Abstract
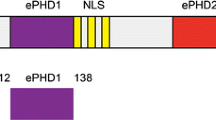
TAR RNA binding protein (TRBP) is a double-stranded RNA binding protein involved in various biological processes like cell growth, development, death, etc. The protein exists as two isoforms TRBP2 and TRBP1. TRBP2 contains additional 21 amino acids at its N-terminus, which are proposed to be involved in its membrane localization, when compared to TRBP1. The resonance assignment (19–228) of the double-stranded RNA binding domains (dsRBD 1 and 2) of TRBP2 has been reported earlier. Here, we report 1H, 13C and 15N resonance assignment for dsRBD1 of TRBP2 (1–105) containing the additional N-terminal residues. This assignment will provide deeper insights to understand the effect of these residues on the structure and dynamics of TRBP2 and would therefore help in further elucidating the differences in the role of these isoforms.




Similar content being viewed by others
References
Benoit MP, Plevin MJ (2013) Backbone resonance assignments of the micro-RNA precursor binding region of human. TRBP Biomol NMR Assign 7:229–233. https://doi.org/10.1007/s12104-012-9416-8
Bhatia P, Raina S, Chugh J, Sharma S (2015) miRNAs: early prognostic biomarkers for type 2 diabetes mellitus? Biomark Med 9:1025–1040
Cavanagh J (2007) Protein NMR spectroscopy: principles and practice, 2nd edn. Academic Press, Amsterdam
Daniels SM, Gatignol A (2012) The multiple functions of TRBP, at the hub of cell responses to viruses, stress, and cancer. Microbiol Mol Biol Rev 76:652–666. https://doi.org/10.1128/MMBR.00012-12
Daniels SM et al (2009) Characterization of the TRBP domain required for dicer interaction and function in RNA interference. BMC Mol Biol 10:38. https://doi.org/10.1186/1471-2199-10-38
Davis IW et al (2007) MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res 35:W375–W383. https://doi.org/10.1093/nar/gkm216
Gatignol A, Buckler-White A, Berkhout B, Jeang KT (1991) Characterization of a human TAR RNA-binding protein that activates the HIV-1 LTR. Science 251:1597–1600
Kim VN, Han J, Siomi MC (2009) Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol 10:126–139. https://doi.org/10.1038/nrm2632
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291 doi. https://doi.org/10.1107/S0021889892009944
Noland CL, Ma E, Doudna JA (2011) siRNA repositioning for guide strand selection by human Dicer complexes. Mol Cell 43:110–121. https://doi.org/10.1016/j.molcel.2011.05.028
Panchal SC, Bhavesh NS, Hosur RV (2001) Improved 3D triple resonance experiments, HNN and HN(C)N, for HN and 15N sequential correlations in (13C, 15N) labeled proteins: application to unfolded proteins. J Biomol NMR 20:135–147
Shen Y et al (2008) Consistent blind protein structure generation from NMR chemical shift data. Proc Natl Acad Sci USA 105:4685–4690. https://doi.org/10.1073/pnas.0800256105
Shen Y, Vernon R, Baker D, Bax A (2009) De novo protein structure generation from incomplete chemical shift assignments. J Biomol NMR 43:63–78. https://doi.org/10.1007/s10858-008-9288-5
Wilson RC, Tambe A, Kidwell MA, Noland CL, Schneider CP, Doudna JA (2015) Dicer-TRBP complex formation ensures accurate mammalian microRNA biogenesis. Mol Cell 57:397–407. https://doi.org/10.1016/j.molcel.2014.11.030
Wishart DS et al (1995) 1H, 13C and 15N chemical shift referencing in biomolecular NMR. J Biomol NMR 6:135–140
Yamashita S et al (2011) Structures of the first and second double-stranded RNA-binding domains of human TAR RNA-binding protein. Protein Sci 20:118–130. https://doi.org/10.1002/pro.543
Acknowledgements
Authors thank Prof Jennifer Doudna for the TRBP clones. JC thanks IISER Pune for the start-up grant. JC acknowledges extra mural grant from Department of Science & Technology, India (EMR/2015/001966). SS acknowledges Ramalingaswami fellowship (BT/RLF/Re-entry/11/2012) and SPPU Research and Development grant to Department of Biotechnology. HP thanks IISER Pune for the fellowship; PVJ and ASN acknowledge Department of Biotechnology, India for their Masters in Biotechnology fellowship. Authors thank High-field NMR facility at IISER Pune funded by DST-FIST and IISER Pune.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paithankar, H., Jadhav, P.V., Naglekar, A.S. et al. 1H, 13C and 15N resonance assignment of domain 1 of trans-activation response element (TAR) RNA binding protein isoform 1 (TRBP2) and its comparison with that of isoform 2 (TRBP1). Biomol NMR Assign 12, 189–194 (2018). https://doi.org/10.1007/s12104-018-9807-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-018-9807-6