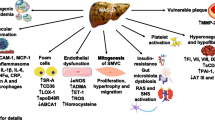
Abstract
Non-alcoholic fatty liver disease (NAFLD) is currently one of the leading forms of chronic liver disease, and its rising frequency worldwide has reached epidemic proportions. NAFLD, particularly its progressive variant NASH (non-alcoholic steatohepatitis), can lead to advanced fibrosis, cirrhosis, and HCC. The pathophysiologic mechanisms that contribute to the development and progression of NAFLD and NASH are complex, and as such myriad therapies are under investigation targeting different pathophysiological mechanisms. Incretin-based therapies, including GLP-1RAs and DPP-4 inhibitors and the inhibition of ASK1 pathway have provided two such novel mechanisms in the management of this disease, and will remain focus of this review.

Similar content being viewed by others
Abbreviations
- AAV8:
-
Adeno-associated virus vector 8
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- ASK-1:
-
Apoptosis signal-regulating kinase-1 (also known as MAP3K5)
- BMI:
-
Body mass index
- Cdc25A/C:
-
Cell division cycle 25A/C
- CFLAR:
-
CASP8 and FADD-like apoptosis regulator (also known as c-FLIP)
- CHOP:
-
CCAAT/enhancer-binding protein (C/EBP) homologous protein
- CREG:
-
Cellular repressor of E1A-stimulated genes
- CT:
-
Computed tomography
- DKK3:
-
Dickkopf-related protein 3
- DPP-4:
-
Dipeptidyl peptidase-4
- ER:
-
Endoplasmic reticulum
- FFA:
-
Free fatty acid
- GIP:
-
Glucose-dependent insulinotropic polypeptide
- GLP-1:
-
Glucagon-like peptide-1
- GLP-1R:
-
Glucagon-like peptide-1 receptor
- GLP-1RA:
-
Glucagon-like peptide-1 receptor agonist
- HCC:
-
Hepatocellular carcinoma
- HFD:
-
High fat diet
- HgA1c:
-
Hemoglobin A1c
- HNF4α:
-
Hepatocyte nuclear factor 4α
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- IL-6:
-
Interleukin 6
- IRS1:
-
Insulin receptor substrate 1
- JNK1:
-
c-Jun N-terminal kinase 1
- LDL:
-
Low density lipoprotein
- LOXL2:
-
Lysyl oxidase-like molecule 2
- MAPK:
-
Mitogen-activated protein kinase
- MKK:
-
Mitogen-activated protein kinase kinase
- MRI:
-
Magnetic resonance imaging
- MRE:
-
MRI elastography
- NAC:
-
N-acetyl-cysteine
- NAFLD:
-
Non-alcoholic fatty liver disease
- NAS:
-
NAFLD activity score
- NASH:
-
Non-alcoholic steatohepatitis
- PDFF:
-
Proton density fat fraction
- PP5:
-
Serine/threonine protein phosphatase 5
- PPARα:
-
Peroxisome proliferator-activated receptor alpha
- RCT:
-
Randomized controlled trial
- ROS:
-
Reactive oxygen species
- T2DM:
-
Type 2 diabetes mellitus
- TNF:
-
Tumor necrosis factor
- TRAF1:
-
TNF receptor-associated factor 1
- TRX:
-
Thioredoxin
References
McPherson S, Hardy T, Henderson E, Burt AD, Day CP, Anstee QM. Evidence of NAFLD progression from steatosis to fibrosing-steatohepatitis using paired biopsies: implications for prognosis and clinical management. J Hepatol 2015;62:1148–1155
Satapathy SK, Sanyal AJ. Epidemiology and natural history of nonalcoholic fatty liver disease. Semin Liver Dis 2015;35:221–235
Baggio LL, Drucker DJ. Biology of incretins: gLP-1 and GIP. Gastroenterology 2007;132:2131–2157
Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006;368:1696–1705
Nauck MA. Incretin-based therapies for type 2 diabetes mellitus: properties, functions, and clinical implications. Am J Med 2011;124:S3–S18
Nauck MA, Homberger E, Siegel EG, Allen RC, Eaton RP, Ebert R, Creutzfeldt W. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab 1986;63:492–498
Vilsboll T, Krarup T, Madsbad S, Holst JJ. Defective amplification of the late phase insulin response to glucose by GIP in obese Type II diabetic patients. Diabetologia 2002;45:1111–1119
Deacon CF, Knudsen LB, Madsen K, Wiberg FC, Jacobsen O, Holst JJ. Dipeptidyl peptidase IV resistant analogues of glucagon-like peptide-1 which have extended metabolic stability and improved biological activity. Diabetologia 1998;41:271–278
Deacon CF. Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: a comparative review. Diabetes Obes Metab 2011;13:7–18
Deacon CF, Hughes TE, Holst JJ. Dipeptidyl peptidase IV inhibition potentiates the insulinotropic effect of glucagon-like peptide 1 in the anesthetized pig. Diabetes 1998;47:764–769
Scheen AJ. Pharmacokinetics of dipeptidylpeptidase-4 inhibitors. Diabetes Obes Metab 2010;12:648–658
Gogebakan O, Osterhoff MA, Schuler R, Pivovarova O, Kruse M, Seltmann AC, Mosig AS, et al. GIP increases adipose tissue expression and blood levels of MCP-1 in humans and links high energy diets to inflammation: a randomised trial. Diabetologia 2015;58:1759–1768
Nauck M. Incretin therapies: highlighting common features and differences in the modes of action of glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Diabetes Obes Metab 2016;18:203–216
Armstrong MJ, Barton D, Gaunt P, Hull D, Guo K, Stocken D, Gough SC, et al. Liraglutide efficacy and action in non-alcoholic steatohepatitis (LEAN): study protocol for a phase II multicentre, double-blinded, randomised, controlled trial. BMJ Open 2013;3:e003995
Eguchi Y, Kitajima Y, Hyogo H, Takahashi H, Kojima M, Ono M, Araki N, et al. Pilot study of liraglutide effects in non-alcoholic steatohepatitis and non-alcoholic fatty liver disease with glucose intolerance in Japanese patients (LEAN-J). Hepatol Res 2015;45:269–278
Russell DH, Larson DF, Cardon SB, Copeland JG. Cyclosporine inhibits prolactin induction of ornithine decarboxylase in rat tissues. Mol Cell Endocrinol 1984;35:159–166
Lee J, Hong SW, Chae SW, Kim DH, Choi JH, Bae JC, Park SE, et al. Exendin-4 improves steatohepatitis by increasing Sirt1 expression in high-fat diet-induced obese C57BL/6J mice. PLoS One 2012;7:e31394
Ding X, Saxena NK, Lin S, Gupta NA, Anania FA. Exendin-4, a glucagon-like protein-1 (GLP-1) receptor agonist, reverses hepatic steatosis in ob/ob mice. Hepatology 2006;43:173–181
Mells JE, Fu PP, Sharma S, Olson D, Cheng L, Handy JA, Saxena NK, et al. Glp-1 analog, liraglutide, ameliorates hepatic steatosis and cardiac hypertrophy in C57BL/6 J mice fed a Western diet. Am J Physiol Gastrointest Liver Physiol 2012;302:G225–G235
Trevaskis JL, Griffin PS, Wittmer C, Neuschwander-Tetri BA, Brunt EM, Dolman CS, Erickson MR, et al. Glucagon-like peptide-1 receptor agonism improves metabolic, biochemical, and histopathological indices of nonalcoholic steatohepatitis in mice. Am J Physiol Gastrointest Liver Physiol 2012;302:G762–G772
Ben-Shlomo S, Zvibel I, Shnell M, Shlomai A, Chepurko E, Halpern Z, Barzilai N, et al. Glucagon-like peptide-1 reduces hepatic lipogenesis via activation of AMP-activated protein kinase. J Hepatol 2011;54:1214–1223
Gupta NA, Mells J, Dunham RM, Grakoui A, Handy J, Saxena NK, Anania FA. Glucagon-like peptide-1 receptor is present on human hepatocytes and has a direct role in decreasing hepatic steatosis in vitro by modulating elements of the insulin signaling pathway. Hepatology 2010;51:1584–1592
Yamamoto T, Nakade Y, Yamauchi T, Kobayashi Y, Ishii N, Ohashi T, Ito K, et al. Glucagon-like peptide-1 analogue prevents nonalcoholic steatohepatitis in non-obese mice. World J Gastroenterol 2016;22:2512–2523
Rahman K, Liu Y, Kumar P, Smith T, Thorn NE, Farris AB, Anania FA. C/EBP homologous protein modulates liraglutide-mediated attenuation of non-alcoholic steatohepatitis. Lab Invest 2016;96:895–908
Samson SL, Bajaj M. Potential of incretin-based therapies for non-alcoholic fatty liver disease. J Diabetes Complic 2013;27:401–406
Korner M, Stockli M, Waser B, Reubi JC. GLP-1 receptor expression in human tumors and human normal tissues: potential for in vivo targeting. J Nucl Med 2007;48:736–743
Burmeister MA, Ferre T, Ayala JE, King EM, Holt RM, Ayala JE. Acute activation of central GLP-1 receptors enhances hepatic insulin action and insulin secretion in high-fat-fed, insulin resistant mice. Am J Physiol Endocrinol Metab 2012;302:E334–E343
Klonoff DC, Buse JB, Nielsen LL, Guan X, Bowlus CL, Holcombe JH, Wintle ME, et al. Exenatide effects on diabetes, obesity, cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin 2008;24:275–286
Gluud LL, Knop FK, Vilsboll T. Effects of lixisenatide on elevated liver transaminases: systematic review with individual patient data meta-analysis of randomised controlled trials on patients with type 2 diabetes. BMJ Open 2014;4:e005325
Armstrong MJ, Houlihan DD, Rowe IA, Clausen WH, Elbrond B, Gough SC, Tomlinson JW, et al. Safety and efficacy of liraglutide in patients with type 2 diabetes and elevated liver enzymes: individual patient data meta-analysis of the LEAD program. Aliment Pharmacol Ther 2013;37:234–242
Jendle J, Nauck MA, Matthews DR, Frid A, Hermansen K, During M, Zdravkovic M, et al. Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes Metab 2009;11:1163–1172
Armstrong MJ, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, Hazlehurst JM, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016;387:679–690
Cui J, Philo L, Nguyen P, Hofflich H, Hernandez C, Bettencourt R, Richards L, et al. Sitagliptin vs. placebo for non-alcoholic fatty liver disease: a randomized controlled trial. J Hepatol 2016;65:369–376
Joy TR, McKenzie CA, Tirona RG, Summers K, Seney S, Chakrabarti S, Malhotra N, et al. Sitagliptin in patients with non-alcoholic steatohepatitis: a randomized, placebo-controlled trial. World J Gastroenterol 2017;23:141–150
Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018;67:328–357
Matsukawa J, Matsuzawa A, Takeda K, Ichijo H. The ASK1-MAP kinase cascades in mammalian stress response. J Biochem 2004;136:261–265
Preziosi ME, Monga SP. Update on the mechanisms of liver regeneration. Semin Liver Dis 2017;37:141–151
Mehta SJ, Jensen CD, Quinn VP, Schottinger JE, Zauber AG, Meester R, Laiyemo AO, et al. Race/ethnicity and adoption of a population health management approach to colorectal cancer screening in a community-based healthcare system. J Gen Intern Med 2016;31:1323–1330
Canbay A, Taimr P, Torok N, Higuchi H, Friedman S, Gores GJ. Apoptotic body engulfment by a human stellate cell line is profibrogenic. Lab Invest 2003;83:655–663
Hirsova P, Gores GJ. Death receptor-mediated cell death and proinflammatory signaling in nonalcoholic steatohepatitis. Cell Mol Gastroenterol Hepatol 2015;1:17–27
Hayakawa R, Hayakawa T, Takeda K, Ichijo H. Therapeutic targets in the ASK1-dependent stress signaling pathways. Proc Jpn Acad Ser B Phys Biol Sci 2012;88:434–453
Tobiume K, Matsuzawa A, Takahashi T, Nishitoh H, Morita K, Takeda K, Minowa O, et al. ASK1 is required for sustained activations of JNK/p38 MAP kinases and apoptosis. EMBO Rep 2001;2:222–228
Goldman EH, Chen L, Fu H. Activation of apoptosis signal-regulating kinase 1 by reactive oxygen species through dephosphorylation at serine 967 and 14-3-3 dissociation. J Biol Chem 2004;279:10442–10449
Saitoh M, Nishitoh H, Fujii M, Takeda K, Tobiume K, Sawada Y, Kawabata M, et al. Mammalian thioredoxin is a direct inhibitor of apoptosis signal-regulating kinase (ASK) 1. EMBO J 1998;17:2596–2606
Tobiume K, Saitoh M, Ichijo H. Activation of apoptosis signal-regulating kinase 1 by the stress-induced activating phosphorylation of pre-formed oligomer. J Cell Physiol 2002;191:95–104
Soga M, Matsuzawa A, Ichijo H. Oxidative stress-induced diseases via the ASK1 signaling pathway. Int J Cell Biol 2012;2012:439587
Samuel VT, Liu ZX, Qu X, Elder BD, Bilz S, Befroy D, Romanelli AJ, et al. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J Biol Chem 2004;279:32345–32353
Zarubin T, Han J. Activation and signaling of the p38 MAP kinase pathway. Cell Res 2005;15:11–18
Li H, Tang QZ, Liu C, Moon M, Chen M, Yan L, Bian ZY, et al. Cellular FLICE-inhibitory protein protects against cardiac remodeling induced by angiotensin II in mice. Hypertension 2010;56:1109–1117
Wang PX, Ji YX, Zhang XJ, Zhao LP, Yan ZZ, Zhang P, Shen LJ, et al. Targeting CASP8 and FADD-like apoptosis regulator ameliorates nonalcoholic steatohepatitis in mice and nonhuman primates. Nat Med 2017;23:439–449
Sakauchi C, Wakatsuki H, Ichijo H, Hattori K. Pleiotropic properties of ASK1. Biochim Biophys Acta 2017;1861:3030–3038
Xiang M, Wang PX, Wang AB, Zhang XJ, Zhang Y, Zhang P, Mei FH, et al. Targeting hepatic TRAF1-ASK1 signaling to improve inflammation, insulin resistance, and hepatic steatosis. J Hepatol 2016;64:1365–1377
Xie L, Wang PX, Zhang P, Zhang XJ, Zhao GN, Wang A, Guo J, et al. DKK3 expression in hepatocytes defines susceptibility to liver steatosis and obesity. J Hepatol 2016;65:113–124
Hirosumi J, Tuncman G, Chang L, Gorgun CZ, Uysal KT, Maeda K, Karin M, et al. A central role for JNK in obesity and insulin resistance. Nature 2002;420:333–336
Kodama Y, Brenner DA. c-Jun N-terminal kinase signaling in the pathogenesis of nonalcoholic fatty liver disease: multiple roles in multiple steps. Hepatology 2009;49:6–8
Schattenberg JM, Singh R, Wang Y, Lefkowitch JH, Rigoli RM, Scherer PE, Czaja MJ. JNK1 but not JNK2 promotes the development of steatohepatitis in mice. Hepatology 2006;43:163–172
Zhang QY, Zhao LP, Tian XX, Yan CH, Li Y, Liu YX, Wang PX, et al. The novel intracellular protein CREG inhibits hepatic steatosis, obesity, and insulin resistance. Hepatology 2017;66:834–854
Aouadi M, Binetruy B, Caron L, Le Marchand-Brustel Y, Bost F. Role of MAPKs in development and differentiation: lessons from knockout mice. Biochimie 2006;88:1091–1098
Kawarazaki Y, Ichijo H, Naguro I. Apoptosis signal-regulating kinase 1 as a therapeutic target. Expert Opin Ther Targets 2014;18:651–664
Loomba R, Lawitz E, Mantry PS, Jayakumar S, Caldwell SH, Arnold H, Diehl AM, et al. The ASK1 Inhibitor Selonsertib in Patients with Nonalcoholic Steatohepatitis: a randomized, phase 2 trial. Hepatology 2017
Jiang CF, Wen LZ, Yin C, Xu WP, Shi B, Zhang X, Xie WF. Apoptosis signal-regulating kinase 1 mediates the inhibitory effect of hepatocyte nuclear factor-4alpha on hepatocellular carcinoma. Oncotarget 2016;7:27408–27421
Nakagawa H, Hirata Y, Takeda K, Hayakawa Y, Sato T, Kinoshita H, Sakamoto K, et al. Apoptosis signal-regulating kinase 1 inhibits hepatocarcinogenesis by controlling the tumor-suppressing function of stress-activated mitogen-activated protein kinase. Hepatology 2011;54:185–195
Sayama K, Hanakawa Y, Shirakata Y, Yamasaki K, Sawada Y, Sun L, Yamanishi K, et al. Apoptosis signal-regulating kinase 1 (ASK1) is an intracellular inducer of keratinocyte differentiation. J Biol Chem 2001;276:999–1004
Eaton GJ, Zhang QS, Diallo C, Matsuzawa A, Ichijo H, Steinbeck MJ, Freeman TA. Inhibition of apoptosis signal-regulating kinase 1 enhances endochondral bone formation by increasing chondrocyte survival. Cell Death Dis 2014;5:e1522
Song J, Cho KJ, Cheon SY, Kim SH, Park KA, Lee WT, Lee JE. Apoptosis signal-regulating kinase 1 (ASK1) is linked to neural stem cell differentiation after ischemic brain injury. Exp Mol Med 2013;45:e69
Zeng T, Peng L, Chao H, Xi H, Fu B, Wang Y, Zhu Z, et al. IRE1alpha-TRAF2-ASK1 complex-mediated endoplasmic reticulum stress and mitochondrial dysfunction contribute to CXC195-induced apoptosis in human bladder carcinoma T24 cells. Biochem Biophys Res Commun 2015;460:530–536
Ishaq M, Kumar S, Varinli H, Han ZJ, Rider AE, Evans MD, Murphy AB, et al. Atmospheric gas plasma-induced ROS production activates TNF-ASK1 pathway for the induction of melanoma cancer cell apoptosis. Mol Biol Cell 2014;25:1523–1531
Lin FR, Huang SY, Hung KH, Su ST, Chung CH, Matsuzawa A, Hsiao M, et al. ASK1 promotes apoptosis of normal and malignant plasma cells. Blood 2012;120:1039–1047
White DE, Burchill SA. Fenretinide-dependent upregulation of death receptors through ASK1 and p38alpha enhances death receptor ligand-induced cell death in Ewing’s sarcoma family of tumours. Br J Cancer 2010;103:1380–1390
Hayakawa Y, Hirata Y, Nakagawa H, Sakamoto K, Hikiba Y, Otsuka M, Ijichi H, et al. Apoptosis signal-regulating kinase 1 regulates colitis and colitis-associated tumorigenesis by the innate immune responses. Gastroenterology 2010;138:1055–1067
Iriyama T, Takeda K, Nakamura H, Morimoto Y, Kuroiwa T, Mizukami J, Umeda T, et al. ASK1 and ASK2 differentially regulate the counteracting roles of apoptosis and inflammation in tumorigenesis. EMBO J 2009;28:843–853
Funding
No Grants or financial support was used in the writing of this manuscript. Dr. Chalasani has consulting agreements and research Grants with several pharmaceutical companies but none represent relevant conflict of interest for this invited review article. Dr. Satapathy has received Grant/research support from Biotest, Conatus, Genfit, Gilead Sciences, Intercept, and Shire; served on the advisory panel for Abbvie, Gilead Sciences, and Intercept; and Speakers bureau for Alexion, and Intercept pharma. None represent relevant conflict of interest for this invited review article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This review article is in compliant with general ethical considerations considered applicable for a review article. Alexander J. Kovalic, Sanjaya K. Satapathy and Naga Chalasani declare no financial conflicts of interest.
Rights and permissions
About this article
Cite this article
Kovalic, A.J., Satapathy, S.K. & Chalasani, N. Targeting incretin hormones and the ASK-1 pathway as therapeutic options in the treatment of non-alcoholic steatohepatitis. Hepatol Int 12, 97–106 (2018). https://doi.org/10.1007/s12072-018-9854-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-018-9854-1