomavirus. Head and Neck Pathol 12, 500–510 (2018). https://doi.org/10.1007/s12105-018-0893-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-018-0893-7