Abstract
Influenza continues to baffle humans by its constantly changing nature. The twenty-first century has witnessed considerable advances in the understanding of the influenza viral pathogenesis, its synergy with bacterial infections and diagnostic methods. However, challenges continue: to find a less expensive and more reliable point-of-care test for use in developing countries, to produce more efficacious antiviral drugs, to explore ways to combat emerging antiviral resistance and to develop vaccines that can either be produced in a shorter production time or can overcome the need for annual matching with the circulating influenza strains. Most importantly for India, as a nation that suffered the highest mortality in the influenza pandemic 1918, there is an urgent need to gear up our existing preparedness for the next pandemic which is capable to hit at any moment in time.
Similar content being viewed by others
Introduction
A century has elapsed since the most fatal epidemic known in mankind which killed over 50 million people – the Spanish influenza pandemic of 1918. India was the focal point of this pandemic in terms of mortality with deaths between 10 and 20 million (about 8% of the population at that time). The epidemic in India originated in Bombay in September 1918 where the troops infected with influenza returned from the First World War [1]. Several pandemics have followed with lower mortality and morbidity in the years 1957 (Asian flu), 1968 (Hongkong flu) and 2009 (Swine flu) [2]. Despite the advances in diagnostics, antiviral drugs and vaccines the threat of a severe pandemic like 1918 looms large as the novel Influenza A viruses of avian or swine origin continue to infect the humans.
Sporadic cases of Influenza occur throughout the year. Transmission of the virus depends on two important environmental factors – temperature and humidity. Epidemics in temperate zones generally occur in dry and cold winters as the influenza virus is known to be more stable in cold with greatest transmission at 5 °C and least above 30 °C [3]. However these transmission dynamics cannot be applied to tropical countries where the average humidity and temperatures are higher and epidemics are observed during the monsoons. Diurnal variations in humidity and temperature and household exposures have been postulated as possible mechanisms in these regions though the exact reasons for monsoon peaks in tropics remain elusive [4].
As of 14th July 2019, the data from National Centre of Disease Control reported over 26,000 cases and 1000 deaths due to influenza since the beginning of this year even when the peak season had just begun [5]. Influenza surveillance studies from India have identified three major patterns of influenza circulation: Srinagar situated in north most part (latitude of 34° N) has its peak circulation like temperate regions during the winter (January–April) probably due to the climatic similarity. Late monsoon peaks have been observed at the opposite end in Chennai and Vellore situated in the South west. Whereas rest of India has peak activity coinciding with the rains (June–October) with minor peaks during winter [6]. This has a significant relevance for vaccination strategy for India.
The incubation period is about 1–2 d with a range of 1–4 d [7]. The viral shedding occurs from the day prior to symptoms and lasts for about 3–5 d in adults and for several weeks in young children and immunocompromised. The primary mode of transmission of the virus is by large size droplets. Particles <10 μm in diameter are more likely to cause infection in the lower respiratory tract.
Influenza Virus
Influenza viruses A, B and C belong to Orthomyxoviridae family and are enveloped negative strand RNA viruses. Negative means they have opposite polarity of the RNA that forms template for protein synthesis required for their viral replication. Though the three viruses have a common genetic ancestry, genetic reassortment is limited to within each specific virus genus and not across the types.
The surface glycoproteins present on the envelope of the virus – hemagglutinin (HA), and neuraminidase (NA) play a critical role in their pathogenesis as well as form the main targets for neutralizing antibodies against the virus.
In 1980, WHO established a standard nomenclature for identifying the influenza viruses. It consists of the antigenic type (A, B or C); host of origin (in case the virus has not been isolated from humans); geographical region of origin; number of lineage; year of isolation and, for Influenza A viruses only, HA and NA subtype, described by letter and number, H1 to H16 known to date, and N1 to N9 [8]. For example, the pandemic Influenza A of 2009 is denoted as A/California/04/2009(H1N1) for Influenza type A, isolated first in California, lineage number 04, year of 2009 and type H1N1.
Pathogenesis
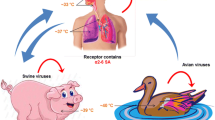
HA binds to the sialic acid residues expressed on the columnar epithelial cells of the respiratory tract and the alveolar type II cells. It is important to note that Avian influenza viruses preferentially bind to sialic acids via alpha 2,3 linkages present on alveolar type II cells and ocular epithelium [9] while human influenza viruses bind to 2,6 linked sialic acids present in the upper respiratory tract epithelium. Due to localisation of the human influenza virus mainly to the upper respiratory tract, there is greater risk of transmission of human influenza viruses than Avian flu [10]. However strains that are able to infect the lower respiratory tract cause more inflammation and severe complications. For example, H1N1 pandemic flu of 1918 was both highly transmissible as well as virulent, while the 2009 H1N1 was highly transmissible but had moderate virulence. As opposed, H5N1 and H7N9 have low transmissibility but high virulence [11].
The HA attachment results in endocytosis of the virion and fusion with the endoplasmic membrane which in turn activates the matrix protein 2 (M2) ion channel. M2 facilitates the virion entry into the host nucleus for replication. The Influenza A viruses carry 8 negative sense RNA segments. These negative sense RNAs therefore require RNA polymerase carried with the virus into the cell to be copied onto the template. Viral replication includes the stages of assembly, budding and scission. NA helps to release these newly formed virions which are either expelled through the tract by droplet secretions or infect other cells. In this manner the virus spreads from cell to cell. The immune response of the host’s body to clear the virus can result in cell apoptosis. In severe cases necrosis of the alveolar epithelium can occur which clinically leads to acute respiratory distress. Either or both the impaired viral clearance and exaggerated host immune response can account for the severity of the illness [12].
Antigenic Shift and Drift
The HA glycoprotein is constantly evolving and the minor antigenic changes occur through accumulation of spontaneous point mutations to form antigenically distinguishable strains with very limited cross-sectional genetic diversity. This phenomenon is referred to as antigenic drift and occurs in Influenza A, B and C viruses. When a sudden genetic reassortment takes place with a creation and introduction of a novel HA, NA or both segments from a zoonotoic reservoir into the currently circulating human viruses, it is called as an antigenic shift. This phenomenon occurs only in Influenza A. For example antigenic shift took place with swine H1N1 in 1918 then later to H2N2 in 1957 and to H3N2 in 1968 with re-introduction and co-circulation of H1N1 since 1977 [13]. The consequences are dramatic as it affects an immunologicaly naive population and results in an unpredictable pandemic.
Clinical Manifestations
Over 50% of infections can be asymptomatic. Symptoms include acute onset of high fever, coryza, cough, headache, prostration, malaise which persist for 7 to 10 d. Fatigue associated with this illness take weeks to resolve.
In general, influenza illness is self limiting. High risk factors for developing complications are elderly, children, pregnant women and those with chronic conditions like asthma, hematological disorders, neurological disorders, metabolic disorders, congenital heart disorders and being immunocompromised [14].
Primary viral pneumonia, acute respiratory distress syndrome (ARDS) and pulmonary edema occur due to bronchiolar and alveolar cytopathology and cytokine storm. Secondary bacterial pneumonia post-influenza usually occurs during resolution, however 32% of patients with viral pneumonia can develop a concomitant bacterial pneumonia which is clinically challenging to distinguish [15]. Streptococcus pneumoniae is the commonest causative organism of secondary bacterial pneumonia in children. The viral-bacterial synergism is not clearly understood but the damaged respiratory epithelial lining with facilitation of access to the receptors, viral suppression of the neutrophil functions and even direct interaction between the virus on the surface of gram positive bacteria like Streptococcus pneumoniae and Staphylococcus aureus have been proposed as possible mechanisms [16].
It has been estimated that 3–5% of children suffer from influenza associated acute otitis media annually. Co-infection with bacteria increases the severity of the infection. Influenza associated myositis presents with severe bilateral myaligia in the lower limbs and reluctance to walk for about 2–3 d. The most common muscles affected are the gastrocnemius and soleus. In these children the creatinine phosphokinase can be high, myoglobinuria can also occur. Rarely rhabdomyolysis has been reported [17]. The commonest neurological complication is febrile seizures, reported in about 5% infants and young children. Influenza associated encephalopathy can occur due to direct infection of the central nervous system through viremia and presents as sudden onset of fever with convulsions and rapid progression into coma and can cause severe neurological deficits in survivors. Imaging may reveal bilateral thalamic necrosis and brainstem involvement. Fulminant myocarditis is a rare complication presenting with arrhythmias and cardiogenic shock. Hematological picture can vary from mild to severe leucopenia, thrombocytopenia including a serious complication of hemophagocytic lymphohistocytosis (HLH).
Death due to influenza occurs either due to the primary virulent infection or secondary bacterial infection or an increase in physiological load in a person with an underlying chronic condition.
Diagnosis
The availability of the nucleic acid amplification test (NAAT) test like the Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) has revolutionised the diagnosis of influenza. These are highly sensitive and specific and are regarded as the gold standard assays. Loop-Mediated Isothermal Amplification-Based Assay (LAMP) based approach have also demonstrated a very high sensitivity of 98% and specificity of 100% when compared to RT-PCR assays [18].
The major limitation of the NAAT bases tests is the high cost in resource-limited settings. The cheaper, Rapid Influenza Diagnostic tests (RIDT) using monoclonal antibodies are available for point of care. But the results are dependent on the prevalence of influenza with greater positive predictive value and false positives occurring in peak periods of influenza circulation.
Virus can be traditionally isolated by inoculation of specimen into permissive cell lines or embryonated eggs or it can be isolated with a quicker turn-around time of 1.4 d by propagation of viruses in mammalian cells grown in shell vials (Shell Vial Culture).
Direct Fluorescent antibody tests have a reasonable good sensitivity of 60–80% in comparison with viral cultures for seasonal flu, its sensitivity varied between 38 and 93% during the pandemic H1N1 when compared to the PCR results. These can differentiate Influenza A and B viruses but cannot be used for subtyping.
Serological tests help to determine presence of influenza specific antibodies following infection and vaccination. Most commonly used are Hemagluttination Inhibition Assay (HIA) and Virus Neutralisation Assay (VN) and Enzyme Immunoassays (EIA), but these tests have lower sensitivities compared to NAAT tests.
Treatment
The neuroaminase inhibitors like Oseltamivir and Zanamivir are the mainstay for treatment and help to inhibit the neuraminase which plays a critical role for cleavage of virion from infected cells and its spread to other host cells. Presently Oseltamivir is recommended as the first-line for treatment of influenza in children. It is less efficacious in treatment of Influenza B than A. The indications for initiating treatment are in those children with suspected influenza with high risk of complications or with severe illness requiring hospitalization. The dosage recommended are based on weight: 30 mg twice a day in less than 15 kg, 45 mg twice a day between 15 and 23 kg, 60 mg twice a day between 24 and 40 kg and 75 mg twice a day above 40 kg and in adults. In infants the recommended dose is 3 mg/kg twice a day while once a day dose is recommended in neonates less than 2 wk. The drug is best administered with food to reduce its gastrointestinal side-effects, commonest being vomiting. Dose modification is required in those with renal insufficiency. The treatment duration is five days, but longer duration can be given based on the clinical response. Such patients requiring longer duration of drug should be evaluated for antiviral resistance. Resistance should also be suspected in contacts of patients with Oseltamivir resistance who develop infection.
The alternative antiviral, Zanamivir given by inhalation is approved for age more than 7 y and should not be given in those with underlying airway disease. The older antivirals, Amantidine and Rimatidine are no longer recommended since 2006 due to the increased global resistance causing treatment failures.
There are several challenges in the pharmaceutical treatment of influenza. Varying efficacy of the antiviral drugs, increasing resistance of antivirals available and unavailability of parenteral formulations for use in severe patients are some of these challenges. Influenza A (H1N1) viruses resistant to Oseltamivir have been reported in children with higher frequency than adults due to relatively higher viral load [19]. The Global Influenza Surveillance and Response System (GISRS) tested 27,000 H1N1 viruses from 2009 to 2011 and detected Oseltamivir resistance in 447 viruses [20]. Of these majority were reported from the Western Pacific region of the WHO. Fourteen percent were from patients who had not been exposed to Oseltamivir, indicating either a spontaneous neuraminase His275Tyr mutation or nosocomial acquisition of the resistant strain. Immunocompromised patients, especially those with hematological malignancies or hematopoietic stem cell transplantation (HSCT) were noted to have the highest risk of Oseltamivir resistance because of suboptimal dosing during prophylaxis or disruption in treatment during viral replication [20].
Baloxavir Marboxil was approved by FDA in October 2018 for use in patients with uncomplicated influenza aged above 12 y and weighing above 40 kg, who present within 48 h of onset of illness [21]. This oral antiviral inhibits viral replication by targeting the endonuclease function encoded by the polymerase acidic subunit of the viral polymerase. It has shown broad antiviral activity against all known types of Influenza (A, B, C and D) and the Influenza A viruses of avian and swine origin which have the potential to cause pandemic [22].
Two intravenous NIs, Peramivir and Zanamivir, are currently undergoing clinical trials [19]. Laninamivir Octanoate has been evaluated in randomised control trials and have shown good tolerance and efficacy in children aged 9 y and under with Oseltamivir-resistant influenza A (H1N1) virus infection [23]. Combination therapy is also being evaluated for treatment of severe influenza strains which have likely susceptibility to M2 inhibitors [19].
Prevention and Infection Control
Annual vaccination against influenza remains the primary mode of prevention against influenza. The vaccines are effective only if there is antigenic matching of the vaccine strains with the circulating virus strains. After the matching is performed by the GISRS, the majority of commercially available vaccines are manufactured by propagation in embryonated chicken eggs. It takes at least 6–8 mo for production of these vaccines. Moreover, the flu seasons are different in both hemispheres i.e., October–May in the northern hemisphere and May–October in the southern hemisphere. Therefore the matching process is completed twice a year, once each for the two hemispheres. Indian surveillance data suggest that, based on the geographical diversity of transmission dynamics, cities above 30° latitude (Srinagar) and those with late monsoon season (Chennai and Vellore) should have winter vaccination strategies, while rest of the country in May–June [6].
At present, trivalent and quadrivalent formulations are available, which include an H1N1 strain, an H3N2 strain and one or two Influenza B strains belonging to evolutionarily diverging lineages – B/Victoria or B/Yamagata. The estimated protection of these vaccines is roughly about 60% in adults and 83% in children [24]. Efforts are underway for production of the “universal” influenza vaccine which are not dependent on this annual labor intensive matching but target certain protein regions common to the seasonal and pre-pandemic strains. The two leading candidates for universal vaccines include the highly conserved stem region of the HA and the M2 protein [25].
Routine chemoprophylaxis to contacts should be avoided, except to control an institutional outbreak, because of concerns of sub therapeutic dosing of the antiviral in case the infection is established. Chemoprophylaxis is best offered to children above 3 mo or adults who have high risk of complications from influenza or in those in whom influenza vaccination is either contraindicated, not available or may not be effective. Post-exposure chemoprophylaxis is not recommended after 48 h of first exposure to a person with influenza. Either oral Oseltamivir (for above 3 mo of age) or inhaled Zanamivir (for above 7 y of age) is recommended for chemoprophylaxis when indicated. Early initiation of empiric treatment for contacts on development of fever or respiratory symptoms is an alternative to post-exposure chemoprophylaxis [26].
Annual influenza vaccination of health care workers is critical for protection and infection control. Cough and sneezing etiquettes and hand hygiene are important for control of influenza infection both in the community and the health care settings. Health education through local media or posters displayed at health care settings can help as reminders. Since influenza primarily spreads by large particle droplet transmission, about 6 ft space surrounding the patient is considered infectious. All the body fluids of the patient are also considered infectious as indirect transmission through contact can also occur. In health care settings, patients can be cohorted or should stay in individual rooms. Droplet precautions and hand hygiene should be performed by attending health care workers and attendants. Gloves and gowns are required for personal protection if there is potential exposure to infected body fluids. Aerosol producing procedures should be minimized, ideally conducted in negative pressure rooms and protection with N95 mask is needed by the health care personnel conducting such procedures.
Surveillance and Pandemic Preparedness
The GISRS of WHO has a robust network of laboratories spread across the globe which is to be notified of any case of human infection due to a new Influenza A virus subtype within 24 h of its detection. These laboratories constantly survey the genetic and antigenic characteristics of circulating influenza viruses. However, there is more work needed to be done for capacity building for diagnostics in the middle- and low-income countries which bear the maximum brunt of the pandemic. Also the production time for vaccine needs to be reduced along with better availability of efficacious and safe antiviral drugs during such a pandemic [2]. In the efforts to address many of these challenges, in 2019, WHO established the Global Influenza Strategy 2019–2030. The four strategic objectives outlined are to promote research, strengthen global surveillance, expand seasonal influenza prevention to protect the vulnerable and strengthen pandemic preparedness [27].
Summary
Influenza, like a chameleon is an ever changing virus and continues to keep humans on their toes. Over the hundred years since the 1918 flu epidemic, there have been considerable advances in the understanding of pathogenesis, diagnostics, antiviral drugs and vaccination strategies. But despite the progress, the global preparedness for the next pandemic remains suboptimal. Not forgetting that the greatest impact of the pandemic was suffered by India, the Indian health authorities need to gear up with greater urgency in capacity building for clinical support and preventive measures.
References
Chandra S, Eva K-NE. The evolution of pandemic influenza: evidence from India, 1918–19. BMC Infect Dis. 2014;14:510.
Jester B, Uyeki T, Jernigan D. Readiness for responding to a severe pandemic 100 years after 1918. Am J Epidemiol. 2018;187:2596–602.
Lowen AC, Steel J. Roles of humidity and temperature in shaping influenza seasonality. J Virol. 2014;88:7692–5.
Ng S, Gordon A. Influenza burden and transmission in the tropics. Curr Epidemiol Rep. 2015;2:89–100.
National Centre for Disease Control. Seasonal Influenza H1N1. Available at: https://ncdc.gov.in/index4.php?lang=1&level=0&linkid=119&lid=276. Accessed 2nd August 2019.
Chadha MS, Potdar VA, Saha S, et al. Dynamics of influenza seasonality at sub-regional levels in India and implications for vaccination timing. PLoS One. 2015;10:e0124122.
Bridges CB, Kuehnert MJ, Hall CB. Transmission of influenza: implications for control in health care settings. Clin Infect Dis. 2003;37:1094–101.
A revision of the system of nomenclature for influenza viruses: a WHO memorandum. Bull World Health Organ. 1980;58:585–91.
Kumlin U, Olofsson S, Dimock K, Arnberg N. Sialic acid tissue distribution and influenza virus tropism. Influenza Other Respir Viruses. 2008;2:147–54.
Mansfield KG. Viral tropism and the pathogenesis of influenza in the Mammalian host. Am J Pathol. 2007;171:1089–92.
Camp JV, Bagci U, Chu YK, et al. Lower respiratory tract infection of the ferret by 2009 H1N1 pandemic influenza A virus triggers biphasic, systemic, and local recruitment of neutrophils. J Virol. 2015;89:8733–48.
Herold S, Becker C, Ridge KM, Budinger GR. Influenza virus-induced lung injury: pathogenesis and implications for treatment. Eur Respir J. 2015;45:1463–78.
Wikramaratna PS, Sandeman M, Recker M, Gupta S. The antigenic evolution of influenza: drift or thrift? Philos Trans R Soc Lond Ser B Biol Sci. 2013;368:20120200.
Yewale V, Dharmapalan D. Complications and prognosis of influenza. In: Rao I, editor. Influenza: Complete Spectrum - II - ECAB. New Delhi: Elsevier Health Sciences; 2011. p. 1–20.
Vander Sluijs KF, van der Poll T, Lutter R, Juffermans NP, Schultz MJ. Bench-to-bedside review: bacterial pneumonia with influenza - pathogenesis and clinical implications. Crit Care. 2010;14:219.
Rowe HM, Meliopoulos VA, Iverson A, Bomme P, Schultz-Cherry S, Rosch JW. Direct interactions with influenza promote bacterial adherence during respiratory infections. Nat Microbiol. 2019;4:1328–36.
Magee H, Goldman RD. Viral myositis in children. Can Fam Physician. 2017;63:365–8.
Vemula SV, Zhao J, Liu J, Wang X, Biswas S, Hewlett I. Current approaches for diagnosis of influenza virus infections in humans. Viruses. 2016;8:96.
Hayden F. Developing new antiviral agents for influenza treatment: what does the future hold? Clin Infect Dis. 2009;48:S3–13.
Hurt AC, Chotpitayasunondh T, Cox NJ, et al. Antiviral resistance during the 2009 influenza A H1N1 pandemic: public health, laboratory, and clinical perspectives. Lancet Infect Dis. 2012;12:240–8.
CDC. Influenza Antiviral Medications: Summary for Clinicians. Available at: https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed 26th Dec 2019.
Mishin VP, Patel MC, Chesnokov A, et al. Susceptibility of influenza a, B, C, and D viruses to baloxavir. Emerg Infect Dis. 2019;25:1969–72.
Sugaya N, Ohashi Y. Long-acting neuraminidase inhibitor laninamivir octanoate (CS-8958) versus oseltamivir as treatment for children with influenza virus infection. Antimicrob Agents Chemother. 2010;54:2575–82.
Houser K, Subbarao K. Influenza vaccines: challenges and solutions. Cell Host Microbe. 2015;17:295–300.
Sautto GA, Kirchenbaum GA, Ross TM. Towards a universal influenza vaccine: different approaches for one goal. Virol J. 2018;15:17.
Uyeki TM, Bernstein HH, Bradley JS, et al. Clinical practice guidelines by the Infectious Diseases Society of America: 2018 update on diagnosis, treatment, chemoprophylaxis, and institutional outbreak management of seasonal influenza. Clin Infect Dis. 2019;68:e1–47.
WHO. Global Influenza Strategy 2019-2030. Geneva: World Health Organization; 2019. Available at: https://apps.who.int/iris/bitstream/handle/10665/311184/9789241515320-eng.pdf?ua=1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dharmapalan, D. Influenza. Indian J Pediatr 87, 828–832 (2020). https://doi.org/10.1007/s12098-020-03214-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-020-03214-1