Abstract
Objective
To assess serum sclerostin levels in relation to severity of arthropathy and bone mineral density (BMD) in children with hemophilic arthropathy.
Methods
This cross-sectional study included 40 male children suffering from Hemophilia A, and 10 matched healthy controls. Assessment of factor VIII deficiency degree, frequency of bleeding, type of treatment, body mass index (BMI), disease severity using the Hemophilia Joint Health Score (HJHS) and lumbar spine (LS) Z score for bone mineral density (BMD) using dual-energy X-ray absorbiometry was done. Serum sclerostin levels were measured for all patients and controls.
Results
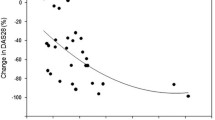
Significant difference of serum sclerostin levels between the patient and control groups with Mean ± SD (0.09 ± 0.07 ng/ml) and (0.04 ± 0.01 ng/ml) (P value = 0.028) respectively was found. Significant positive correlations between serum sclerostin levels and the patients’ age, and HJHS (P value <0.05) were found, while it had negative correlation with DEXA Z score, not reaching a significant value. LS-BMD-Z score levels ranged from (−4.5 to 1.2), with 15 patients with low BMD Z score (less than −2) representing 37.5% of total patients.
Conclusions
Serum sclerostin levels are elevated in hemophilic children denoting bone metabolism affection and correlates with increased age, and HJHS. Increased levels of serum sclerostin may identify hemophilic patients at high risk for developing osteoporosis.

Similar content being viewed by others
References
Hoyer LW. Hemophilia A. New Engl J Med. 1994;330:38–47.
Gallacher SJ, Deighan C, Wallace AM, et al. Association of severe haemophilia A with osteoporosis: a densitometric and biochemical study. Q J Med. 1994;87:181–6.
Wallny TA, Scholz DT, Oldenburg J, et al. Osteoporosis in haemophilia–an underestimated co-morbidity? Haemophilia. 2007;13:79–84.
Nair AP, Jijina F, Ghosh K, Madkaikar M, Shrikhande M, Nema M. Osteoporosis in young hemophiliacs from western India. Am J Hematol. 2007;82:453–7.
Baron R, Rawadi G. Targeting the Wnt/β-catenin pathway to regulate bone formation in the adult skeleton. Endocrinology. 2007;148:2635–43.
Krishnan V, Bryant HU, MacDougald OA. Regulation of bone mass by Wnt signaling. J Clin Invest. 2006;116:1202–9.
Van Bezooijen RL, Roelen BA, Visser A, et al. Sclerostin is an osteocyte-expressed negative regulator of bone formation, but not a classical BMP antagonist. J Exp Med. 2004;199:805–14.
Semënov M, Tamai K, He X. SOST is a ligand for LRP5/LRP6 and a Wnt signaling inhibitor. J Biol Chem. 2005;280:26770–5.
Poole KE, van Bezooijen RL, Loveridge N. Sclerostin is a delayed secreted product of osteocytes that inhibits bone formation. FASEB J. 2005;19:1842–4.
van Bezooijen RL, ten Dijke P, Papapoulos SE, Löwik CW. SOST/sclerostin, an osteocyte-derived negative regulator of bone formation. Cytokine Growth Factor Rev. 2005;16:319–27.
Wijenayaka AR, Kogawa M, Lim HP, Bonewald LF, Findlay DM, Atkins GJ. Sclerostin stimulates osteocyte support of osteoclast activity by a RANKL-dependent pathway. PLoS One. 2011;6:e25900.
Baron R, Kneissel M. WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med. 2013;19:179–92.
Brunkow ME, Gardner JC, Van Ness J, et al. Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot–containing protein. Am J Hum Genet. 2001;68:577–89.
Staehling-Hampton K, Proll S, Paeper BW, et al. A 52-kb deletion in the SOST-MEOX1 intergenic region on 17q12-q21 is associated with van Buchem disease in the Dutch population. Am J Med Genet. 2002;110:144–52.
Li X, Ominsky MS, Warmington KS, et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J Bone Miner Res. 2009;24:578–88.
Padhi D, Jang G, Stouch B, Fang L, Posvar E. Single-dose, placebo-controlled, randomized study of AMG 785, a sclerostin monoclonal antibody. J Bone Miner Res. 2011;26:19–26.
El-Bakry S, Saber N, Zidan H, Samaha D. Sclerostin as an innovative insight towards understanding rheumatoid arthritis. Egypt Rheumatol. 2016;38:71–5.
Visnjic D, Kalajzic Z, Rowe DW. Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood. 2004;103:3258–64.
Paschou SA, Anagnostis P, Karras S, et al. Bone mineral density in men and children with haemophilia a and B: A systematic review and meta-analysis. Osteoporos Int. 2014;25:2399–407.
Lewiecki EM, Gordon CM, Baim S, et al. International Society for Clinical Densitometry 2007 adult and pediatric official positions. Bone. 2008;43:1115–21.
Bishop N, Braillon P, Burnham J, et al. Dual-energy X-ray absorptiometry assessment in children and adolescents with diseases that may affect the skeleton: the 2007 ISCD pediatric official positions. J Clin Densitom. 2008;11:29–42.
Bachrach LK, Sills IN. Bone densitometry in children and adolescents. Pediatrics. 2011;127:189–94.
Hilliard P, Funk S, Zourikian N, et al. Hemophilia joint health score reliability study. Hemophilia. 2006;12:518–25.
Fewtrell MS. Bone densitometry in children assessed by dual x ray absorptiometry: uses and pitfalls. Arch Dis Child. 2003;88:795–8.
Giordano P, Brunetti G, Lassandro G, et al. High serum sclerostin levels in children with haemophilia A. Br J Haematol. 2016;172:293–5.
Clarke BL, Drake MT. Clinical utility of serum sclerostin measurements. Bonekey Rep. 2013;2:361.
Hassab HMA, El-Gendy WM, El-Noueam KI, Abd El Ghany HM, Elwan MMA. Serum cartilage oligomeric matrix protein reflects radiological damage and functional status in hemophilic arthropathy patients. Egypt Rheumatol. 2016;38:241–5.
Barnes C, Wong P, Egan B, et al. Reduced bone density among children with severe hemophilia. Pediatrics. 2004;114:e177–81.
Tlacuilo-Parra A, Morales-Zambrano R, Tostado-Rabago N, Esparza-Flores MA, Lopez-Guido B, Orozco-Alcala J. Inactivity is a risk factor for low bone mineral density among haemophilic children. Br J Haematol. 2008;140:562–7.
EL Naeem RSA, El Hefnawy HEL, El Aziz OAA, El Mikkawy DMEED, Tantawy AAG, El-Ghany SMA. Assessment of bone mineral density and functional status in children with hemophilic arthropathy. Glob Adv Res J Med Med Sci. 2016;5:035–41.
Author information
Authors and Affiliations
Contributions
DME El-M: Collection of patients from clinic, clinical assessment and HJHS assessment, prepare patients data for statistical analysis; MAE: Helped in clinical data assessment and writing the manuscript; SMA El-G: Collection of blood samples; DS: Laboratory analysis of serum sclerostin levels. DME El- M will act as guarantor for this paper.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Source of Funding
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Mikkawy, D.M.E., Elbadawy, M.A., Abd El-Ghany, S.M. et al. Serum Sclerostin Level and Bone Mineral Density in Pediatric Hemophilic Arthropathy. Indian J Pediatr 86, 515–519 (2019). https://doi.org/10.1007/s12098-019-02855-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-019-02855-1