Abstract
Background
Solitary pulmonary nodules (SPNs) frequently bother oncologists. The differentiation of malignant from benign nodules with non-invasive approach remains a tough challenge. This study was designed to assess the diagnostic accuracy of dynamic computed tomography (CT), dynamic magnetic resonance imaging (MRI), fluorine 18 fluorodeoxyglucose (18F-FDG) positron emission tomography (PET), and technetium 99 m (99mTc) depreotide single photon emission computed tomography (SPECT) for SPNs.
Methods
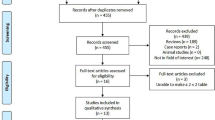
Electronic databases of MEDLINE, PubMed, EMBASE, and Cochrane Library were searched to identify relevant trials. The primary evaluation index of diagnostic accuracy was areas under the summary receiver-operating characteristic (SROC) curve. The results were analyzed utilizing Stata 12.0 statistical software.
Results
Seventy-three trials incorporating 7956 individuals were recruited. Sensitivities, specificities, positive likelihood ratios, negative likelihood ratios, diagnostic score, diagnostic odds ratios, and areas under the SROC curve with 95% confidence intervals were, respectively, 0.92 (0.89–0.95), 0.64 (0.54–0.74), 2.60 (1.98–3.42), 0.12 (0.08–0.17), 3.10 (2.62–3.59), 22.24 (13.67–36.17), and 0.91 (0.88–0.93) for CT; 0.92 (0.86–0.95), 0.85 (0.77–0.90), 6.01 (3.90–9.24), 0.10 (0.06–0.17), 4.12 (3.41–4.82), 61.39 (30.41–123.93), and 0.94 (0.92–0.96) for MRI; 0.90 (0.86–0.93), 0.73 (0.65–0.79), 3.28 (2.56–4.20), 0.14 (0.10–0.19), 3.16 (2.69–3.64), 23.68 (14.74–38.05), and 0.90 (0.87–0.92) for 18F-FDG PET; and 0.93 (0.88–0.96), 0.70 (0.56–0.81), 3.12 (2.03–4.81), 0.10 (0.06–0.17), 3.43 (2.63–4.22), 30.74 (13.84–68.27), and 0.93 (0.91–0.95) for 99mTc-depreotide SPECT.
Conclusion
The dynamic MRI, dynamic CT, 18F-FDG PET, and 99mTc-depreotide SPECT were favorable non-invasive approaches to distinguish malignant SPNs from benign. Moreover, from the viewpoint of cost-effectiveness and avoiding radiation, the dynamic MRI was recommendable for SPNs.





Similar content being viewed by others
References
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology-Lung Cancer Screening (2020 Version 1). https://guide.medlive.cn/; 2020 Accessed 20 March 2020.
The Lung Cancer Group of Chinese Medical Association Respiratory Neurology, Chinese expert alliance of prevention and control for lung cancer. Chinese expert consensus on the diagnosis and treatment of pulmonary nodules (2018 edition). Chin Tubercul Respir J. 2018;41(10):763–771.
Cronin P, Dwamena BA, Kelly AM, Carlos RC. Solitary pulmonary nodules: meta-analytic comparison of cross-sectional imaging modalities for diagnosis of malignancy. Radiology. 2008;246(3):772–82.
Deeks JJ, Macaskill P, Irwig L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol. 2005;58(9):882–93.
Mack MJ, Hazelrigg SR, Landreneau RJ, Acuff TE. Thoracoscopy for the diagnosis of the indeterminate solitary pulmonary nodule. Ann Thorac Surg. 1993;56(4):825–30.
Lempel JK, Raymond DP, Usman A, Susan OM, Bolen MA, Ruffin G, et al. Video-assisted thoracic surgery resection without intraoperative fluoroscopy after CT-guided microcoil localization of peripheral pulmonary nodules. J Vasc Interv Radiol. 2018;29(10):1423–8.
Keagy BA, Starek PJ, Murray GF, Battaglini JW, Lores ME, Wilcox BR. Major pulmonary resection for suspected but unconfirmed malignancy. Ann Thorac Surg. 1984;38(4):314–6.
Nasim F, Ost DE. Management of the solitary pulmonary nodule. Curr Opin Pulm Med. 2019;25(4):344–53.
Ost D, Fein AM, Feinsilver SH. Clinical practice. The solitary pulmonary nodule. N Engl J Med. 2003;348(25):2535–42.
Gambhir SS, Shepherd JE, Shah BD, Hart E, Hoh CK, Valk PE, et al. Analytical decision model for the cost-effective management of solitary pulmonary nodules. J Clin Oncol. 1998;16(6):2113–255.
Dewes P, Frellesen C, Al-Butmeh F, Albrecht MH, Scholtz JE, Metzger SC, et al. Comparative evaluation of non-contrast CAIPIRINHA-VIBE 3T-MRI and multidetector CT for detection of pulmonary nodules: in vivo evaluation of diagnostic accuracy and image quality. Eur J Radiol. 2016;85(1):193–8.
Usuda K, Sagawa M, Motono N, Ueno M, Tanaka M, Machida Y, et al. Diagnostic performance of diffusion weighted imaging of malignant and benign pulmonary nodules and masses: comparison with positron emission tomography. Asian Pac J Cancer Prev. 2014;15(11):4629–35.
Mori T, Nomori H, Ikeda K, Kawanaka K, Shiraishi S, Katahira K, et al. Diffusion-weighted magnetic resonance imaging for diagnosing malignant pulmonary nodules/masses: comparison with positron emission tomography. J Thorac Oncol. 2008;3(4):358–64.
Ohba Y, Nomori H, Mori T, Shiraishi K, Namimoto T, Katahira K. Diffusion-weighted magnetic resonance for pulmonary nodules: 1.5 vs. 3 Tesla. Asian Cardiovasc Thorac Ann. 2011;19(2):108–14.
Tozaki M, Ichiba N, Fukuda K. Dynamic magnetic resonance imaging of solitary pulmonary nodules: utility of kinetic patterns in differential diagnosis. J Comput Assist Tomogr. 2005;29(1):13–9.
Ohno Y, Fujisawa Y, Yui M, Takenaka D, Koyama H, Sugihara N, et al. Solitary pulmonary nodule: comparison of quantitative capability for differentiation and management among dynamic CE-perfusion MRI at 3 T system, dynamic CE-perfusion ADCT and FDG-PET/CT. Eur J Radiol. 2019;115:22–30.
Ohno Y, Nishio M, Koyama H, Seki S, Tsubakimoto M, Fujisawa Y, et al. Solitary pulmonary nodules: comparison of dynamic first-pass contrast-enhanced perfusion area-detector CT, dynamic first-pass contrast-enhanced MR imaging, and FDG PET/CT. Radiology. 2015;274(2):563–75.
Ohno Y, Koyama H, Takenaka D, Nogami M, Maniwa Y, Nishimura Y, et al. Dynamic MRI, dynamic multidetector-row computed tomography (MDCT), and coregistered 2-[fluorine-18]-fluoro-2-deoxy-D-glucose-positron emission tomography (FDG-PET)/CT: comparative study of capability for management of pulmonary nodules. J Magn Reson Imaging. 2008;27(6):1284–95.
Schaefer JF, Vollmar J, Schick F, Vonthein R, Seemann MD, Aebert H, et al. Solitary pulmonary nodules: dynamic contrast-enhanced MR imaging–perfusion differences in malignant and benign lesions. Radiology. 2004;232(2):544–53.
Ohno Y, Koyama H, Fujisawa Y, Yoshikawa T, Seki S, Sugihara N, et al. Dynamic contrast-enhanced perfusion area detector CT for non-small cell lung cancer patients: influence of mathematical models on early prediction capabilities for treatment response and recurrence after chemoradiotherapy. Eur J Radiol. 2016;85(1):176–86.
Ohno Y, Fujisawa Y, Koyama H, Kishida Y, Seki S, Sugihara N, et al. Dynamic contrast-enhanced perfusion area-detector CT assessed with various mathematical models: its capability for therapeutic outcome prediction for non-small cell lung cancer patients with chemoradiotherapy as compared with that of FDG-PET/CT. Eur J Radiol. 2017;86:83–91.
Ohno Y, Koyama H, Fujisawa Y, Yoshikawa T, Inokawa H, Sugihara N, et al. Hybrid Type iterative reconstruction method vs. filter back projection method: capability for radiation dose reduction and perfusion assessment on dynamic first-pass contrast-enhanced perfusion chest area-detector CT. Eur J Radiol. 2016;85(1):164–75.
Gibson G, Kumar AR, Steinke K, Bashirzadeh F, Roach R, Windsor M, et al. Risk stratification in the investigation of pulmonary nodules in a high-risk cohort: positron emission tomography/computed tomography outperforms clinical risk prediction algorithms. Intern Med J. 2017;47(12):1385–92.
Predina JD, Newton AD, Keating J, Barbosa EM Jr, Okusanya O, Xia L, et al. Intraoperative molecular imaging combined with positron emission tomography improves surgical management of peripheral malignant pulmonary nodules. Ann Surg. 2017;266(3):479–88.
Lillington GA, Gould MK. Managing solitary pulmonary nodules: accurate predictions and divergent conclusions. Mayo Clin Proc. 1999;74(4):435–6.
Nomori H, Watanabe K, Ohtsuka T, Naruke T, Suemasu K, Uno K. Visual and semiquantitative analyses for F-18 fluorodeoxyglucose PET scanning in pulmonary nodules 1 cm to 3 cm in size. Ann Thorac Surg. 2005;79(3):984–8.
Raslan OA, Parkar ND, Muzaffar R, Doherty C, Osman MM. Case 227: endobronchial carcinoid tumor with incidental metastatic breast cancer detected with somatostatin receptor scintigraphy ((111) in pentreotide). Radiology. 2016;278(3):949–55.
Krenning EP, Kwekkeboom DJ, Bakker WH, Breeman WA, Kooij PP, Oei HY, et al. Somatostatin receptor scintigraphy with [111In-DTPA-d-Phe1]-and [123I-Tyr3]-octreotide: the Rotterdam experience with more than 1000 patients. Eur J Nucl Med. 1993;20(8):716–31.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, Q., Zhong, L. & Xie, X. The value of four imaging modalities to distinguish malignant from benign solitary pulmonary nodules: a study based on 73 cohorts incorporating 7956 individuals. Clin Transl Oncol 23, 296–310 (2021). https://doi.org/10.1007/s12094-020-02418-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02418-3