Abstract
We propose a stochastic model for interacting species in a metacommunity in order to study the factors affecting the intensity of the competition/colonization trade-off as a coexistence mechanism in metacommunities. We particularly focus on the role of the number of local communities and the number of refuges for the inferior competitor. The stochastic component is associated with the dispersal process and is represented by Poisson random measures. Thus, this stochastic model includes two dynamic scales: a continuous one, which refers to the interactions among species, and a low frequency one, referring to dispersal following a Poisson scheme. We show the well-posedness of the model and that it is possible to study its long-term behavior using Lyapunov exponents; the extinction of a species is associated with a negative slope in the time trajectory of the Lyapunov exponent, otherwise, it is equal to zero. We show that the competition/colonization trade-off is a function of the dispersal rate of the inferior competitor, and that it becomes less intense as the number of local communities increases, while the opposite is true with an increase in the number of refuges for the inferior competitor. We also show that under a priority effect type of scenario, dispersal can reverse priority effects and generate coexistence. Our results emphasize the importance of coexistence mechanisms related to the topology of the system of local communities, and its relationship with dispersal, in affecting the result of competition in local communities.







Similar content being viewed by others
References
Aiken CM, Navarrete SA (2014) Coexistence of competitors in marine metacommunities: environmental variability, edge effects, and the dispersal niche. Ecol 95(8):2289–2302
Amarasekare P (2003) Competitive coexistence in spatially structured environments: a synthesis. Ecol Lett 6(12):1109–1122
Amarasekare P (2004) Spatial variation and density-dependent dispersal in competitive coexistence. Proc R Soc London Ser B Biological Sci 271(1547):1497–1506
Amarasekare P, Hoopes MF, Mouquet N, Holyoak M (2004) Mechanisms of coexistence in competitive metacommunities. Am Nat 164(3):310–326
Amarasekare P, Nisbet RM (2001) Spatial heterogeneity, source-sink dynamics, and the local coexistence of competing species. Am Nat 158(6):572–584
Applebaum D (2009) Lévy processes and stochastic calculus. Cambridge University Press, Cambridge
Bansaye V, Méléard S (2015) Stochastic models for structured populations. Springer, Berlin
Bao J, Mao X, Yin G, Yuan C (2011) Competitive lotka–Volterra population dynamics with jumps. Nonlinear Anal Theor Methods Appl 74(17):6601–6616
Barter E, Gross T (2017) Spatial effects in meta-foodwebs. Sci Rep 7(1):1–10
Benaïm M, Schreiber SJ (2019) Persistence and extinction for stochastic ecological models with internal and external variables. J Math Biol 79(1):393–431
Boettiger C (2018) From noise to knowledge: how randomness generates novel phenomena and reveals information. Ecol Lett 21(8):1255–1267
Bolker BM, Pacala SW (1999) Spatial moment equations for plant competition: understanding spatial strategies and the advantages of short dispersal. Am Nat 153(6):575–602
Brechtel A, Gross T, Drossel B (2019) Far-ranging generalist top predators enhance the stability of meta-foodwebs. Sci Rep 9(1):1–15
Cadotte MW (2006) Dispersal and species diversity: a meta-analysis. Am Nat 167(6):913–924
Calcagno V, Mouquet N, Jarne P, David P (2006) Coexistence in a metacommunity: the competition–colonization trade-off is not dead. Ecol Lett 9(8):897–907
Cattiaux P, Méléard S (2010) Competitive or weak cooperative stochastic lotka–Volterra systems conditioned on non-extinction. J Math Biol 60(6):797–829
Chesson P (1994) Multispecies competition in variable environments. Theor Popul Biol 45 (3):227–276
Chesson P (2000) General theory of competitive coexistence in spatially-varying environments. Theor Popul Biol 58(3):211–237
Chesson P (2018) Updates on mechanisms of maintenance of species diversity. J Ecol 106 (5):1773–1794
Chesson PL (1982) The stabilizing effect of a random environment. J Math Biol 15(1):1–36
Cornell HV, Lawton JH (1992) Species interactions, local and regional processes, and limits to the richness of ecological communities: a theoretical perspective. J Anim Ecol 61(1):1–12
Courchamp F, Berec L, Gascoigne J (2008) Allee effects in ecology and conservation. Oxford University Press, Oxford
De Masi A, Galves A, Löcherbach E, Presutti E (2015) Hydrodynamic limit for interacting neurons. J Stat Phys 158(4):866–902
De Meester L, Vanoverbeke J, Kilsdonk LJ, Urban MC (2016) Evolving perspectives on monopolization and priority effects. Trends Ecol Evol 31(2):136–146
Dieckmann U, Law R (1996) The dynamical theory of coevolution: a derivation from stochastic ecological processes. J Math Biol 34(5-6):579–612
Du NH, Sam VH (2006) Dynamics of a stochastic lotka–Volterra model perturbed by white noise. J Math Anal Appl 324(1):82–97
Durrett R, Levin S (1998) Spatial aspects of interspecific competition. Theor Popul Biol 53 (1):30–43
Economo EP, Keitt TH (2008) Species diversity in neutral metacommunities: a network approach. Ecol Lett 11(1):52–62
Economo EP, Keitt TH (2010) Network isolation and local diversity in neutral metacommunities. Oikos 119(8):1355–1363
Forbes AE, Chase JM (2002) The role of habitat connectivity and landscape geometry in experimental zooplankton metacommunities. Oikos 96(3):433–440
Fournier N, Löcherbach E (2016) On a toy model of interacting neurons. Annales de l’Institut Henri poincaré Probabilités et Statistiques 52(4):1844–1876
Freilich MA, Rebolledo R, Corcoran D, Marquet PA (2020) Reconstructing ecological networks with noisy dynamics. Proc R Soc A 476(2237):20190739
Gravel D, Massol F, Leibold MA (2016) Stability and complexity in model meta-ecosystems. Nat Commun 7(1):1–8
Ha S-Y, Liu J-G (2009) A simple proof of the cucker-smale flocking dynamics and mean-field limit. Commun Math Sci 7(2):297–325
Hanski I (1982) Dynamics of regional distribution: the core and satellite species hypothesis. Oikos 38(2):210–221
Hanski I, Ovaskainen O (2000) The metapopulation capacity of a fragmented landscape. Nature 404(6779):755–758
Haskovec J (2013) Flocking dynamics and mean-field limit in the cucker–smale-type model with topological interactions. Phys D Nonlinear Phenom 261:42–51
Hastings A (1980) Disturbance, coexistence, history, and competition for space. Theor Popul Biol 18(3):363–373
Hening A, Nguyen DH (2018a) Coexistence and extinction for stochastic Kolmogorov systems. Ann Appl Probab 28(3):1893–1942
Hening A, Nguyen DH (2018b) Stochastic lotka–Volterra food chains. J Math Biol 77(1):135–163
Hening A, Nguyen DH, Yin G (2018) Stochastic population growth in spatially heterogeneous environments: the density-dependent case. J Math Biol 76(3):697–754
Hofbauer J, Sigmund K (1998) Evolutionary games and population dynamics. Cambridge University Press, Cambridge
Holt RD (1993) Ecology at the mesoscale: the influence of regional processes on local communities. In: Ricklefs, Schluter (eds) Species Diversity: Historical and Geographical Perspectives. University of Chicago, p 77–88
Holt RD (1996) Food webs in space: an island biogeographic perspective. In: Polis GA, Winemiller KO (eds) Food webs. Springer, pp 313–323
Holt RD (2009) Toward a trophic island biogeography. In: Losos JB, Ricklefs RE (eds) The theory of island biogeography revisited. Princeton University Press Princeton, NJ, pp 143–185
Holt RD, Hoopes MF, Holyoak M, Leibold MA, Holt RD (2005) Food web dynamics in a metacommunity context. In: Metacommunities: spatial dynamics and ecological communities. University of Chicago Press Chicago, Illinois, pp 68–94
Holyoak M (2000) Habitat subdivision causes changes in food web structure. Ecol Lett 3(6):509–515
Hubbell SP (2001) The unified neutral theory of biodiversity and biogeography (MPB-32), volume 32. Princeton University Press
Hubbell SP (2005) Neutral theory in community ecology and the hypothesis of functional equivalence. Funct Ecol 19(1):166–172
Ikeda N, Watanabe S (2014) Stochastic differential equations and diffusion processes. Elsevier, Amsterdam
Jocque M, Field R, Brendonck L, De Meester L (2010) Climatic control of dispersal–ecological specialization trade-offs: a metacommunity process at the heart of the latitudinal diversity gradient? Glob Ecol Biogeogr 19(2):244–252
Keymer JE, Marquet PA, Velasco-Hernández JX, Levin SA (2000) Extinction thresholds and metapopulation persistence in dynamic landscapes. Am Nat 156(5):478–494
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D et al (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7(7):601–613
Levin SA (1974) Dispersion and population interactions. Am Nat 108(960):207–228
Levins R (1969) The effect of random variations of different types on population growth. Proc Natl Acad Sci 62(4):1061–1065
Levins R, Culver D (1971) Regional coexistence of species and competition between rare species. Natl Acade Sci 68(6):1246–1248
Li X, Mao X (2009) Population dynamical behavior of non-autonomous lotka-Volterra competitive system with random perturbation. Discrete Contin Dyn Syst-A 24(2):523
Lions P-L, Sznitman A-S (1984) Stochastic differential equations with reflecting boundary conditions. Commun Pur Appl Math 37(4):511–537
Loreau M, Mouquet N (1999) Immigration and the maintenance of local species diversity. Am Nat 154(4):427–440
Luo Q, Mao X (2007) Stochastic population dynamics under regime switching. J Math Anal Appl 334(1):69–84
MacArthur RH, Wilson EO (1963) An equilibrium theory of insular zoogeography. Evolution 17(4):373–387
Mao X (2007) Stochastic differential equations and applications. Elsevier, Amsterdam
Mao X, Marion G, Renshaw E (2002) Environmental Brownian noise suppresses explosions in population dynamics. Stoch Process Appl 97(1):95–110
Mao X, Sabanis S, Renshaw E (2003) Asymptotic behaviour of the stochastic lotka–Volterra model. J Math Anal Appl 287(1):141–156
Marquet PA (1997) A source-sink patch occupancy metapopulation model. Rev Chil Hist Nat 70:371–380
Marquet PA, Allen AP, Brown JH, Dunne JA, Enquist BJ, Gillooly JF, Gowaty PA, Green JL, Harte J, Hubbell SP et al (2014) On theory in ecology. Bioscience 64(8):701–710
Marquet PA, Espinoza G, Abades SR, Ganz A, Rebolledo R (2017) On the proportional abundance of species: integrating population genetics and community ecology. Sci Rep 7(1):1–10
Marquet PA, Tejo M, Rebolledo R (2020) What is the species richness distribution?. In: Dobson A, Holt RD, Tilman D (eds) Unsolved problems in ecology. Princeton University Press, pp 177–188
Mena-Lorca J, Velasco-Hernández JX, Marquet PA (2006) Coexistence in metacommunities: a tree-species model. Math Biosci 202(1):42–56
Mohd MH, Murray R, Plank MJ, Godsoe W (2016) Effects of dispersal and stochasticity on the presence–absence of multiple species. Ecol Model 342:49–59
Murray JD (2007) Mathematical biology: i. an introduction, vol 17. Springer Science & Business Media, Berlin
Murrell DJ, Law R (2003) Heteromyopia and the spatial coexistence of similar competitors. Ecol Lett 6(1):48–59
Nagylaki T (1979) The island model with stochastic migration. Genetics 91(1):163–176
Nee S, May RM (1992) Dynamics of metapopulations: habitat destruction and competitive coexistence. J Anim Ecol 61(1):37–40
Neuhauser C, Pacala SW (1999) An explicitly spatial version of the lotka-Volterra model with interspecific competition. Ann Appl Probab 9(4):1226–1259
Ovaskainen O, Meerson B (2010) Stochastic models of population extinction. Trends Ecol Evol 25(11):643–652
Pakdaman K, Perthame B, Salort D (2009) Dynamics of a structured neuron population. Nonlinearity 23(1):55
Pakdaman K, Perthame B, Salort D (2013) Relaxation and self-sustained oscillations in the time elapsed neuron network model. SIAM J Appl Math 73(3):1260–1279
Pillai P, Gonzalez A, Loreau M (2011) Metacommunity theory explains the emergence of food web complexity. Proc Natl Acad Sci 108(48):19293–19298
Plitzko SJ, Drossel B (2015) The effect of dispersal between patches on the stability of large trophic food webs. Theor Ecol 8(2):233–244
Quininao C, Touboul J (2015) Limits and dynamics of randomly connected neuronal networks. Acta Appl Math 136(1):167–192
Rebolledo R, Navarrete SA, Kéfi S., Rojas S, Marquet PA (2019) An open-system approach to complex biological networks. SIAM J Appl Math 79(2):619–640
Richter-Dyn N, Goel NS (1972) On the extinction of a colonizing species. Theor Popul Biol 3(4):406–433
Ricklefs RE (1987) Community diversity: relative roles of local and regional processes. Science 235(4785):167–171
Saavedra S, Rohr RP, Bascompte J, Godoy O, Kraft NJ, Levine JM (2017) A structural approach for understanding multispecies coexistence. Ecol Monogr 87(3):470–486
Schreiber SJ (2017) Coexistence in the face of uncertainty. In: Recent progress and modern challenges in applied mathematics, modeling and computational science. Springer, pp 349–384
Schreiber SJ, Killingback TP (2013) Spatial heterogeneity promotes coexistence of rock–paper–scissors metacommunities. Theor Popul Biol 86:1–11
Seeley TD, Visscher PK, Schlegel T, Hogan PM, Franks NR, Marshall JA (2012) Stop signals provide cross inhibition in collective decision-making by honeybee swarms. Science 335(6064):108–111
Singh P, Baruah G (2020) Higher order interactions and species coexistence. Theoretical Ecology, 1–13
Skorokhod AV (1961) Stochastic equations for diffusion processes in a bounded region. Theory Probab Its Appl 6(3):264–274
Skorokhod AV (1962) Stochastic equations for diffusion processes in a bounded region. ii. Theory Probab Its Appl 7(1):3–23
Southwood TR (1977) Habitat, the templet for ecological strategies? J Anim Ecol 46(2):337–365
Tanaka H (2002) Stochastic differential equations with reflecting boundary condition in convex regions. In: Stochastic processes: selected papers of Hiroshi Tanaka. World Scientific, pp 157–171
Tejo M, Niklitschek-Soto S, Vásquez C, Marquet PA (2017) Single species dynamics under climate change. Theor Ecol 10(2):181–193
Tilman D (1994) Competition and biodiversity in spatially structured habitats. Ecol 75(1):2–16
Turnbull LA, Rees M, Crawley MJ (1999) Seed mass and the competition/colonization trade-off: a sowing experiment. J Ecol 87:899–912
Venail PA, MacLean RC, Bouvier T, Brockhurst MA, Hochberg ME, Mouquet N (2008) Diversity and productivity peak at intermediate dispersal rate in evolving metacommunities. Nature 452(7184):210–214
Warren BH, Simberloff D, Ricklefs RE, Aguilée R, Condamine FL, Gravel D, Morlon H, Mouquet N, Rosindell J, Casquet J et al (2015) Islands as model systems in ecology and evolution: prospects fifty years after macArthur-Wilson. Ecol Lett 18(2):200–217
Wilson DS (1992) Complex interactions in metacommunities, with implications for biodiversity and higher levels of selection. Ecol 73(6):1984–2000
Wilson EO, MacArthur RH (1967) The theory of island biogeography. Princeton University Press, Princeton
Xiong J, Li X, Wang H (2019) The survival analysis of a stochastic Lotka-Volterra competition model with a coexistence equilibrium. Math Biosci 1(k1k2):1–k1k2
Zhou Y, Kot M (2011) Discrete-time growth-dispersal models with shifting species ranges. Theor Ecol 4(1):13–25
Acknowledgments
We acknowledge funding from projects FONDECYT 1161023 and 1200925 and Project EcoDep. We also acknowledge funding from a postdoctoral fellowship to Cristóbal Quiñinao (FONDECYT 3170435), and support from projects CIMFAV-CIDI and Redes CONICYT 180018 to Rolando Rebolledo and AFB170008. We thank Sergio Navarrete for stimulating discussions on the role of dispersal and spatial structure in metacommunities and two anonymous reviewers for their insightful comments, suggestions, and criticisms.
Funding
FONDECYT 1161023, 1200925, FONDECYT 3170435), CIMFAV-CIDI, AFB170008, and Redes CONICYT 180018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Authors’ contributions
All authors participated in the design and analysis, and jointly wrote the manuscript.
Data availability
Not applicable.
Code availability
All codes used in the simulations are available upon request.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1: Proofs
Proof of Proposition 1
Let ∥⋅∥ be the usual Euclidean norm in \(\mathbb {R}^{d}\), and for real matrices Q = (qlk)l,k= 1,..,d define the norm \(\parallel \cdot \parallel _{\max \limits }\) as \(\parallel Q\parallel _{\max \limits }=\underset {1\leq l,k\leq d}{\max \limits }\mid q_{lk}\mid \). Notice that for an \(\mathbb {R}^{d}\)-vector Y, we have \(\parallel QY\parallel \leq \sqrt {d}\parallel Q\parallel _{\max \limits }\parallel Y\parallel \). Let \(\boldsymbol {x}\in \mathbb {R}^{IJ}\). Then, we have the following inequalities: \(\parallel {\Lambda }(\boldsymbol {x})\boldsymbol {x}\parallel \leq \sqrt {IJ}\parallel {\Lambda }(\boldsymbol {x})\parallel _{\max \limits }\parallel \boldsymbol {x}\parallel \) and \(\parallel B(\boldsymbol {x},z)\parallel =\parallel \text {vec}(B(\boldsymbol {x},z))\parallel \leq \sqrt {IJ}\parallel B(\boldsymbol {x},z)\parallel _{\max \limits }\leq \sqrt {IJ}\parallel \beta (z)\parallel _{\max \limits }\parallel \boldsymbol {x}\parallel \), where
Using those inequalities, we have that:
where \(K(\boldsymbol {x})=\max \limits \{\parallel F(\boldsymbol {x})\parallel ^{2},IJ\parallel {\Lambda }(\boldsymbol {x})\parallel _{\max \limits }^{2},IJ{\int \limits }_{0}^{\infty }\parallel \beta (z)\parallel _{\max \limits }^{2}dz\}\). By (A.1), ∥X(0) ∥ is bounded, so we can choose a large K0 ≫ K(X(0)) such that D(x) ≤ K0(1+ ∥x ∥2), for all x belonging to the set \(\{\boldsymbol {x}\in \mathbb {R}^{IJ}:K(\boldsymbol {x})\leq K_{0}\}\). This means that (4) has a local linear growth.
On the other hand, by (A.2) F(⋅) is a locally Lipschitz function. Let L(x) = Λ(x)x, for all \(\boldsymbol {x}\in \mathbb {R}^{IJ}\). Now, note that for any \(\boldsymbol {x},\boldsymbol {y}\in \mathbb {R}^{IJ}\) we have that L(x) − L(y) = [Λ(x) + Λ(y)][x −y]/2 + [Λ(x) −Λ(y)][x + y]/2, and then, \(\parallel L(\boldsymbol {x})-L(\boldsymbol {y})\parallel \leq (\sqrt {IJ}/2)\parallel {\Lambda }(\boldsymbol {x})+{\Lambda }(\boldsymbol {y})\parallel _{\max \limits }\parallel \boldsymbol {x}-\boldsymbol {y}\parallel +(\sqrt {IJ}/2)\parallel {\Lambda }(\boldsymbol {x})-{\Lambda }(\boldsymbol {y})\parallel _{\max \limits }\parallel \boldsymbol {x}+\boldsymbol {y}\parallel \), where
by Hölder inequality. That is,
and thus for any \(\boldsymbol {x},\boldsymbol {y}\in \mathbb {R}^{IJ}\) such that ∥x ∥∨∥y ∥≤ M, where M is a constant such that M ≫∥X(0) ∥, there exists a constant HM such that ∥ L(x) − L(y) ∥≤ HM ∥x −y ∥. So, it means that L(⋅) is locally Lipschitz as well. Therefore, conditions of Theorem 9.1 in Chapter IV from Ikeda and Watanabe (2014) (linear growth and Lipschitz conditions) are locally satisfied, and hence, we have existence and uniqueness of a local solution. \(\square \)
Proof of Proposition 2
Recalling the formulation Xij(t) of (3), we define the following quantity:
which models the total biomass of species j over the whole system (i.e., archipelago) at time t. We recall that no biomass is transferred from outside the archipelago and all the emigrant biomass of any species in any community at most lands over some of the other communities. Therefore, the stochastic part of (3) must satisfy:
almost surely, since \({\sum }_{i=1}^{I}\varepsilon _{ii^{\prime }}\leq 0\) for any \(i^{\prime }\). Let Y = (Y1,...,YJ)T. Now, in (6), let Ξ be the J × IJ matrix such that ΞX = Y. We have that:
by (16). Then we get:
By (A.3), it is clear that \(\sup _{t\in \mathbb {R}_{+}}\max \limits _{j=1,..,J}Y_{j}(t)<\infty \), almost surely, and (7) follows from the fact that \(\max \limits _{j=1,..,J}Y_{j}(\cdot )\geq \max \limits _{i=1,..,I;j=1,..,J}X_{ij}(\cdot )\), almost surely.
Now, we shall prove that extinction occurs in the long term. First, notice that under structural assumptions made in (6), we can write in (3) fij(x) in the form \(\tilde {f}_{ij}(x)x\). Therefore, for any \(t\in \mathbb {R}_{+}\), i = 1,...,I and j = 1,...,J we have:
where \({\mathscr{M}}_{ij}([0,b_{j}],t)=-({\int \limits }_{0}^{\infty }1_{\left \{z\leq b_{j}\right \}}N_{ij}(dz,t)-tb_{j})=tb_{j}-N_{ij}([0,b_{j}],t)\) is a square-integrable martingale. This implies that:
where
and RJ is the bounded subset of \(\mathbb {R}^{J}_{+}\) which delimits the state space of (Xi1(⋅),...,XiJ(⋅))T for any i = 1,...,I. On the other hand, we have that:
and thus,
Notice that it must satisfy \(b_{j}\exp \{-1\}+\inf _{\mathsf {x}\in R^{J}}G_{ij}(\mathsf {x})\leq 0\) if Xij(0) > 0, since otherwise \(\mathbb {E}(X_{ij}(t))\rightarrow \infty \) when \(t\rightarrow \infty \), which contradicts the fact that Xij(⋅) is bounded. So, if Xij(0) > 0, \(\exp \left \{t\inf _{\mathsf {x}\in R^{J}}G_{ij}(\mathsf {x})+{\mathscr{M}}_{ij}([0,b_{j}],t)\right \}\) never reaches zero in finite time, and then neither does Xij(⋅). \(\square \)
Proof of proposition 3
First notice that the vector b(x) can be decomposed into b(x) = βx, where \(\boldsymbol {\beta }={\int \limits }_{0}^{\infty }\beta (z)dz\) and β(z) is the IJ × IJ matrix defined in (14). Also, we have that \(\parallel B(\boldsymbol {x},z)\parallel \leq \sqrt {IJ}\parallel \beta (z)\parallel _{\max \limits }\parallel \boldsymbol {x}\parallel \).
We start by showing the existence of a Lyapunov exponent for (6). Consider its representation given by (11). According to Theorem 6.8.2 in Applebaum (2009), we have to show that the alternative condition to the Assumption 6.8.1, given in Equation (6.42), is satisfied for our system (11) for all x ∈R. That is, the existence of a positive constant \(L^{\prime }\) such that \(\boldsymbol {x}^{\mathrm {T}}{\Gamma }(\boldsymbol {x})\boldsymbol {x}+\boldsymbol {x}^{\mathrm {T}}\boldsymbol {b}(\boldsymbol {x})+{\int \limits }_{0}^{\infty }\parallel B(\boldsymbol {x},z)\parallel ^{2}dz\leq L^{\prime }(1+\parallel \boldsymbol {x}\parallel ^{2})\). In fact, by the above, we have that:
where \(L^{\prime \prime }=\max \limits \{\sup _{\boldsymbol {x}\in \boldsymbol {R}}{\Gamma }({\boldsymbol {x}}),\parallel \boldsymbol {\beta }\parallel ,IJ{\int \limits }_{0}^{\infty }\parallel \beta (z)\parallel _{\max \limits }^{2}dz\}\). Clearly it also implies that the process (12) has a Lyapunov exponent.
Now, consider that there exists a constant c > 0 such that \({\mathscr{L}}\langle u_{IJ},\boldsymbol {x}\rangle \leq -c\langle u_{IJ},\boldsymbol {x}\rangle \) for all x ∈R. Define \(\mathbb {E}_{\boldsymbol {x}}(\langle u_{IJ},\boldsymbol {X}(t)\rangle ):=\mathbb {E}(\langle u_{IJ},\boldsymbol {X}(t)\rangle \mid \boldsymbol {X}(0)=\boldsymbol {x})\), for all \(t\in \mathbb {R}_{+}\). Applying it to (12) we obtain:
Then, by Gronwall’s inequality we get:
which implies that 〈uIJ,X(⋅)〉 goes exponentially fast to 0. \(\square \)
Proof of Lemma 4
For a chosen uIJ, there exists \(i^{\prime }=1,...,I\) and \(j^{\prime }=1,...,J\) such that \(\mathbb {E}_{\boldsymbol {x}}(X_{i^{\prime }j^{\prime }}(\cdot ))\leq \mathbb {E}_{\boldsymbol {x}}(\langle u_{IJ},\boldsymbol {X}(\cdot )\rangle )\), where \(\mathbb {E}_{\boldsymbol {x}}(X_{i^{\prime }j^{\prime }}(\cdot ))\) satisfies (17) for \(i=i^{\prime }\) and \(j=j^{\prime }\) given \(X_{i^{\prime }j^{\prime }}(0)=\boldsymbol {x}\). Therefore, extinction can only occur in an exponentially fast way. \(\square \)
Appendix 2. Some results of a classical competitive Lotka-Volterra system
In the present appendix we recall some results of a classical competitive Lotka-Volterra system and its corresponding parameter space, in order to enlighten the long-term solutions and the reader realize how the presence of dispersal can transform such parameter space and bifurcation diagrams. Notice that as we will start treating the classic case, in a local community without dispersal, first we do not consider the subscript i. Consider then the equations:
For any steady state (x1,x2), the eigenvalues of the Jacobian matrix are given by:
where tr(J) and \(\det (J)\) stand for the trace and determinant of the matrix
In the following we simply write T = tr(J) and \(D=\det (J)\). The standard trace-determinant analysis tells us that
-
Case \(4\det (J)>\text {tr}(J)^{2}\): the system has two complex eigenvalues, If tr(J) < 0 then the steady state is stable. On the other hand, if tr(J) > 0 then the steady state is unstable.
-
Case \(\det (J)<0\): in this scenario, both eigenvalues are positive and have different sign, thus the steady state is unstable.
-
Case \(0<4\det (J)<\text {tr}(J)^{2}\): if tr(J) > 0 then the two eigenvalues are positive and the steady state is unstable. Otherwise, tr(J) < 0 then the configuration is asymptotically stable.
Recall also that steady states are either (0,0), (0,−r2/ α22), (−r1/α11, 0) or
Then we have:
-
(0,0): it follows that T = r1 + r2 and D = r1r2, in which case the extinction scenario is unconditionally unstable.
-
(0,−r2/α22): we have
$$ T=r_{1}-r_{2}\frac{\alpha_{12}}{\alpha_{22}}-r_{2}\text{ and }D=-r_{2}\left( r_{1}-r_{2}\frac{\alpha_{12}}{\alpha_{22}}\right). $$Stability of this fixed point is reduced to the condition
$$ r_{1}<r_{2}\frac{\alpha_{12}}{\alpha_{22}}. $$ -
(−r1/α11, 0): similarly,
$$ T=-r_{1}+r_{2}-r_{1}\frac{\alpha_{22}}{\alpha_{11}}\text{ and }D=-r_{1}\left( r_{2}-r_{1}\frac{\alpha_{21}}{\alpha_{11}}\right), $$by consequence, the stability of this fixed point holds true if and only if
$$ r_{2}<r_{1}\frac{\alpha_{21}}{\alpha_{11}}. $$ -
\((\bar x_{1},\bar x_{2})\): it follows that
$$ T=\alpha_{11}\bar x_{1}+\alpha_{22}\bar x_{2}\text{ and }D=\big(\alpha_{11}\alpha_{22}-\alpha_{12}\alpha_{21}\big)\bar x_{1}\bar x_{2}, $$the existence and stability of this final fixed point is only under the conditions
$$ \alpha_{11}\alpha_{22}>\alpha_{12}\alpha_{21},\quad r_{1}\alpha_{22}<r_{2}\alpha_{12}\quad\text{and}\quad r_{2}\alpha_{11}<r_{1}\alpha_{21}. $$Notice that, since αii < 0 then second and third conditions are equivalent to
$$ r_{1}>r_{2}\frac{\alpha_{12}}{\alpha_{22}}\quad\text{and}\quad r_{2}>\frac{r_{1}\alpha_{21}}{\alpha_{11}}, $$thus we have the instability of all previous scenarios.
Analysis of the phase plane
If we come back to the numerical example with 2 species in a local community without dispersal, the system of equations are:
thus defining the interacting matrix
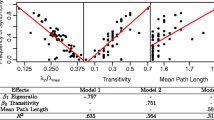
Parameters rj’s are intrinsic for each species, thus we can consider them as fixed. Therefore, we can make an analysis of the stability of the fixed points under the two nonnegative quantities:
where α21 = λ21, α12 = λ12, α11 = −r1/K1 and α22 = −r2/K2. It follows that the coexistence of the two population can happen only under the restriction
and similar conditions appear for any of the other fixed points. We summarize this parameter dependence of fixed points, in Fig. 8, where without loss of generality we have assumed that r1 ≥ r2. Long-term behavior correspond to the classical Lotka-Volterra competition: a) coexistence, b) species 1 wins, c) species 2 wins, and d) strong competition (where the species with the largest initial biomass wins), that correspond to each of the zones in Fig. 8. The conditions for each scenario are:
-
(a)
p1 < r2/r1 and p2 < r1/r2. Since in this case p1p2 < 1, coexistence of species is expected.
-
(b)
p1 > r2/r1 and p2 < r1/r2. It follows that species 1 kicks in and at large times it is expected to converge asymptotically towards (−r1/α11, 0) = (K1, 0).
-
(c)
p1 < r2/r1 and p2 > r1/r2. Species 2 dominates and the solution is expected to converge asymptotically towards (0,−r2/α22, 0) = (0,K2). Moreover, since p1 < r2/r1 and p1p2 > 1 we get that
$$ \bar x_{1} = \frac{-r_{1}\alpha_{22}+r_{2}\alpha_{12}}{\alpha_{11}\alpha_{22}-\alpha_{12}\alpha_{21}} = \frac{1}{\alpha_{11}}\frac{r_{2}p_{2}-r_{1}}{1-p_{1}p_{2}}>0, $$and
$$ \bar x_{2} = \frac{-r_{2}\alpha_{11}+r_{1}\alpha_{21}}{\alpha_{11}\alpha_{22}-\alpha_{12}\alpha_{21}}=\frac{1}{\alpha_{22}}\frac{r_{1}p_{1}-r_{2}}{1-p_{1}p_{2}}<0, $$i.e., the second coordinate of the coexistence scenario vanishes and
-
d)
p1 > r2/r1 and p2 > r1/r2. In this last zone conditions imply that both winner-takes-all scenario are possible, and that the coexistence fixed point \((\bar x_{1},\bar x_{2})\) exists but it is unstable. Therefore, according to initial condition, we expect to converge at large times towards either (0,K2) or (K1, 0).
Outcomes of the model for a single community and two populations when r1 > r2 and on the parameter space p1 = −λ21K1/r1 v/s p2 = −λ12K2/r2. Long-term behavior on each zone: (a) coexistence, (b) species 1 wins, (c) species 2 wins and (d) strong competition. Points 1 and 3 are the coordinates for the analyzes carried out in “Coexistence and the competition/colonization trade-off” and “Effect of the metacommunity spatial structure” and Appendix C, respectively
Appendix 3. Additional results for two species competition with priority effects
Results for the dynamics of two species in a central community within a metacommunity architecture with three local communities
For each pair (b1,b2) with bi taking values from 0.1 to 0.9 with incremental step 0.1, we performed 150 simulations. We report the empirical average of biomass of both species on the central community.
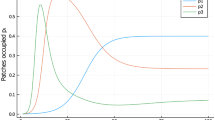
The results of the model are shown in the upper subplots of Fig. 9. One observes that the species with larger dispersal rates dominates the long-term solutions in the central community. In the bottom panel of Fig. 9 we plot the probability that species 1 wins as a function of the difference b1 − b2. We find a shape very similar to the ones described in literature (see, e.g., Calcagno et al. 2006).
Simulations made for parameters r1 = r2 = 2, Ki1 = Ki2 = 10 (for all i = 1,2,3). All εij are 0 except for ε21 = ε23 = −ε11 = −ε33 = 0.1 and λ21 = λ12 = − 0.21. (top) We show the empirical mean calculated for species 1 and 2 in a central community under a strong competition scenario where species with higher initial biomass will win according to the classical Lotka-Volterra model of competition without stochastic dispersal. (down) We show the probability that species 1 wins as a function of the difference between colonization rates b1 − b2. As expected, under a symmetric dynamics the species with larger bi dominates at long times. Dots represents the empirical mean of 150 simulations of the model, for the three-community architecture where the dispersion parameter of species 1 varies. The solid line corresponds to a nonlinear statistical fit
Results for two species competition under priority effects in a metacommunity with two local communities
The first case is a symmetrical one, in which the outcome is the expected one for the classical Lotka-Volterra model (i.e., for each community, the species with the larger initial biomass will win). In our example, species 1 wins in community 2 due to its larger initial biomass condition and species 2 wins in community 2 for analogous reasons (Fig. 10 upper panel). Here, species 1 cannot disperse and species 2 dispersal rate b2 is low. In the second scenario, a small increase in b2 and no dispersal of species 1, will reverse the competence in community 1, and species 2 will win in both communities (Fig. 10, middle panels). And in the third scenario, species 1 only lives in community 1, and a stronger dispersal intensity of species 2 and no dispersal of species 1, will cause coexistence in community 1 (Fig. 10, bottom panel). This last outcome is due to the rapid and massive exchange of biomass of species 2 from one community to another, which can maintain its biomass levels in both communities, specifically causing in community 1 that the biomass condition for winning frequently changes: sometimes species 1 locally dominates due to a temporary larger biomass and sometimes species 2 locally dominates due to the same reason. In Fig. 11 we can visualize the final outcome of the dynamics of both species in community 1 and under each scenario, through the slope (in the long term) of the graph \(\log (X_{ij}(t))\) vs t, which corresponds to the (estimated) Lyapunov exponent.
Here we show the biomass dynamics of two species in two communities under a priority effect scenario whereby the species that wins in competition is the one with higher initial biomass. The three panels represents three cases of the dynamics of the two species in the two communities, where the dispersal parameter of the inferior competitor species (the one with the lowest initial biomass) varies in each of these columns. The simulations where made over a mean of 50 trajectories and we considered the common parameters r1 = 1.6, r2 = 1, Ki1 = 12, Ki2 = 10 (for all i = 1.2), ε12 = ε21 = −ε11 = −ε22 = 0.5, λ12 = − 0.168 and λ21 = − 0.0875 in (8) for the three cases, with initial conditions given by X11(0) = 10, X12(0) = 1, X21(0) = 1 and X22(0) = 10. The first case is one where the dispersal rate of species 2 is low, b2 = 0.003, and no dispersal of species 1. In the second case we consider only a small increase in the dispersal rate of species 2 (b2 = 0.05). Notice that this subtle change makes this species to increase in biomass in community 1 reversing the priority effect. Finally, in the third case, in local community 2 only species 2 exists but as we increase its dispersal rate (b2 = 1) the priority effect coexistence in local community 1 is now possible
Convergence of Lyapunov exponents of species 1 and species 2 in community 1 for the corresponding cases of Fig. 10, from top to bottom. In continuous line is depicted one typical trajectory and in dashed line the mean of 50 trajectories, where we can see a softened version of \(\log (X_{ij}(t))\) vs t. The slopes of the long-term tendencies tell us the destination of the species: slope ≈ 0 will mean persistence and negative slope indicates extinction
Rights and permissions
About this article
Cite this article
Tejo, M., Quiñinao, C., Rebolledo, R. et al. Coexistence, dispersal and spatial structure in metacommunities: a stochastic model approach. Theor Ecol 14, 279–302 (2021). https://doi.org/10.1007/s12080-020-00496-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-020-00496-1