Abstract
Purpose
The present study aimed to investigate the efficacy and severity of adverse effects of HCAG and CAG re-induction chemotherapy in elderly low- and intermediate-risk group patients diagnosed with acute myeloid leukemia (AML) following induction failure.
Methods
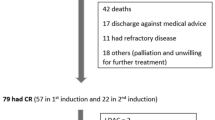
A total of 94 AML patients were enrolled in the study, of whom 46 were treated with HCAG chemotherapy, while 48 were treated with CAG chemotherapy.
Result
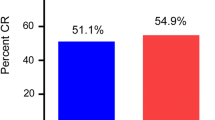
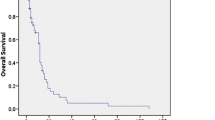
The complete remission (CR) was 39.6% in the patients with HCAG, while the CR was 33.3% in the CAG group. The overall remission (ORR) was 63.0% and 43.5% in patients of the HCAG and CAG groups, respectively (P = 0.038). The median survival time of progression free survival (PFS) was 8.0 (95% CI 3.843–10.157) months in the HCAG group and 7.0 (95% CI 2.682–13.318) months in the CAG group (P = 0.032). A total of 31 patients in the HCAG group suffered from grade 4 hematological toxicity, whereas 29 patients were treated with CAG (P = 0.622). A total of 27 (58.7%) cases indicated apparent pulmonary infection in the HCAG group, while 25 (52.1%) were noted with this complication in the CAG group (P = 0.519). Oral cavity toxicity was evident for 13 (28.3%) and 11 (23.0%) cases in the HCAG and CAG groups, respectively (P = 0.216).
Conclusion
The HCAG regimen was more effective than the CAG regimen in elderly low- and intermediate-risk group patients diagnosed with acute myeloid leukemia although the HCAG regimen exhibited similar toxicity with that of the CAG group.




Similar content being viewed by others
References
Burnett AK, Russell N, Hills RK, et al. A randomised comparison of the novel nucleoside analogue sapacitabine with low-dose cytara-bine in older patients with acute myeloid leukaemia. Leukemia. 2015;29(6):1312–9.
Chishti AH. Malaria selectively targets pregnancy receptors. Blood. 2015;125(2):217–8.
Latagliata R, Petti MC, Mandelli F. Acute myeloid leukemia in the elderly: ‘Per aspera adastra’? Leuk Res. 1999;23:603–13.
Kantarjian H, Ravandi F, O′Brien S, et al. Intensive chemotherapy does not benefit most older patients (age 70 years or older) with acute myeloid leukemia. Blood. 2010;116(22):4422–9.
Oran B, Weisdorf DJ. Survival for older patients with acute myeloid leukemia: a population-based study. Haematologica. 2012;97(12):1916–24.
Juliusson G, Antunovic P, Derolf A, Lehmann S, Möllgård L, Stockelberg D, Tidefelt U, Wahlin A, Höglund M. Age and acute myeloid leukemia: real world data on decision to treat and outcomes from Swedish Acute Leukemia Registry. Blood. 2009;113(18):4179–87.
Sekeres MA. Treatment of older adults with acute myeloid leukemia: state of the art and current perspectives. Haematologica. 2008;93:1769–72.
Leith CP, Kopecky KJ, Chen IM, et al. Frequency and clinical significance of the expression of the multidrug resistance proteins MDR1/P-glycoprotein, MRP1, and LRP in acute myeloid leukemia: a Southwest Oncology Group Study. Blood. 1999;94:1086–99.
Buchner T, Berdel WE, Haferlach C, et al. Agerelated risk profile and chemotherapy dose response in acute myeloid leukemia: a study by the German Acute Myeloid Leukemia Cooperative Group. J Clin Oncol. 2009;27:61–9.
Kantarjian H, O’Brien S, Cortes J, et al. Results of intensive chemotherapy in 998 patients age 65 years or older with acute myeloid leukemia or high-risk myelodysplastic syndrome: predictive prognostic models for outcome. Cancer. 2006;106:1090–8.
Kadia TM, Faderl S, Ravandi F, et al. Final results of a phase 2 trial of clofarabine and low-dose cytarabine alternating with decitabine in older patients with newly diagnosed acute myeloid leukemia. Cancer. 2015;121(14):2375–82.
Suzushima H, Wada N, Yamasaki H, et al. Low-dose cytarabine and aclarubicin in combination with grannlocyte colony-stimu1ating factor for elderly patients with previously untreated acute myeloid leukemia. Leuk Res. 2010;34(5):610–4.
Zhu HH, Jiang H, Huang XJ, et al. Cytarabine, aclarubicin and granulocyte colony—stimulating factor regimen represents an effective and safe salvage regimen for patients with acute myeloid leukemia refractory to first course of induction chemotherapy. Leuk Lymphoma. 2013;54(11):2452–7.
Zhu HH, Jiang H, et al. Homoharringtonine, aclarubicin and cytarabine (HAA) regimen as the first course of induction therapy is highly effective for acute myeloid leukemia with t(8;21). Leuk Res. 2016;44:40–4.
Fan CL, Yu W, et al. Efficacy of HAA regimen in the treatment of 64 patients with refractory/relapsed acute myeloid leukemia. Zhonghua Xue Ye Xue Za Zhi. 2016;37(2):100–4.
Zhang JY, Yu K, Li LJ, et al. Comparison of efficacy of HCAG and FLAG re-induction chemotherapy in acute myeloid leukemia patients of low-and intermediate-risk groups. Clin Transl Oncol. 2019;21:1543–50.
Zhang X, Li Y, Zhang Y, et al. Etoposide in combination with low-dose CAG(eytarabine, aclarubicin, G-CSF)for the treatment of relapsed or refractory acute myeloid leukemia”: a multicenter, randomized control trial in southwest China. Leuk Res. 2013;37(6):657–64.
Fresno M, Jimenez A, Vazquez D. Inhibition of translation in eukaryotic systems by harringtonine. Eur J Biochem. 1977;72(2):323–30.
Ye XJ, Lin MF. Homoharringtonine induces apoptosis of endothelium and down-regulates VEGF expression of K562 cells. J Zhejiang Univ Sci. 2004;5(2):230–4.
Xie WZ, Lin MF, Huang H, Cai Z. Homoharringtonine-induced apoptosis of human leukemia HL-60 cells is associated with down-regulation of telomerase. Am J Chin Med. 2006;34(2):233–44.
Cai Z, Bao HY, Lin MF. Correlation between survivin mRNA expression and homoharringtonine induced apoptosis of malignant hematopoietic cells. Chin Med J (Engl). 2005;118(7):548–54.
Gurel G, Blaha G, Moore PB, Steitz TA. U2504 determines the species specificity of the A-site cleft antibiotics: the structures of tiamulin, homoharringtonine, and bruceantin bound to the ribosome. J Mol Biol. 2009;389:146–56.
Lü S, Wang J. Homoharringtonine and omacetaxine for myeloid hematological malignancies. J Hematol Oncol. 2014;7:2.
Zhang T, Shen S, Zhu Z, Lu S, Yin X, Zheng J, et al. Homoharringtonine binds to and increases myosin—9 in myeloid leukemia. Br J Pharmacol. 2016;173:212–21.
Wang L, You LS, Ni WM, Ma QL, Tong Y, Mao LP, et al. β-Catenin and AKT are promising targets for combination therapy in acute myeloid leukemia. Leuk Res. 2013;37:1329–40.
Wang J, Lfi S, Yan J, et al. A homoharringtonine-based induction regimen for the treatment of elderly patients with acute myeloid leukemia: a single center experience from China. J Hematol Oncol. 2009;2:32.
Sugiyama T, Kohara H, Noda M, Nagasawa T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity. 2006;25(6):977–88.
Xu J, Lv TT, et al. Efficacy of common salvage chemotherapy regimens in patients with refractory or relapsed acute myeloid leukemia A retrospective cohort study. Medicine. 2018;97(39):e12102.
Rollig C, Thiede C, Gramatzki M, et al. A novel prognostic model in elderly patients with acute myeloid leukemia:results of 909 patients entered into the prospective AML96 trial. Blood. 2010;116(6):971–8. https://doi.org/10.1182/blood-2010-01-267302.
Funding
Funding was funded by Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (Grant no. 2020376298).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
The study protocol was approved by the Medical Research and Ethics Committee of the Lishui Municipal Central Hospital.
Informed consent
Written informed consent from each patient or family member was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J.Y., Li, L., Liu, W. et al. Comparison of efficacy of HCAG and CAG re-induction chemotherapy in elderly low- and intermediate-risk group patients diagnosed with acute myeloid leukemia. Clin Transl Oncol 23, 48–57 (2021). https://doi.org/10.1007/s12094-020-02383-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02383-x