Abstract
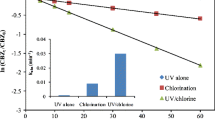
Carbamazepine (CBZ) is an anti-epileptic and anti-convulsant drug widely used for the treatment of epilepsy and other bipolar disorders. Ozone as an advanced oxidation process has been widely used for the degradation of CBZ resulting in the formation of transformation products (ozonides). The present research aims to isolate and identify potential microorganism, capable of degradation of CBZ and its transformation products. The cell viability and cytotoxicity of pure CBZ and their ozone transformation products were evaluated using the cells of Pseudomonas sp. strain KSH-1 through cell viability assay tests. The cells metabolic activity was assessed at varying CBZ concentrations (~ 10–25 ppm, pure CBZ) and cumulatively for ozone transformation products. For pure CBZ, % cell viability decreases as CBZ concentration increases, while, in case of post-ozonated CBZ transformation products, the viability decreases initially and then increases upon exposure of ozone with a maximum cell viability of 97 ± 2.8% evaluated for 2 h post-ozonated samples.




Similar content being viewed by others
References
Papageorgiou M, Kosma C, Lambropoulou D (2016) Seasonal occurrence, removal, mass loading and environmental risk assessment of 55 pharmaceuticals and personal care products in a municipal wastewater treatment plant in Central Greece. Sci Total Environ 543:547–569. https://doi.org/10.1016/j.scitotenv.2015.11.047
Rudrashetti AP, Jadeja NB, Gandhi D, Juwarkar AA, Sharma A, Kapley A, Pandey RA (2017) Microbial population shift caused by sulfamethoxazole in engineered-Soil Aquifer Treatment (e-SAT) system. World J Microbiol Biotechnol 33:121. https://doi.org/10.1007/s11274-017-2284-8
Cunningham VL, Perino C, D’Aco VJ, Hartmann A, Bechter R (2010) Human health risk assessment of carbamazepine in surface waters of North America and Europe. Regul Toxicol Pharmacol 56:343–351. https://doi.org/10.1016/j.yrtph.2009.10.006
Dwivedi K, Morone A, Pratape V, Chakrabarti T, Pandey RA (2017) Carbamazepine and oxcarbazepine removal in pharmaceutical wastewater treatment plant using a mass balance approach: A case study. Korean J Chem Eng 34:2662–2671. https://doi.org/10.1007/s11814-017-0190-2
Clara M, Strenn B, Kreuzinger N (2004) Carbamazepine as a possible anthropogenic marker in the aquatic environment: investigations on the behaviour of Carbamazepine in wastewater treatment and during groundwater infiltration. Water Res 38:947–954. https://doi.org/10.1016/j.watres.2003.10.058
Caliman FA, Gavrilescu M (2009) Pharmaceuticals, personal care products and endocrine disrupting agents in the environment: a review. CLEAN Soil Air Water 37:277–303. https://doi.org/10.1002/clen.200900038
Li A, Cai R, Cui D, Qiu T, Pang C, Yang J, Ma F, Ren N (2013) Characterization and biodegradation kinetics of a new cold-adapted carbamazepine-degrading bacterium, Pseudomonas sp. CBZ-4. J Environ Sci 25:2281–2290. https://doi.org/10.1016/S1001-0742(12)60293-9
Prakash J, Sharma R, Ray S, Koul S, Kalia VC (2017) Wastewater: a potential bioenergy resource. Indian J Microbiol. https://doi.org/10.1007/s12088-017-0703-z
Dwivedi K, Morone A, Chakrabarti T, Pandey RA (2016) Evaluation and optimization of Fenton pretreatment integrated with granulated activated carbon (GAC) filtration for carbamazepine removal from complex wastewater of pharmaceutical industry. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2016.12.054
Klavarioti M, Mantzavinos D, Kassinos D (2009) Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ Int 35:402–417. https://doi.org/10.1016/j.envint.2008.07.009
Espejo A, Aguinaco A, Amat AM, Beltrán FJ (2014) Some ozone advanced oxidation processes to improve the biological removal of selected pharmaceutical contaminants from urban wastewater. J Environ Sci Health A Tox Hazard Subst Environ Eng 49:410–421. https://doi.org/10.1080/10934529.2014.854652
Dwivedi K, Rudrashetti A, Sharma A, Lokhande S, Chakrabarti T, Pandey RA (2016) Evaluation of fenton and ozone treatment processes for the degradation of psychiatric drugs carbamazepine (CBZ) and oxcarbazepine (oxCBZ) in simulated wastewater (SWW). J Environ Sci Eng 58:1–10
McDowell DC, Huber MM, Wagner M, von Gunten U, Ternes TA (2005) Ozonation of Carbamazepine in drinking water: identification and kinetic study of major oxidation products. Environ Sci Technol 39:8014–8022. https://doi.org/10.1021/es050043l
Hubner U, Seiwert B, Reemtsma T, Jekel M (2014) Ozonation products of carbamazepine and their removal from secondary effluents by soil aquifer treatment: indications from column experiments. Water Res 49:34–43. https://doi.org/10.1016/j.watres.2013.11.016
Dai G, Wang B, Fu C, Dong R, Huang J, Deng S, Wang Y, Yu G (2016) Pharmaceuticals and personal care products (PPCPs) in urban and suburban rivers of Beijing, China: occurrence, source apportionment and potential ecological risk. Environ Sci Process Impacts 18:445–455. https://doi.org/10.1039/c6em00018e
Vasiliadou IA, Molina R, Martínez F, Melero JA (2013) Biological removal of pharmaceutical and personal care products by a mixed microbial culture: sorption, desorption and biodegradation. Biochem Eng J 81:108–119. https://doi.org/10.1016/j.bej.2013.10.010
van Meerloo J, Kaspers GJ, Cloos J (2011) Cell sensitivity assays: the MTT assay. Methods Mol Biol 731:237–245. https://doi.org/10.1007/978-1-61779-080-5_20
Rudrashetti AP, Dwivedi K, Gandhi D, Juwarkar AA, Sharma A, Pandey RA (2016) Ozone treatment of sulfamethoxazole in aqueous matrix and toxicity of its degradation products on Pseudomonas aeruginosa and Enterobacter xiangfangensis species. J Environ Sci Eng 58:73–78
Alharbi SK, Price WE, Kang J, Fujioka T, Nghiem LD (2016) Ozonation of carbamazepine, diclofenac, sulfamethoxazole and trimethoprim and formation of major oxidation products. Desalin Water Treat 57:29340–29351. https://doi.org/10.1080/19443994.2016.1172986
Acknowledgements
Kshitiz Dwivedi is supported by fellowship from Department of Science and Technology (DST) under the program INSPIRE (Grant No. DST-INSPIRE/IF-120178). We express our sincere thanks to Dr. Sanjeev Kanojiya from Sophisticated Analytical Instrument Facility (SAIF, supported by DST), CSIR-Central Drug Research Institute (CDRI), Lucknow for assistance with LC–MS and LC–MS/MS analysis. The authors would like to acknowledge Knowledge Resource Centre (KRC), CSIR-NEERI, for plagiarism check for which the accession number is CSIR-NEERI/KRC/2017/OCT/EBGD/3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dwivedi, K., Rudrashetti, A.P., Chakrabarti, T. et al. Transformation Products of Carbamazepine (CBZ) After Ozonation and their Toxicity Evaluation Using Pseudomonas sp. Strain KSH-1 in Aqueous Matrices. Indian J Microbiol 58, 193–200 (2018). https://doi.org/10.1007/s12088-018-0715-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-018-0715-3