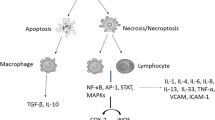
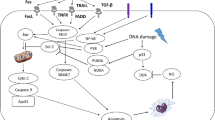
Abstract
Nowadays, using ionizing radiation (IR) is necessary for clinical, agricultural, nuclear energy or industrial applications. Accidental exposure to IR after a radiation terror or disaster poses a threat to human. In contrast to the old dogma of radiation toxicity, several experiments during the last two recent decades have revealed that intercellular signaling and communications play a key role in this procedure. Elevated level of cytokines and other intercellular signals increase oxidative damage and inflammatory responses via reduction/oxidation interactions (redox system). Intercellular signals induce production of free radicals and inflammatory mediators by some intermediate enzymes such as cyclooxygenase-2 (COX-2), nitric oxide synthase (NOS), NADPH oxidase, and also via triggering mitochondrial ROS. Furthermore, these signals facilitate cell to cell contact and increasing cell toxicity via cohort effect. Nitric oxide is a free radical with ability to act as an intercellular signal that induce DNA damage and changes in some signaling pathways in irradiated as well as non-irradiated adjacent cells. Targeting of these mediators by some anti-inflammatory agents or via antioxidants such as mitochondrial ROS scavengers opens a window to mitigate radiation toxicity after an accidental exposure. Experiments which have been done so far suggests that some cytokines such as IL-1β, TNF-α, TGF-β, IL-4 and IL-13 are some interesting targets that depend on irradiated organs and may help mitigate radiation toxicity. Moreover, animal experiments in recent years indicated that targeting of toll like receptors (TLRs) may be more useful for radioprotection and mitigation. In this review, we aimed to describe the role of intercellular interactions in oxidative injury, inflammation, cell death and killing effects of IR. Moreover, we described evidence on potential mitigation of radiation injury via targeting of these mediators.

Similar content being viewed by others
References
Ameziane-El-Hassani R, Talbot M, de Souza Dos Santos MC, Al Ghuzlan A, Hartl D, Bidart J-M, De Deken X, Miot F, Diallo I, De Vathaire F, Schlumberger M, Dupuy C (2015) NADPH oxidase DUOX1 promotes long-term persistence of oxidative stress after an exposure to irradiation. Proc Natl Acad Sci U S A 112:5051–5056
Anscher MS (2010) Targeting the TGF-β1 Pathway to Prevent Normal Tissue Injury After Cancer Therapy. The Oncologist 15:350–359
Anscher MS, Thrasher B, Rabbani Z, Teicher B, Vujaskovic Z (2006) Antitransforming growth factor-beta antibody 1D11 ameliorates normal tissue damage caused by high-dose radiation. Int J Radiat Oncol Biol Phys 65:876–881
Archambeau JO, Tovmasyan A, Pearlstein RD, Crapo JD, Batinic-Haberle I (2013) Superoxide dismutase mimic, MnTE-2-PyP(5+) ameliorates acute and chronic proctitis following focal proton irradiation of the rat rectum. Redox Biol 1:599–607
Autsavapromporn N, de Toledo SM, Little JB, Jay-Gerin JP, Harris AL, Azzam EI (2011) The role of gap junction communication and oxidative stress in the propagation of toxic effects among high-dose alpha-particle-irradiated human cells. Radiat Res 175:347–357
Autsavapromporn N, de Toledo SM, Jay-Gerin JP, Harris AL, Azzam EI (2013a) Human cell responses to ionizing radiation are differentially affected by the expressed connexins. J Radiat Res 54:251–259
Autsavapromporn N, Suzuki M, Plante I, Liu C, Uchihori Y, Hei TK, Azzam EI, Murakami T (2013b) Participation of gap junction communication in potentially lethal damage repair and DNA damage in human fibroblasts exposed to low- or high-LET radiation. Mutat Res 756:78–85
Azzam EI, de Toledo SM, Gooding T, Little JB (1998) Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat Res 150:497–504
Azzam EI, de Toledo SM, Little JB (2001) Direct evidence for the participation of gap junction-mediated intercellular communication in the transmission of damage signals from alpha -particle irradiated to nonirradiated cells. Proc Natl Acad Sci U S A 98:473–478
Azzam EI, de Toledo SM, Spitz DR, Little JB (2002) Oxidative metabolism modulates signal transduction and micronucleus formation in bystander cells from α-particle-irradiated normal human fibroblast cultures. Cancer Res 62:5436–5442
Barnaby F (1995) The effects of the atomic bombings of Hiroshima and Nagasaki. Med War 11:1–9
Battino M, Bullon P, Wilson M, Newman H (1999) Oxidative injury and inflammatory periodontal diseases: the challenge of anti-oxidants to free radicals and reactive oxygen species. Crit Rev Oral Biol Med 10:458–476
Berhane H, Shinde A, Kalash R, Xu K, Epperly MW, Goff J, Franicola D, Zhang X, Dixon T, Shields D, Wang H, Wipf P, Li S, Gao X, Greenberger JS (2014) Amelioration of radiation-induced oral cavity mucositis and distant bone marrow suppression in fanconi anemia Fancd2-/- (FVB/N) mice by intraoral GS-nitroxide JP4-039. Radiat Res 182:35–49
Blyth BJ, Sykes PJ (2011) Radiation-induced bystander effects: what are they, and how relevant are they to human radiation exposures? Radiat Res 176:139–157
Boerma M, Wang J, Sridharan V, Herbert JM, Hauer-Jensen M (2013) Pharmacological induction of transforming growth factor-beta1 in rat models enhances radiation injury in the intestine and the heart. PLoS One 8:e70479
Boerma M, Sridharan V, Mao XW, Nelson GA, Cheema AK, Koturbash I, Singh SP, Tackett AJ, Hauer-Jensen M (2016) Effects of ionizing radiation on the heart. Mutat Res 770:319–327
Brand MD, Affourtit C, Esteves TC, Green K, Lambert AJ, Miwa S, Pakay JL, Parker N (2004) Mitochondrial superoxide: production, biological effects, and activation of uncoupling proteins. Free Radic Biol Med 37:755–767
Burdelya LG, Krivokrysenko VI, Tallant TC, Strom E, Gleiberman AS, Gupta D, Kurnasov OV, Fort FL, Osterman AL, Didonato JA, Feinstein E, Gudkov AV (2008) An agonist of toll-like receptor 5 has radioprotective activity in mouse and primate models. Science 320:226–230
Calabrese V, Mancuso C, Calvani M, Rizzarelli E, Butterfield DA, Stella AM (2007) Nitric oxide in the central nervous system: neuroprotection versus neurotoxicity. Nat Rev Neurosci 8:766–775
Cali B, Ceolin S, Ceriani F, Bortolozzi M, Agnellini AH, Zorzi V, Predonzani A, Bronte V, Molon B, Mammano F (2015) Critical role of gap junction communication, calcium and nitric oxide signaling in bystander responses to focal photodynamic injury. Oncotarget 6:10161–10174
Chai Y, Calaf G, Zhou H, Ghandhi S, Elliston C, Wen G, Nohmi T, Amundson S, Hei T (2012) Radiation induced COX-2 expression and mutagenesis at non-targeted lung tissues of gpt delta transgenic mice. Br J Cancer 108:91–98
Chai Y, Lam R, Calaf G, Zhou H, Amundson S, Hei T (2013) Radiation-induced non-targeted response in vivo: role of the TGFβ-TGFBR1-COX-2 signalling pathway. Br J Cancer 108:1106–1112
Chang J, Feng W, Wang Y, Luo Y, Allen AR, Koturbash I, Turner J, Stewart B, Raber J, Hauer-Jensen M, Zhou D, Shao L (2015) Whole-body proton irradiation causes long-term damage to hematopoietic stem cells in mice. Radiat Res 183:240–248
Chaudhry MA, Omaruddin RA (2012) Differential regulation of microRNA expression in irradiated and bystander cells. Mol Biol (Mosk) 46:634–643
Cheki M, Yahyapour R, Farhood B, Rezaeyan A, Shabeeb D, Amini P, Rezapoor S, Najafi M (2018) COX-2 in Radiotherapy; a potential target for radioprotection and radiosensitization. Curr Mol Pharmacol. https://doi.org/10.2174/1874467211666180219102520
Cho HJ, Lee WH, Hwang OMH, Sonntag WE, Lee YW (2017) Role of NADPH oxidase in radiation-induced pro-oxidative and pro-inflammatory pathways in mouse brain. Int J Radiat Biol 93:1257–1266
Chung SI, Horton JA, Ramalingam TR, White AO, Chung EJ, Hudak KE, Scroggins BT, Arron JR, Wynn TA, Citrin DE (2016) IL-13 is a therapeutic target in radiation lung injury. Sci Rep 6
Collins-Underwood JR, Zhao W, Kooshki M, Robbins M (2007) Modulation of NADPH oxidase by ionizing radiation and its role in radiation-induced oxidative stress and inflammation in brain endothelium. Cancer Res 67:1381–1381
Dancea HC, Shareef MM, Ahmed MM (2009) Role of Radiation-induced TGF-beta Signaling in Cancer Therapy. Mol Cell Pharmacol 1
Dayal D, Martin SM, Owens KM, Aykin-Burns N, Zhu Y, Boominathan A, Pain D, Limoli CL, Goswami PC, Domann FE, Spitz DR (2009) Mitochondrial complex II dysfunction can contribute significantly to genomic instability after exposure to ionizing radiation. Radiat Res 172:737–745
de Toledo SM, Buonanno M, Harris AL, Azzam EI (2017) Genomic instability induced in distant progeny of bystander cells depends on the connexins expressed in the irradiated cells. Int J Radiat Biol 93:1182–1194
Dorr H, Meineke V (2011) Acute radiation syndrome caused by accidental radiation exposure - therapeutic principles. BMC Med 9:126
Douple EB, Mabuchi K, Cullings HM, Preston DL, Kodama K, Shimizu Y, Fujiwara S, Shore RE (2011) Long-term radiation-related health effects in a unique human population: lessons learned from the atomic bomb survivors of Hiroshima and Nagasaki. Disaster Med Public Health Prep 5(Suppl 1):S122–S133
Eldabaje R, Le DL, Huang W, Yang LX (2015) Radiation-associated Cardiac Injury. Anticancer Res 35:2487–2492
Fadok VA, Bratton DL, Konowal A, Freed PW, Westcott JY, Henson PM (1998) Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J Clin Invest 101:890–898
Fang Z, Xu A, Wu L, Hei TK, Hong M (2016) The role of protein kinase C alpha translocation in radiation-induced bystander effect. Sci Rep 6:25817
Fardid R, A S, Mosleh-Shirazi MA, Sharifzadeh S, Okhovat MA, Najafi M, Rezaeyan A, Abaszadeh A (2017) Melatonin ameliorates the production of COX-2, iNOS, and the formation of 8-OHdG in non-targeted lung tissue after pelvic irradiation. Cell J 19:324–331
Flechsig P, Dadrich M, Bickelhaupt S, Jenne J, Hauser K, Timke C, Peschke P, Hahn EW, Grone HJ, Yingling J, Lahn M, Wirkner U, Huber PE (2012) LY2109761 attenuates radiation-induced pulmonary murine fibrosis via reversal of TGF-beta and BMP-associated proinflammatory and proangiogenic signals. Clin Cancer Res 18:3616–3627
Gauter-Fleckenstein B, Fleckenstein K, Owzar K, Jiang C, Reboucas JS, Batinic-Haberle I, Vujaskovic Z (2010a) Early and late administration of MnTE-2-PyP5+ in mitigation and treatment of radiation-induced lung damage. Free Radic Biol Med 48:1034–1043
Gauter-Fleckenstein B, Fleckenstein K, Owzar K, Jiang C, Rebouças JS, Batinic-Haberle I, Vujaskovic Z (2010b) Early and late administration of MnTE-2-PyP 5+ in mitigation and treatment of radiation-induced lung damage. Free Radic Biol Med 48:1034–1043
Ghandhi SA, Yaghoubian B, Amundson SA (2008) Global gene expression analyses of bystander and alpha particle irradiated normal human lung fibroblasts: Synchronous and differential responses. BMC Med Genet 1:63–63
Ghobadi A, Shirazi A, Najafi M, Kahkesh MH, Rezapoor S (2017) Melatonin ameliorates radiation-induced oxidative stress at targeted and nontargeted lung tissue. J Med Phys 42(4):241–244
Ghosh S, Maurya DK, Krishna M (2008) Role of iNOS in bystander signaling between macrophages and lymphoma cells. Int J Radiat Oncol Biol Phys 72:1567–1574
Goff JP, Epperly MW, Dixon T, Wang H, Franicola D, Shields D, Wipf P, Li S, Gao X, Greenberger JS (2011) Radiobiologic effects of GS-nitroxide (JP4-039) on the hematopoietic syndrome. In Vivo 25:315–323
Golden E, Pellicciotta I, Demaria S, Barcellos-Hoff MH, Formenti S (2012) The convergence of radiation and immunogenic cell death signaling pathways. Front Oncol 2
Grammaticos P, Giannoula E, Fountos GP (2013) Acute radiation syndrome and chronic radiation syndrome. Hell J Nucl Med 16:56–59
Groves AM, Johnston CJ, Misra RS, Williams JP, Finkelstein JN (2016) Effects of IL-4 on pulmonary fibrosis and the accumulation and phenotype of macrophage subpopulations following thoracic irradiation. Int J Radiat Biol 92:754–765
Guo J, Chen Y, Lei X, Xu Y, Liu Z, Cai J, Gao F, Yang Y (2017) Monophosphoryl lipid a attenuates radiation injury through TLR4 activation. Oncotarget 8:86031–86042
Han W, Wu L, Chen S, Bao L, Zhang L, Jiang E, Zhao Y, Xu A, Hei TK, Yu Z (2007) Constitutive nitric oxide acting as a possible intercellular signaling molecule in the initiation of radiation-induced DNA double strand breaks in non-irradiated bystander cells. Oncogene 26:2330–2339
Hardee ME, Marciscano AE, Medina-Ramirez CM, Zagzag D, Narayana A, Lonning SM, Barcellos-Hoff MH (2012) Resistance of glioblastoma-initiating cells to radiation mediated by the tumor microenvironment can be abolished by inhibiting transforming growth factor-beta. Cancer Res 72:4119–4129
Havaki S, Kotsinas A, Chronopoulos E, Kletsas D, Georgakilas A, Gorgoulis VG (2015) The role of oxidative DNA damage in radiation induced bystander effect. Cancer Lett 356:43–51
He J, Jiang B-H (2016) Interplay between Reactive oxygen Species and MicroRNAs in Cancer. Curr Pharmacol Rep 2:82–90
Hildebrandt G, Loppnow G, Jahns J, Hindemith M, Anderegg U, Saalbach A, Kamprad F (2003) Inhibition of the iNOS pathway in inflammatory macrophages by low-dose X-irradiation in vitro. Is there a time dependence? Strahlenther Onkol 179:158–166
Hong C-W, Kim Y-M, Pyo H, Lee J-H, Kim S, Lee S, Noh JM (2013) Involvement of inducible nitric oxide synthase in radiation-induced vascular endothelial damage. J Radiat Res 54:1036–1042
Horton JA, Li F, Chung EJ, Hudak K, White A, Krausz K, Gonzalez F, Citrin D (2013) Quercetin inhibits radiation-induced skin fibrosis. Radiat Res 180:205–215
Hosseinimehr SJ, Fathi M, Ghasemi A, Shiadeh SN, Pourfallah TA (2017) Celecoxib mitigates genotoxicity induced by ionizing radiation in human blood lymphocytes. Res Pharm Sci 12:82–87
Huaux F, Liu T, McGarry B, Ullenbruch M, Phan SH (2003) Dual roles of IL-4 in lung injury and fibrosis. J Immunol 170:2083–2092
Jang CW, Chen CH, Chen CC, Chen JY, Su YH, Chen RH (2002) TGF-beta induces apoptosis through Smad-mediated expression of DAP-kinase. Nat Cell Biol 4:51–58
Jiang J, Belikova NA, Hoye AT, Zhao Q, Epperly MW, Greenberger JS, Wipf P, Kagan VE (2008) A mitochondria-targeted nitroxide/hemigramicidin S conjugate protects mouse embryonic cells against gamma irradiation. Int J Radiat Oncol Biol Phys 70:816–825
Jiang J, Stoyanovsky DA, Belikova NA, Tyurina YY, Zhao Q, Tungekar MA, Kapralova V, Huang Z, Mintz AH, Greenberger JS, Kagan VE (2009) A mitochondria-targeted triphenylphosphonium-conjugated nitroxide functions as a radioprotector/mitigator. Radiat Res 172:706–717
Jiang Y, Chen X, Tian W, Yin X, Wang J, Yang H (2014) The role of TGF-β1–miR-21–ROS pathway in bystander responses induced by irradiated non-small-cell lung cancer cells. Br J Cancer 111:772–780
Kamiya K, Ozasa K, Akiba S, Niwa O, Kodama K, Takamura N, Zaharieva EK, Kimura Y, Wakeford R (2015) Long-term effects of radiation exposure on health. Lancet 386:469–478
Karki R, Igwe OJ (2013) Toll-like receptor 4-mediated nuclear factor kappa B activation is essential for sensing exogenous oxidants to propagate and maintain oxidative/nitrosative cellular stress. PloS one 8
Khayyal MT, El-Ghazaly MA, El-Hazek RM, Nada AS (2009) The effects of celecoxib, a COX-2 selective inhibitor, on acute inflammation induced in irradiated rats. Inflammopharmacology 17:255–266
Klune JR, Dhupar R, Cardinal J, Billiar TR, Tsung A (2008) HMGB1: endogenous danger signaling. Mol Med 14:476–484
Koh RY, Lim CL, Uhal BD, Abdullah M, Vidyadaran S, Ho CC, Seow HF (2015) Inhibition of transforming growth factor-beta via the activin receptor-like kinase-5 inhibitor attenuates pulmonary fibrosis. Mol Med Rep 11:3808–3813
Kostyuk SV, Ermakov AV, Alekseeva AY, Smirnova TD, Glebova KV, Efremova LV, Baranova A, Veiko NN (2012) Role of extracellular DNA oxidative modification in radiation induced bystander effects in human endotheliocytes. Mutat Res 729:52–60
Krivokrysenko VI, Toshkov IA, Gleiberman AS, Krasnov P, Shyshynova I, Bespalov I, Maitra RK, Narizhneva NV, Singh VK, Whitnall MH, Purmal AA, Shakhov AN, Gudkov AV, Feinstein E (2015) The Toll-Like Receptor 5 Agonist Entolimod Mitigates Lethal Acute Radiation Syndrome in Non-Human Primates. PLoS One 10:e0135388
Krysko DV, Garg AD, Kaczmarek A, Krysko O, Agostinis P, Vandenabeele P (2012) Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer 12:860–875
Kurkjian CJ, Guo H, Montgomery ND, Cheng N, Yuan H, Merrill JR, Sempowski GD, Brickey WJ, Ting JP (2017) The Toll-Like Receptor 2/6 Agonist, FSL-1 Lipopeptide, Therapeutically Mitigates Acute Radiation Syndrome. Sci Rep 7:17355
Leach JK, Van Tuyle G, Lin P-S, Schmidt-Ullrich R, Mikkelsen RB (2001) Ionizing Radiation-induced, Mitochondria-dependent Generation of Reactive Oxygen/Nitrogen. Cancer Res 61:3894–3901
Lee JC, Krochak R, Blouin A, Kanterakis S, Chatterjee S, Arguiri E, Vachani A, Solomides CC, Cengel KA, Christofidou-Solomidou M (2009) Dietary flaxseed prevents radiation-induced oxidative lung damage, inflammation and fibrosis in a mouse model of thoracic radiation injury. Cancer Biol Ther 8:47–53
Li G, Tang D, Lotze MT (2013) Menage a Trois in stress: DAMPs, redox and autophagy. Semin Cancer Biol 23:380–390
Lorimore SA, Coates PJ, Scobie GE, Milne G, Wright EG (2001) Inflammatory-type responses after exposure to ionizing radiation in vivo: a mechanism for radiation-induced bystander effects? Oncogene 20:7085–7095
Mahmood J, Jelveh S, Zaidi A, Doctrow SR, Hill RP (2013) Mitigation of radiation-induced lung injury with EUK-207 and genistein: effects in adolescent rats. Radiat Res 179:125–134
Maier P, Hartmann L, Wenz F, Herskind C (2016) Cellular Pathways in Response to Ionizing Radiation and Their Targetability for Tumor Radiosensitization. Int J Mol Sci 17:102
Martin M, Lefaix J-L, Delanian S (2000) TGF-β1 and radiation fibrosis: a master switch and a specific therapeutic target? Int J Radiat Oncol Biol Phys 47:277–290
Medhora M, Gao F, Jacobs ER, Moulder JE (2012) Radiation damage to the lung: mitigation by angiotensin-converting enzyme (ACE) inhibitors. Respirology 17:66–71
Melisi D, Ishiyama S, Sclabas GM, Fleming JB, Xia Q, Tortora G, Abbruzzese JL, Chiao PJ (2008) LY2109761, a novel transforming growth factor beta receptor type I and type II dual inhibitor, as a therapeutic approach to suppressing pancreatic cancer metastasis. Mol Cancer Ther 7:829–840
Mi S, Li Z, Liu H, Hu ZW, Hua F (2012) Blocking IL-17A protects against lung injury-induced pulmonary fibrosis through promoting the activation of p50NF-kappaB. Yao Xue Xue Bao 47:739–744
Mittra I, Khare NK, Raghuram GV, Chaubal R, Khambatti F, Gupta D, Gaikwad A, Prasannan P, Singh A, Iyer A, Singh A, Upadhyay P, Nair NK, Mishra PK, Dutt A (2015) Circulating nucleic acids damage DNA of healthy cells by integrating into their genomes. J Biosci 40:91–111
Mittra I, Samant U, Sharma S, Raghuram GV, Saha T, Tidke P, Pancholi N, Gupta D, Prasannan P, Gaikwad A, Gardi N, Chaubal R, Upadhyay P, Pal K, Rane B, Shaikh A, Salunkhe S, Dutt S, Mishra PK, Khare NK, Nair NK, Dutt A (2017) Cell-free chromatin from dying cancer cells integrate into genomes of bystander healthy cells to induce DNA damage and inflammation. Cell Death Discov 3:17015
Mothersill C, Seymour CB (1998) Cell-cell contact during gamma irradiation is not required to induce a bystander effect in normal human keratinocytes: evidence for release during irradiation of a signal controlling survival into the medium. Radiat Res 149:256–262
Mothersill C, Seymour C (2012) Are Epigenetic Mechanisms Involved in Radiation-Induced Bystander Effects? Front Genet 3:74
Mothersill C, Stamato TD, Perez ML, Cummins R, Mooney R, Seymour CB (2000) Involvement of energy metabolism in the production of 'bystander effects' by radiation. Br J Cancer 82:1740–1746
Moulder JE (2003) Pharmacological intervention to prevent or ameliorate chronic radiation injuries. Semin Radiat Oncol 13:73–84
Moulder JE (2004) Post-irradiation approaches to treatment of radiation injuries in the context of radiological terrorism and radiation accidents: a review. Int J Radiat Biol 80:3–10
Multhoff G, Radons J (2012) Radiation, inflammation, and immune responses in cancer. Front Oncol 2
Najafi M, Fardid R, Hadadi G, Fardid M (2014) The mechanisms of radiation-induced bystander effect. J Biomed Phys Eng 4:163–172
Najafi M, Fardid R, Takhshid MA (2016) Radiation-Induced Oxidative Stress at Out-of-Field. Cell J 18:340–345
Najafi M, Salajegheh A, Rezaeyan A (2017a) Bystander Effect and Second Primary Cancers following Radiotherapy: What are its Significances? J Med Phys 42:55–56
Najafi M, Shirazi A, Motevaseli E, Rezaeyan AH, Salajegheh A, Rezapoor S (2017b) Melatonin as an anti-inflammatory agent in radiotherapy. Inflammopharmacology 25:403–413
Najafi M, Motevaseli E, Shirazi A, Geraily G, Rezaeyan A, Norouzi F, Rezapoor S, Abdollahi H (2018a) Mechanisms of inflammatory responses to radiation and normal tissues toxicity: clinical implications. Int J Radiat Biol 94:335–356
Najafi M, Cheki M, Rezapoor S, Geraily G, Motevaseli E, Carnovale C, Clementi E, Shirazi A (2018b) Metformin: prevention of genomic instability and cancer: a review. Mutat Res Genet Toxicol Environ Mutagen 827:1–8. https://doi.org/10.1016/j.mrgentox.2018.01.007
Nishioka A, Ogawa Y, Mima T, Jin YJ, Sonobe H, Kariya S, Kubota K, Yoshida S, Ueno H (2004) Histopathologic amelioration of fibroproliferative change in rat irradiated lung using soluble transforming growth factor-beta (TGF-beta) receptor mediated by adenoviral vector. Int J Radiat Oncol Biol Phys 58:1235–1241
Nishioka A, Ogawa Y, Kariya S, Hamada N, Nogami M, Inomata T, Ueno H (2015) Reduction of fibroproliferative changes in irradiated rat lung with soluble transforming growth factor-beta receptor. Mol Med Rep 11:2659–2663
Oikonomou N, Harokopos V, Zalevsky J, Valavanis C, Kotanidou A, Szymkowski DE (2006) Soluble TNF mediates the transition from pulmonary inflammation to fibrosis. PLoS One 1
Park JS, Gamboni-Robertson F, He Q, Svetkauskaite D, Kim JY, Strassheim D, Sohn JW, Yamada S, Maruyama I, Banerjee A, Ishizaka A, Abraham E (2006) High mobility group box 1 protein interacts with multiple Toll-like receptors. Am J Physiol Cell Physiol 290:C917–C924
Pazhanisamy SK, Li H, Wang Y, Batinic-Haberle I, Zhou D (2011) NADPH oxidase inhibition attenuates total body irradiation-induced haematopoietic genomic instability. Mutagenesis 26:431–435
Peter RU, Braun-Falco O, Birioukov A, Hacker N, Kerscher M, Peterseim U, Ruzicka T, Konz B, Plewig G (1994) Chronic cutaneous damage after accidental exposure to ionizing radiation: the Chernobyl experience. J Am Acad Dermatol 30:719–723
Piccinini A, Midwood K (2010) DAMPening inflammation by modulating TLR signalling. Mediat Inflamm 2010
Pietrofesa R, Turowski J, Tyagi S, Dukes F, Arguiri E, Busch TM, Gallagher-Colombo SM, Solomides CC, Cengel KA, Christofidou-Solomidou M (2013) Radiation mitigating properties of the lignan component in flaxseed. BMC Cancer 13:179
Piwkowska A, Rogacka D, Jankowski M, Angielski S (2013) Metformin reduces NAD(P)H oxidase activity in mouse cultured podocytes through purinergic dependent mechanism by increasing extracellular ATP concentration. Acta Biochim Pol 60:607–612
Prise KM, O'Sullivan JM (2009) Radiation-induced bystander signalling in cancer therapy. Nat Rev Cancer 9:351–360
Prise KM, Schettino G, Folkard M, Held KD (2005) New insights on cell death from radiation exposure. Lancet Oncol 6:520–528
Rabender C, Mezzaroma E, Mauro AG, Mullangi R, Abbate A, Anscher M, Hart B, Mikkelsen R (2016) IPW-5371 Proves Effective as a Radiation Countermeasure by Mitigating Radiation-Induced Late Effects. Radiat Res 186:478–488
Rajagopalan MS, Gupta K, Epperly MW, Franicola D, Zhang X, Wang H, Zhao H, Tyurin VA, Pierce JG, Kagan VE, Wipf P, Kanai AJ, Greenberger JS (2009) The mitochondria-targeted nitroxide JP4-039 augments potentially lethal irradiation damage repair. In: In Vivo, vol 23, pp 717–726
Reeves A, Zagurovskaya M, Gupta S, Shareef MM, Mohiuddin M, Ahmed MM (2007) Inhibition of transforming growth factor-beta signaling in normal lung epithelial cells confers resistance to ionizing radiation. Int J Radiat Oncol Biol Phys 68:187–195
Robbins ME, Zhao W (2004) Chronic oxidative stress and radiation-induced late normal tissue injury: a review. Int J Radiat Biol 80:251–259
Rube CE, Uthe D, Schmid KW, Richter KD, Wessel J, Schuck A, Willich N, Rube C (2000) Dose-dependent induction of transforming growth factor beta (TGF-beta) in the lung tissue of fibrosis-prone mice after thoracic irradiation. Int J Radiat Oncol Biol Phys 47:1033–1042
Rwigema JC, Beck B, Wang W, Doemling A, Epperly MW, Shields D, Goff JP, Franicola D, Dixon T, Frantz MC, Wipf P, Tyurina Y, Kagan VE, Wang H, Greenberger JS (2011) Two strategies for the development of mitochondrion-targeted small molecule radiation damage mitigators. Int J Radiat Oncol Biol Phys 80:860–868
Sakai Y, Yamamori T, Yoshikawa Y, Bo T, Suzuki M, Yamamoto K, Ago T, Inanami O (2018) NADPH oxidase 4 mediates ROS production in radiation-induced senescent cells and promotes migration of inflammatory cells. Free Radic Res 52:92–102
Schuster N, Krieglstein K (2002) Mechanisms of TGF-beta-mediated apoptosis. Cell Tissue Res 307:1–14
Shakhov AN, Singh VK, Bone F, Cheney A, Kononov Y, Krasnov P, Bratanova-Toshkova TK, Shakhova VV, Young J, Weil MM, Panoskaltsis-Mortari A, Orschell CM, Baker PS, Gudkov A, Feinstein E (2012) Prevention and mitigation of acute radiation syndrome in mice by synthetic lipopeptide agonists of Toll-like receptor 2 (TLR2). PLoS One 7:e33044
Shinde A, Berhane H, Rhieu BH, Kalash R, Xu K, Goff J, Epperly MW, Franicola D, Zhang X, Dixon T, Shields D, Wang H, Wipf P, Parmar K, Guinan E, Kagan V, Tyurin V, Ferris RL, Zhang X, Li S, Greenberger JS (2016) Intraoral Mitochondrial-Targeted GS-Nitroxide, JP4-039, Radioprotects Normal Tissue in Tumor-Bearing Radiosensitive Fancd2(-/-) (C57BL/6) Mice. Radiat Res 185:134–150
Szondy Z, Sarang Z, Kiss B, Garabuczi E, Koroskenyi K (2017) Anti-inflammatory Mechanisms Triggered by Apoptotic Cells during Their Clearance. Front Immunol 8:909
Tang D, Shi Y, Kang R, Li T, Xiao W, Wang H, Xiao X (2007) Hydrogen peroxide stimulates macrophages and monocytes to actively release HMGB1. J Leukoc Biol 81:741–747
Tartier L, Gilchrist S, Burdak-Rothkamm S, Folkard M, Prise KM (2007) Cytoplasmic irradiation induces mitochondrial-dependent 53BP1 protein relocalization in irradiated and bystander cells. Cancer Res 67:5872–5879
Tian W, Yin X, Wang L, Wang J, Zhu W, Cao J, Yang H (2015) The key role of miR-21-regulated SOD2 in the medium-mediated bystander responses in human fibroblasts induced by alpha-irradiated keratinocytes. Mutat Res 780:77–85
Tomita M, Matsumoto H, Funayama T, Yokota Y, Otsuka K, Maeda M, Kobayashi Y (2015) Nitric oxide-mediated bystander signal transduction induced by heavy-ion microbeam irradiation. Life Sci Space Res (Amst) 6:36–43
Toshkov IA, Gleiberman AS, Mett VL, Hutson AD, Singh AK, Gudkov AV, Burdelya LG (2017) Mitigation of Radiation-Induced Epithelial Damage by the TLR5 Agonist Entolimod in a Mouse Model of Fractionated Head and Neck Irradiation. Radiat Res 187:570–580
Valentin J (2005) Protecting people against radiation exposure in the event of a radiological attack. A report of The International Commission on Radiological Protection. Ann ICRP 35(1-110):iii–iiv
Wang B-Z, Wang L-P, Han H, Cao F-L, Li G-Y, Xu J-L, Wang X-W, Wang L-X (2014a) Interleukin-17A antagonist attenuates radiation-induced lung injuries in mice. Exp Lung Res 40:77–85
Wang, L. P., Wang, Y. W., Wang, B. Z., Sun, G. M., Wang, X. Y. & Xu, J. L. 2014b. Expression of interleukin-17A in lung tissues of irradiated mice and the influence of dexamethasone. ScientificWorldJournal, 2014, 251067.
Wang L, He L, Bao G, He X, Fan S, Wang H (2016) Ionizing Radiation Induces HMGB1 Cytoplasmic Translocation and Extracellular Release. Guo Ji Fang She Yi Xue He Yi Xue Za Zhi 40:91–99
Weyemi U, Redon CE, Aziz T, Choudhuri R, Maeda D, Parekh PR, Bonner MY, Arbiser JL, Bonner WM (2015) Inactivation of NADPH oxidases NOX4 and NOX5 protects human primary fibroblasts from ionizing radiation-induced DNA damage. Radiat Res 183:262–270
Willis, J., Epperly, M. W., Fisher, R., Zhang, X., Shields, D., Hou, W., Wang, H., Li, S., Wipf, P., Parmar, K., Guinan, E., Steinman, J. & Greenberger, J. S. 2018. Amelioration of Head and Neck Radiation-Induced Mucositis and Distant Marrow Suppression in Fanca(-/-) and Fancg(-/-) Mice by Intraoral Administration of GS-Nitroxide (JP4-039). Radiat Res.
Wu Y, Doroshow JH (2014a) Abstract 5358: IL-4/IL-13 induce Duox2/DuoxA2 expression and reactive oxygen production in human pancreatic and colon cancer cells. Cancer Res 74:5358
Wu, Y. & Doroshow, J. H. 2014b. IL-4/IL-13 induce Duox2/DuoxA2 expression and reactive oxygen production in human pancreatic and colon cancer cells. AACR.
Wu Q, Allouch A, Martins I, Modjtahedi N, Deutsch E, Perfettini J-L (2017) Macrophage biology plays a central role during ionizing radiation-elicited tumor response. Biomed J 40:200–211
Xavier S, Piek E, Fujii M, Javelaud D, Mauviel A, Flanders KC, Samuni AM, Felici A, Reiss M, Yarkoni S, Sowers A, Mitchell JB, Roberts AB, Russo A (2004) Amelioration of radiation-induced fibrosis: inhibition of transforming growth factor-beta signaling by halofuginone. J Biol Chem 279:15167–15176
Xu S, Ding N, Pei H, Hu W, Wei W, Zhang X, Zhou G, Wang J (2014) MiR-21 is involved in radiation-induced bystander effects. RNA Biol 11:1161–1170
Xu W, Wang T, Xu S, Xu S, Wu L, Wu Y, Bian P (2015) Radiation-induced epigenetic bystander effects demonstrated in Arabidopsis thaliana. Radiat Res 183:511–524
Yahyapour R, Amini P, Rezapoor S, Rezaeyan A, Farhood B, Cheki M, Fallah H, Najafi M (2017a) Targeting of inflammation for radiation protection and mitigation. Curr Mol Pharmacol. https://doi.org/10.2174/1874467210666171108165641
Yahyapour R, Motevaseli E, Rezaeyan A, Abdollahi H, Farhood B, Cheki M, Najafi M, Villa V (2017b) Mechanisms of radiation bystander and non-targeted effects: implications to radiation carcinogenesis and radiotherapy. Curr Radiopharm 11(1):34–45
Yahyapour R, Amini P, Rezapour S, Cheki M, Rezaeyan A, Farhood B, Shabeeb D, Musa AE, Fallah H, Najafi M (2018a) Radiation-induced inflammation and autoimmune diseases. Mil Med Res 5:9
Yahyapour R, Motevaseli E, Rezaeyan A, Abdollahi H, Farhood B, Cheki M, Rezapoor S, Shabeeb D, Musa AE, Najafi M, Villa V (2018b) Reduction-oxidation (redox) system in radiation-induced normal tissue injury: molecular mechanisms and implications in radiation therapeutics. Clin Transl Oncol. https://doi.org/10.1007/s12094-017-1828-6
Yahyapour R, Salajegheh A, Safari A, Abbasi S, Amini P, Rezaeyan A, Amraee A, Najafi M (2018c) Radiation-induced non-targeted effect and carcinogenesis; Implications in Clinical Radiotherapy. J Biomed Phys Eng. https://doi.org/10.22086/jbpe.v0i0.713
Yakovlev VA, Rabender CS, Sankala H, Gauter-Fleckenstein B, Fleckenstein K, Batinic-Haberle I, Jackson I, Vujaskovic Z, Anscher MS, Mikkelsen RB, Graves PR (2010) Proteomic analysis of radiation-induced changes in rat lung: Modulation by the superoxide dismutase mimetic MnTE-2-PyP(5+). Int J Radiat Oncol Biol Phys 78:547–554
Yang H, Asaad N, Held KD (2005) Medium-mediated intercellular communication is involved in bystander responses of X-ray-irradiated normal human fibroblasts. Oncogene 24:2096–2103
Yang H, Hreggvidsdottir HS, Palmblad K, Wang H, Ochani M, Li J, Lu B, Chavan S, Rosas-Ballina M, Al-Abed Y, Akira S, Bierhaus A, Erlandsson-Harris H, Andersson U, Tracey KJ (2010) A critical cysteine is required for HMGB1 binding to Toll-like receptor 4 and activation of macrophage cytokine release. Proc Natl Acad Sci U S A 107:11942–11947
Yang, H., Wang, H., Chavan, S. S. & Andersson, U. 2015. High Mobility Group Box Protein 1 (HMGB1): The Prototypical Endogenous Danger Molecule. Molecular Medicine, 21, S6-S12.
Yoshida T, Goto S, Kawakatsu M, Urata Y, Li T-s (2012) Mitochondrial dysfunction, a probable cause of persistent oxidative stress after exposure to ionizing radiation. Free Radic Res 46:147–153
Zhang M, Kleber S, Rohrich M, Timke C, Han N, Tuettenberg J, Martin-Villalba A, Debus J, Peschke P, Wirkner U, Lahn M, Huber PE (2011) Blockade of TGF-beta signaling by the TGFbetaR-I kinase inhibitor LY2109761 enhances radiation response and prolongs survival in glioblastoma. Cancer Res 71:7155–7167
Zhang H, Wang Y-a, Meng A, Yan H, Wang X, Niu J, Li J, Wang H (2013) Inhibiting TGFβ1 has a protective effect on mouse bone marrow suppression following ionizing radiation exposure in vitro. J Radiat Res 54:630–636
Zhao Y, de Toledo SM, Hu G, Hei TK, Azzam EI (2014) Connexins and cyclooxygenase-2 crosstalk in the expression of radiation-induced bystander effects. Br J Cancer 111:125–131
Zhou H, Ivanov VN, Gillespie J, Geard CR, Amundson SA, Brenner DJ, Yu Z, Lieberman HB, Hei TK (2005) Mechanism of radiation-induced bystander effect: role of the cyclooxygenase-2 signaling pathway. Proc Natl Acad Sci U S A 102:14641–14646
Zhou H, Ivanov VN, Lien Y-C, Davidson M, Hei TK (2008a) Mitochondrial Function and NF-κB Mediated Signaling in Radiation-Induced Bystander Effects. Cancer Res 68:2233–2240
Zhou H, Ivanov VN, Lien Y-C, Davidson M, Hei TK (2008b) Mitochondrial function and nuclear factor-κB–mediated signaling in radiation-induced bystander effects. Cancer Res 68:2233–2240
Zhou R, Yazdi AS, Menu P, Tschopp J (2011) A role for mitochondria in NLRP3 inflammasome activation. Nature 469:221–225
Zorov DB, Juhaszova M, Sollott SJ (2006) Mitochondrial ROS-induced ROS release: an update and review. Biochim Biophys Acta 1757:509–517
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No
Rights and permissions
About this article
Cite this article
Farhood, B., Goradel, N.H., Mortezaee, K. et al. Intercellular communications-redox interactions in radiation toxicity; potential targets for radiation mitigation. J. Cell Commun. Signal. 13, 3–16 (2019). https://doi.org/10.1007/s12079-018-0473-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-018-0473-3