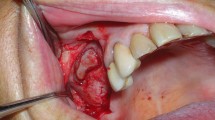
Abstract
A modification is made to the conventional way of doing Draf III by completely avoiding burrs by using only gouges and punches to reduce the postoperative narrowing followed by local instillation of budesonide solution. The results are compared. The comparison was made in terms of surgical duration, frontal ostium size, and recurrence of pathology. This is a prospective non-randomised comparative study in a private practice setting. 25 patients (15 males and 10 females) who underwent Draf III surgery between April 2012 and March 2017 were included in the study. Among them, 14 patients were assigned surgery only with punches and gouges. All the patients were given budesonide nasal instillation postoperatively. Outcomes measured included surgical duration, frontal neo-ostium size and, recurrence of pathology. They were followed up for a period of 14 months. The Student’s independent t test and χ2 test for independence of attributes were used for statistical analysis. The mean surgical duration for modified Draf III was significantly shorter than conventional Draf III (p value < 0.01). The frontal ostium remained patent in modified Draf III than the Draf III using burrs with a statistical significance (p value < 0.01). The number of cases reporting the absence of recurrence was significantly higher (p value < 0.001) in modified Draf III. Modified Draf III technique, which completely avoids the burr, takes only lesser surgical duration, keeps the frontal ostium patent and drastically reduces recurrence of pathology. Combining postoperative budesonide instillation after modified Draf III helps in achieving promising results.





Similar content being viewed by others
References
Bachert C (2016) Innovative therapeutic targets in chronic sinusitis with nasal polyps. Braz J Otorhinolaryngol 82:251–252
Wynn R, Har-El G (2004) Recurrence rates after endoscopic sinus surgery for massive sinus polyposis. Laryngoscope 114(5):811–813
Kennedy DW (1985) Functional endoscopic sinus surgery. Technique Arch Otolaryngol 111(10):643–649
Stammberger H, Posawetz W (1990) Functional endoscopic sinus surgery. Concept, indications and results of the Messerklinger technique. Eur Arch Otorhinolaryngol 247(2):63–76
Snidvongs K, Pratt E, Chin D et al (2012) Corticosteroid nasal irrigations after endoscopic sinus surgery in the management of chronic rhinosinusitis. Int Forum Allergy Rhinol 2(5):415–421
Fandiño CM, Macdonald KI, Lee J et al (2013) The use of postoperative topical corticosteroids in chronic rhinosinusitis with nasal polyps: a systematic review and meta-analysis. Am J Rhinol Allergy 27(5):e146–e157
Bassiouni A, Wormald PJ (2013) Role of frontal sinus surgery in nasal polyp recurrence. Laryngoscope 123(1):36–41
Draf W (1991) Endonasal micro-endoscopic frontal sinus surgery: the Fulda concept. Oper Tech Otolaryngol 2:234–240
Shen PH, Weitzel EK, Lai JT et al (2011) Retrospective study of full house functional endoscopic sinus surgery for revision endoscopic sinus surgery. Int Forum Allergy Rhinol 1(6):498–503
AlQahtani A, Bignami M, Terranova P, Digilio E, Basilico F, Abdulrahman S, Castelnuovo P (2014) Newly designed double-vascularized nasoseptal flap to prevent restenosis after endoscopic modified Lothrop procedure (Draf III): laboratory investigation. Eur Arch Otorhinolaryngol 271(11):2951–2955
Lee JT, Kennedy DW, Palmer JN, Feldman M, Chiu AG (2006) The incidence of concurrent osteitis in patients with chronic rhinosinusitis: a clinicopathological study. Am J Rhinol 20(3):278–282
Rajapaksa SP, Ananda A, Cain TM, Oates L, Wormald PJ (2004) Frontal ostium neo-osteogenesis and restenosis after modified endoscopic Lothrop procedure in an animal model. Clin Otolaryngol Allied Sci 29(4):386–388
Dubin MG, Kuhn FA (2005) Endoscopic modified Lothrop (Draf III) with frontal sinus punches. Laryngoscope 115(9):1702–1703
Lavigne F, Cameron L, Renzi PM et al (2002) Intrasinus administration of topical budesonide to allergic patients with chronic rhino sinusitis following surgery. Laryngoscope 112(5):858–864
Raghavan U, Logan BM (2000) New method for the effective instillation of nasal drops. J Laryngol Otol 114(6):456–459
DeConde AS, Smith TL (2016) Outcomes after frontal sinus surgery: an evidence-based review. Otolaryngol Clin North Am 49(4):1019–1033
Patel V, Choby G, Shih LC et al (2018) Equivalence in outcomes between Draf 2B vs Draf 3 frontal sinusotomy for refractory chronic frontal rhinosinusitis. Int Forum Allergy Rhinol 8(1):25–31
Ting JY, Wu A, Metson R (2014) Frontal sinus drillout (modified Lothrop procedure): long-term results in 204 patients. Laryngoscope 124(5):1066–1070
Georgalas C, Hansen F, Videler WJ et al (2011) Long terms results of Draf type III (modified endoscopic Lothrop) frontal sinus drainage procedure in 122 patients: a single centre experience. Rhinology 49(2):195–201
Kang TW, Chung JH, Cho SH et al (2017) The effectiveness of budesonide nasal irrigation after endoscopic sinus surgery in chronic Rhinosinusitis with asthma. Clin Exp Otorhinolaryngol 10(1):91–96. https://doi.org/10.21053/ceo.2016.00220
Jaksha AF, Weitzel EK, Laury AM (2016) Recent advances in the surgical management of rhinosinusitis. Research 5:F1000 Faculty Rev 2377. https://doi.org/10.12688/f1000research.9163.1
Acknowledgments
The authors wish to acknowledge Mr. Aman Paul George, Medical Student, for secretarial assistance and Mr. Iyer for the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Research Involving Human Participants and/or Animals
This study have been approved by the appropriate institutional and/or national research ethics committee and have been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Varghese, G., Sahadevan, A. Comparison of the Modified Version of DRAF III to the Conventional DRAF III: A 5 Year Study. Indian J Otolaryngol Head Neck Surg 71, 334–340 (2019). https://doi.org/10.1007/s12070-019-01628-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-019-01628-8