Abstract
Objective
To determine the incidence of serous tubal intraepithelial carcinoma (STIC) after risk reduction salpingo-oophorectomy(RRSO), and to describe oncological outcomes after RRSO.
Materials and methods
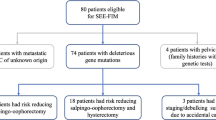
BRCA pathogenic mutation carriers who had undergone an RRSO were evaluated in this retrospective multicenter observational study. Patients were only included when fallopian tubes were analyzed following the protocol for Sectioning and Extensively Examining the FIMbria (SEE-FIM). Surgeries were performed between June 2010 and April 2017 at eight Spanish hospitals.
Results
A total of 359 patients met the inclusion criteria. STIC was diagnosed in 3 (0.8%) patients; one of them underwent surgical staging due to positive peritoneal washing, with absence of disease at the final pathology report. None of the three patients received adjuvant chemotherapy and were free of disease at last follow-up. Fallopian tube and ovarian carcinoma were diagnosed in 5 (1.4%) and 1 (0.3%), respectively. At a median (range) follow-up time of 29 (3–92) months, five patients had a newly diagnosed breast cancer. Other types of cancer, which were diagnosed during the follow-up time, included: serous primary peritoneal carcinoma (n = 1), serous endometrial carcinoma (n = 1), colon (n = 1), pancreas (n = 1), jaw (n = 1), and lymphoma (n = 1). Seven patients died due to different types of cancer: breast (n = 4), pancreas (n = 1), jaw (n = 1), and colon (n = 1).
Conclusion
The incidence of STIC after RRSO in BRCA mutation carriers is low (0.8%) and it presents an excellent oncological outcome. Patients after RRSO, however, run the risk to develop other types of cancer during follow-up and should be properly advised before the prophylactic surgery.

Similar content being viewed by others
References
Kurman RJ. Origin and molecular pathogenesis of ovarian high-grade serous carcinoma. Ann Oncol. 2013;24(Suppl 10):x16–21.
Morrison JC, Blanco LZ Jr, Vang R, Ronnett BM. Incidental serous tubal intraepithelial carcinoma and early invasive serous carcinoma in the nonprophylactic setting: analysis of a case series. Am J Surg Pathol. 2015;39(4):442–53.
Li HX, Lu ZH, Shen K, Cheng WJ, Malpica A, Zhang J, et al. Advances in serous tubal intraepithelial carcinoma: correlation with high grade serous carcinoma and ovarian carcinogenesis. Int J Clin Exp Pathol. 2014;7(3):848–57.
Patrono MG, Corzo C, Iniesta M, Ramirez PT. Management of preinvasive lesions. Clin Obstet Gynecol. 2017;60(4):771–9.
Patrono MG, Iniesta MD, Malpica A, Lu KH, Fernandez RO, Salvo G, et al. Clinical outcomes in patients with isolated serous tubal intraepithelial carcinoma (STIC): a comprehensive review. Gynecol Oncol. 2015;139(3):568–72.
Zakhour M, Danovitch Y, Lester J, Rimel BJ, Walsh CS, Li AJ, et al. Occult and subsequent cancer incidence following risk-reducing surgery in BRCA mutation carriers. Gynecol Oncol. 2016;143(2):231–5.
Medeiros F, Muto MG, Lee Y, Elvin JA, Callahan MJ, Feltmate C, et al. The tubal fimbria is a preferred site for early adenocarcinoma in women with familial ovarian cancer syndrome. Am J Surg Pathol. 2006;30(2):230–6.
National Comprehensive Cancer Network (NCCN). Genetic/familial high-risk assessment: breast and ovarian. https://www.nccn.org/store/login/login.aspx?. https://www.nccn.org/professionals/physician_gls/pdf/genetics_screening.pdf. Accessed Jan 2018.
Mehrad M, Ning G, Chen EY, Mehra KK, Crum CP. A pathologist’s road map to benign, precancerous, and malignant intraepithelial proliferations in the fallopian tube. Adv Anat Pathol. 2010;17(5):293–302.
College of American Pathologists. Protocol for the examination of specimens from patients with carcinoma of the fallopian tube. http://www.cap.org/ShowProperty?nodePath=/UCMCon/Contribution%20Folders/WebContent/pdf/fallopiantube-15protocol-3102.pdf. Accessed 10 March 2018.
Prat J. FIGO Committee on Gynecologic Oncology. Staging classification for cancer of the ovary, fallopian tube, and peritoneum. Int J Gynaecol Obstet. 2014;124(1):1–5.
Powell CB, Swisher EM, Cass I, McLennan J, Norquist B, Garcia RL, et al. Long term follow up of BRCA1 and BRCA2 mutation carriers with unsuspected neoplasia identified at risk reducing salpingo-oophorectomy. Gynecol Oncol. 2013;129(2):364–71.
King J, Wynne CH, Assersohn L, Jones A. Hormone replacement therapy and women with premature menopause—a cancer survivorship issue. Eur J Cancer. 2011;47(11):1623–32.
Domchek SM, Friebel TM, Singer CF, Evans DG, Lynch HT, Isaacs C, et al. Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA. 2010;304(9):967–75.
Eisen A, Lubinski J, Gronwald J, Moller P, Lynch HT, Klijn J, et al. Hereditary breast cancer clinical study group. Hormone therapy and the risk of breast cancer in BRCA1 mutation carriers. J Natl Cancer Inst. 2008;100(19):1361–7.
Rebbeck TR, Friebel T, Wagner T, Lynch HT, Garber JE, Daly MB, et al. Effect of short-term hormone replacement therapy on breast cancer risk reduction after bilateral prophylactic oophorectomy in BRCA1 and BRCA2 mutation carriers: the PROSE study group. J Clin Oncol. 2005;23(31):7804–10.
Chlebowski RT, Prentice RL. Menopausal hormone therapy in BRCA1 mutation carriers: uncertainty and caution. J Natl Cancer Inst. 2008;100(19):1341–3.
Thompson D, Easton DF, Breast Cancer Linkage Consortium. Cancer Incidence in BRCA1 mutation carriers. J Natl Cancer Inst. 2002;94(18):1358–65.
Brose MS, Rebbeck TR, Calzone KA, Stopfer JE, Nathanson KL, Weber BL. Cancer risk estimates for BRCA1 mutation carriers identified in a risk evaluation program. J Natl Cancer Inst. 2002;94(18):1365–72.
Blok F, Roes EM, van Leenders GJ, van Beekhuizen HJ. The lack of clinical value of peritoneal washing cytology in high risk patients undergoing risk-reducing salpingo-oophorectomy: a retrospective study and review. BMC Cancer. 2016;14(16):18.
Reitsma W, de Bock GH, Oosterwijk JC, Bart J, Hollema H, Mourits MJ. Support of the ‘fallopian tube hypothesis’ in a prospective series of risk-reducing salpingo-oophorectomy specimens. Eur J Cancer. 2013;49(1):132–41.
Jazaeri AA, Bryant JL, Park H, Li H, Dahiya N, Stoler MH, et al. Molecular requirements for transformation of fallopian tube epithelial cells into serous carcinoma. Neoplasia. 2011;13(10):899–911.
Powell CB, Chen LM, McLennan J, Crawford B, Zaloudek C, Rabban JT, et al. Risk-reducing salpingo-oophorectomy (RRSO) in BRCA mutation carriers: experience with a consecutive series of 111 patients using a standardized surgical-pathological protocol. Int J Gynecol Cancer. 2011;21(5):846–51.
The Pelvic-Ovarian Cancer Interception (POINT) Project. http://www.bwhpathology.org/POINT/point_purpose.aspx. Accessed Jan 2018.
Acknowledgements
The authors want to thank Lara Vargas, MD (Gynecology Department, Sanatorio Allende, Córdoba, Argentina) for her contribution in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
The study was previously approved by the Internal Review Board in accordance with the ethical standards of the Helsinki Declaration of 1975, revised in 1983.
Informed consent
For this type of study formal consent was not required.
Rights and permissions
About this article
Cite this article
Minig, L., Cabrera, S., Oliver, R. et al. Pathology findings and clinical outcomes after risk reduction salpingo-oophorectomy in BRCA mutation carriers: a multicenter Spanish study. Clin Transl Oncol 20, 1337–1344 (2018). https://doi.org/10.1007/s12094-018-1865-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-018-1865-9