Abstract
Introduction
The US Food and Drug Administration banned most “sweet” flavorants for use in cigarettes due to the concern that sweet flavors appeal to young, beginning smokers. However, many of the same flavors, including fruity and confection-associated aromas (e.g., vanilla), are still used in e-cigarettes. Sweet flavors may have a number of effects, including enhancement of the taste of other ingredients. The current work focused on the impact of model flavorants on the taste of a mixture of propylene glycol and vegetable glycerine, solvents used in most e-cigarettes and related products.
Methods
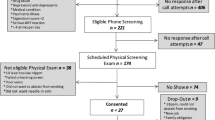
A device delivered mixtures of propylene glycol and vegetable glycerine into the mouth in parallel with puffs of clean air (control) or odorized air. Aromas included two “fruity” esters (“pineapple” and “banana”), two confection-associated aromas (“vanilla” and “caramel/malty”), menthol (not a “sweet” aroma, but commonly used in e-cigarettes), and a “burnt” aroma not expected to enhance flavor. Twenty young adults, aged 18–25, rated the sweetness, bitterness, and pleasantness of all stimuli (within-subjects design).
Results
Both fruity aromas significantly enhanced sweetness, both confection-associated aromas significantly enhanced pleasantness, and the caramel/malty aroma significantly reduced bitterness. Menthol and the “burnt” aroma had no measurable effects on the taste of solvent mixtures.
Conclusion
Some flavorants modulated the taste of solvents commonly used in e-cigarettes in ways consistent with an enhanced sensory profile.
Implications
If similar effects occur in actual products, improved flavor profiles could facilitate continued use, particularly in non-smokers experimenting with e-cigarettes and related products.



Similar content being viewed by others
References
111th Congress (2009) Family smoking prevention and tobacco control and federal retirement reform. https://www.gpo.gov/fdsys/pkg/PLAW-111publ31/html/PLAW-111publ31.htm. Accessed 27 July 2017.
Ambrose BK, Day HR, Rostron B, Conway KP, Borek N, Hyland A, Villanti AC (2015) Flavored tobacco product use among US youth aged 12-17 years, 2013-2014. JAMA 314(17):1871. doi:10.1001/jama.2015.13802
Audrain-McGovern J, Strasser AA, Wileyto EP (2016) The impact of flavoring on the rewarding and reinforcing value of e-cigarettes with nicotine among young adult amokers. Drug Alcohol Depend 166:263–267. doi:10.1016/j.drugalcdep.2016.06.030
Bartoshuk LM, Duffy VB, Green BG, Hoffman HJ, Ko CW, Lucchina LA, Marks LE, Snyder DJ, Weiffenbach JM (2004) Valid across-group comparisons with labeled scales: the gLMS versus magnitude matching. Physiol Behav 82(1):109–114. doi:10.1016/j.physbeh.2004.02.033
Berg CJ (2016) Preferred flavors and reasons for E-cigarette use and discontinued use among never, current, and former smokers. Int J Public Health 61(2):225–236. doi:10.1007/s00038-015-0764
Bingham AF, Birch GG, de Graaf C, Behan JM, Perring KD (1990) Sensory studies with sucrose-maltol mixtures. Chem Senses 15(4):447–456. doi:10.1093/chemse/15.4.447
Bohn MJ, Babor TF, Kranzler HR (1995) The Alcohol Use Disorders Identification Test (AUDIT): validation of a screening instrument for use in medical settings. J Stud Alcohol 56(4):423–432. doi:10.15288/jsa.1995.56.423
Carpenter CM, Wayne GF, Pauly JL, Koh HK, Connolly GN (2005) New cigarette brands with flavors that appeal to youth: tobacco marketing strategies. Health Aff (Project Hope) 24(6):1601–1610. doi:10.1377/hlthaff.24.6.1601
Clark CC, Lawless HT (1994) Limiting response alternatives in time-intensity scaling: an examination of the halo-dumping effect. Chem Senses 19(6):583–594. doi:10.1093/chemse/19.6.583
Connolly GN (2004) Sweet and spicy flavours: new brands for minorities and youth. Tob Control 13(3):211–212. doi:10.1136/tc.2004.009191
Duffy DB, Rawal S, Park J, Brand MH, Sharafi M, Bolling BW (2016) Characterizing and improving the sensory and hedonic responses to polyphenol-rich aronia berry juice. Appetite 107:116–125
Duke JC, Lee YO, Kim AE, Watson KA, Nonnemaker JM, Porter L (2014) Exposure to electronic cigarette television advertisements among youth and young adults. Pediatrics 134(1):e29–e36. doi:10.1542/peds.2014-0269
Dunnett CW (1955) A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc 50:1096–1121
FDA, Center for Tobacco Products (2016a) http://www.fda.gov/TobaccoProducts/Labeling/RulesRegulationsGuidance/ucm394909.htm
FDA, Center for Tobacco Products (2016b) https://www.fda.gov/TobaccoProducts/Labeling/ProductsIngredientsComponents/ucm456610.htm
Frank RA (2002) Response context affects judgments of flavor components in foods and beverages. Food Qual Pref 14:139–145. doi:10.1016/S0950-3293(02)00073-3
Frank RA, Byram J (1988) Taste-smell interactions are tastant and odorant dependent. Chem Senses 13(3):445–455. doi:10.1093/chemse/13.3.445
Giovenco DP, Hammond D, Corey CG, Ambrose BK, Delnevo CD (2015) E-cigarette market trends in traditional U.S. retail channels, 2012-2013. Nicotine Tob Res 17(10):1279–1283. doi:10.1093/ntr/ntu282
Green BG, Shaffer GS, Gilmore MM (1993) Derivation and evaluation of a semantic scale of oral sensation magnitude with apparent ratio properties. Chem Senses 18:683–702. doi:10.1093/chemse/18.6.683
Green BG, Dalton P, Cowart B, Shaffer G, Rankin K, Higgins J (1996) Evaluating the ‘labeled magnitude scale’ for measuring sensations of taste and smell. Chem Senses 21(3):323–334. doi:10.1093/chemse/21.3.323
Green BG, Nachtigal D, Hammond S, Lim J (2012) Enhancement of Retronasal odors by taste. Chem Senses 37(1):77–86. doi:10.1093/chemse/bjr068
Isogai T, Wise PM (2016) The effects of odor quality and temporal asynchrony on modulation of taste intensity by retronasal odor. doi:10.1093/chemse/bjw059
Kim H, Lim J, Buehler SS, Brinkman MC, Johnson NM, Wilson L, Cross KS, Clark PL (2016) Role of sweet and other flavours in liking and disliking of electronic cigarettes. Tob Control 25:ii55–ii61. doi:10.1136/tobaccocontrol-2016-053221
King BA, Patel R, Nguyen KH, Dube SR (2015) Trends in awareness and use of electronic cigarettes among US adults, 2010-2013. Nicotine Tob Res 17(2):219–227. doi:10.1093/ntr/ntu191
Klein SM, Giovino GA, Barker DC, Tworek C, Cummings KM, O'Connor RJ (2008) Use of flavored cigarettes among older adolescent and adult smokers: United States, 2004--2005. Nicotine Tob Res 10(7):1209–1214. doi:10.1080/14622200802163159
Kong G, Morean ME, Cavallo DA, Camenga DR, Krishnan-Sarin S (2015) Reasons for electronic cigarette experimentation and discontinuation among adolescents and young adults. Nicotine Tob Res 17(7):847–854. doi:10.1093/ntr/ntu257
Kreslake JM, Wayne GF, Connolly GN (2008) The menthol smoker: tobacco industry research on consumer sensory perception of menthol cigarettes and its role in smoking behavior. Nicotine Tob Res 10(4):705–715. doi:10.1080/14622200801979134
Krishnan-Sarin S, Morean ME, Camenga DR (2014) E-cigarette use among high school and middle school adolescents in Connecticut. Nicotine Tob Res:1–9. doi:10.1093/ntr/ntu243
Labbe D, Rytz A, Morgenegg C, Ali S, Martin A (2007) Subthreshold olfactory stimulation can enhance sweetness. Chem Senses 32(3):205–214. doi:10.1093/chemse/bjl040
Lawless HT, Clark CC (1992) Psychological biases in time intensity scaling. Food Technol 46:81–90
Lewis MJ, Wackowski O (2006) Dealing with an innovative industry: a look at flavored cigarettes promoted by mainstream brands. Am J Public Health 96(2):244–251. doi:10.2105/AJPH.2004.061200
Mcneill A, Brose LS, Calder R, Hitchman SC (2015) E-cigarettes: an evidence update. A report commissioned by Public Health England. Public Health England: 111. www.gov.uk/government/uploads/system/uploads/attachment_data/file/454516/Ecigarettes_an_evidence_update_A_report_commissioned_by_Public_Health_England.pdf
Mermelstein R, Fiore MC, Bernstein SL, Delnevo C, Croyle RT, Emmons KM, Eissenberg TE, Hyland A, Ostroff JS, Prochaska J, Ribisi K, Schnoll RA, Sindelar J, Vidrine JI, Winickoff JP, Web Hooper M (2016) Tobacco control research priorities for the next decade: Working group recommendations for 2016–2025. Report to the NCI Board of Scientific Advisors Web site. http://cancercontrol.cancer.gov/pdf/nci-tobacco-control-research-priorities-rpt-feb-2016.pdf
Minaker LM, Rashid A, Hammond D, Manske S (2014) Flavored tobacco use among Canadian students in grades 9 through 12: prevalence and patterns from the 2010-2011 youth smoking survey. Prev Chronic Dis 11(4):1–9. doi:10.5888/pcd11.140094
Mukai J, Tokuyama E, Ishizaka T, Okada S, Ochida T (2007) Inhibitory effect of aroma on the bitterness of branched-chain amino acid solutions. Chem Pharm Bull 55(11):1581–1584. doi:10.1248/cpb.55.1581
O’Connor RJ, Ashare RL, Cummings KM, Hawk LW (2007) Comparing smoking behaviors and exposures from flavored and unflavored cigarettes. Addict Behav 32(4):869–874. doi:10.1016/j.addbeh.2006.06.009
Oliver AJ, Jensen JA, Vogel RI, Anderson AJ, Hatsukami DK (2013) Flavored and nonflavored smokeless tobacco products: rate, pattern of use, and effects. Nicotine Tob Res 15(1):88–92. doi:10.1093/ntr/nts093
Pepper JK, Reiter PL, McRee A, Cameron LD, Gilkey MB, Brewer NT (2013) Adolescent males’ awareness of and willingness to try electronic cigarettes. J Adolesc Health 52(2):144–150. doi:10.1016/j.jadohealth.2012.09.014
Pepper JK, Ribisl KM, Brewer NT (2016). Adolescents’ interest in trying flavoured e-cigarettes. Tobacco Control. Published online first: 15 September 2016. doi: 10.1136/tobaccocontrol-2016-053174
Plevkova J, Kollarik M, Poliacek I, Brozmanova M, Surdenikova L, Tatar M (2013) The role of trigeminal nasal TRPM8-expressing afferent neurons in the antitussive effects of Menthol. J Appl Physiol (1985) 115(2):268–274. doi:10.1152/japplphysiol.01144.2012
Prescott J (1999) Flavour as a psychological construct: implications for perceiving and measuring the sensory qualities of foods. Food Qual Pref 10:349–356. doi:10.1016/S0950-3293(98)00048-2
Prescott J (2012) Multimodal chemosensory interactions and perception of flavor. In: The neural bases of multisensory processes. CRC Press, Boca Raton, pp 703–716 ncbi.nlm.nih.gov
Prescott J, Tylaor A, Roberts D (2004) Psychological processes in flavour perception. In: Flavour perception. Blackwell Publishing, London, pp 256–277
Rosbrook K, Green BG (2016) Sensory effect of menthol and nicotine in an e-cigarette. Nicotine Tob Res. doi:10.1093/ntr/ntw019
Sakai N, Kobayakawa T, Gotow N, Saito S, Imada S (2001) Enhancement of sweetness ratings of aspartame by a vanilla odor presented either by orthonasal or retronasal routes. Percept Mot Skills 92(3 Pt 2):1002–1008. doi:10.2466/pms.2001.92.3c.1002
Skinner HA (1982) The drug abuse screening test. Addict Behav 7(4):363–371. doi:10.1016/0306-4603(82)90005-3
Stevenson RJ, Prescott J, Boakes RA (1995) The acquisition of taste properties by odors. Learn Motiv 26:1–23. doi:10.1016/S0023-9690(05)80006-2
U.S. Department of Health and Human Services (2012) Preventing tobacco use among youth and young adults: a report of the surgeon general. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 3[accessed 2016 Apr 14]
U.S. Department of Health and Human Services (2014) The health consequences of smoking—50 years of progress: a report of the surgeon general. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 17 [accessed 2016 Apr 14]. aahb.wildapricot.org
Wang L, Yongcheng Z, Li Q, Zeng DD, Leischow SJ, Okamoto J (2015) An examination of electronic cigarette content on social media: analysis of E-cigarette flavor content on Reddit. Int J Environ Res Public Health 12(11):14933–14952. doi:10.3390/ijerph121114916
Willis DN, Liu B, Ha MA, Jordt SE, Morris JB (2011) Menthol attenuates respiratory irritation responses to multiple cigarette smoke irritants. FASEB J 25(12):4434–4444. doi:10.1096/fj.11-188383
Wise PM, Breslin PA (2011) Relationships among taste qualities assessed with response-context effects. Chem Senses 36(7):581–587. doi:10.1093/chemse/bjr024
Wise PM, Preti G, Eades J, Wysocki CJ (2011) The effect of menthol vapor on nasal sensitivity to chemical irritation. Nicotine Tob Res 13(10):989–997. doi:10.1093/ntr/ntr107
Acknowledgements
This work was funded in part by P50-CA-179546, NIH/NCI/FDA, University of Pennsylvania Tobacco Center of Regulatory Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded in part by P50-CA-179546, NIH/NCI/FDA, and in part by funds from [the authors’ institutions, not specifically listed to comply with double-blind peer review].
Conflict of Interest
PMW currently receives research funding from major food, beverage, and food ingredients companies. None of these companies provided funding for or were in any way involved in the current research. AAS has active research grants from the US National Institutes of Health (NIH) and US Food and Drug Administration (FDA) and has previously been funded by NIH and FDA, as well as a peer-reviewed GRAND grant sponsored by Pfizer (2008–2011). HN has recently been hired as a full-time employee by Kerry, a flavor and nutrition company. PDR declares that she has no potential conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments. All procedures were approved by an Institutional Review Board (IRB) at the University of [not included here to comply with double-blind peer review].
Informed Consent
Written, informed consent was obtained from all individual participants in the study, using IRB approved forms, before any study procedures were performed.
Rights and permissions
About this article
Cite this article
Rao, P.D., Husile, N., Strasser, A.A. et al. Pilot Experiment: the Effect of Added Flavorants on the Taste and Pleasantness of Mixtures of Glycerol and Propylene Glycol. Chem. Percept. 11, 1–9 (2018). https://doi.org/10.1007/s12078-017-9231-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12078-017-9231-9