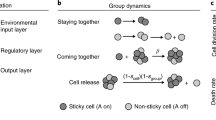
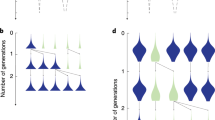
Abstract
Collective integration and processing of information have increased through the history of life, through both the formation of aggregates in which the entities may have very different properties and which jointly coarse-grained environmental variables (ranging from widely varying metabolism in microbial consortia to the ecological diversity of species on reefs) and through collectives of similar entities (such as cells within an organism or social groups). Such increases have been implicated in significant transitions in the history of life, including aspects of the origin of life, the generation of pangenomes among microbes and microbial communities such as stromatolites, multicellularity and social insects. This contribution provides a preliminary overview of the dominant modes of collective information processing in the history of life, their phylogenetic distribution and extent of convergence, and the effects of new modes for integrating and acting upon information on the tempo of evolutionary change.
Similar content being viewed by others
References
Allwood AC, Walter MR, Kamber BS, Marshall CP, Burch IW (2006) Stromatolite reef from the Early Archaean era of Australia. Nature 441:714–718
Arendt D (2008) The evolution of cell types in animals: emerging principles from molecular studies. Nat Rev Genet 8:868–882
Arendt D, Tosches MA, Marlow H (2016) From nerve net to nerve ring, nerve cord and brain: evolution of the nervous system. Nat Rev Neurosci 17:61–72. https://doi.org/10.1038/nrn.2015.15
Berezikov E (2011) Evolution of microRNA diversity and regulation in animals. Nat Rev Genet 12:846–860
Bergstrom CT, Rosvall M (2011) The transmission sense of information. Biol Philos 26:159–176. https://doi.org/10.1007/s10539-009-9180-z
Bolouri H, Davidson EH (2002) Modeling transcriptional regulatory networks. BioEssays 24:1118–1129
Boyd R (2018) A different kind of animal. Princeton University Press, Princeton
Calcott B, Sterelny K (eds) (2011) The major transitions in evolution revisited. MIT Press, Cambridge
Conway Morris S (2010) Evolution: like any other science it is predictable. Philos Trans R Soc Lond B Biol Sci 365:133–145. https://doi.org/10.1098/rstb.2009.0154
Cornforth DM et al (2014) Combinatorial quorum sensing allows bacteria to resolve their social and physical environment. Proc Natl Acad Sci USA 111:4280–4284. https://doi.org/10.1073/pnas.1319175111
Couzin ID (2009) Collective cognition in animal groups. Trends Cog Sci 13:36–43. https://doi.org/10.1016/j.tics.2008.10.002
Daniels BC, Ellison CJ, Krakauer DC, Flack JC (2016) Quantifying collectivity. Curr Opin Neurobiol 37:106–113. https://doi.org/10.1016/j.conb.2016.01.012
Darwin C (1881) The formation of vegetable mould, through the action of worMS. John Murray, London
Davidson EH, Erwin DH (2010) Evolutionary innovation and stability in animal gene networks. J Exp Zool (Mol Dev Evol) 314B:182–186
de Muizon C (1993) Walrus-like feeding adaptation in a new cetacean from the Pliocene of Peru. Nature 365:745–748
Degnan BM, Vervoort M, Larroux C, Richards GS (2009) Early evolution of metazoan transcription factors. Curr Opin Genet Dev 19:591–599. https://doi.org/10.1016/j.gde.2009.09.008
Delwiche CF (1999) Tracing the thread of plastid diversity through the tapestry of life. Am Nat 154(Suppl):S164–S177
Douglas AE (2014) Symbiosis as a general principle in eukaryotic evolution. Cold Spring Harbor Perspect Biol 6:a016113
Eme L, Spang A, Lombard J, Stairs CW, Ettema TJG (2017) Archaea and the origin of eukaryotes. Nat Rev Microbiol 15:711–723. https://doi.org/10.1038/nrmicro.2017.133
Erkenbrack EM, Davidson EH (2015) Evolutionary rewiring of gene regulatory network linkages at divergence of the echinoid subclasses. Proc Natl Acad Sci USA 112:E4075–E4084. https://doi.org/10.1073/pnas.1509845112
Erwin DH (2006) Dates and rates: temporal resolution in the deep time stratigraphic record. Ann Rev Earth Planet Sci 34:569–589
Erwin DH (2008) Macroevolution of ecosystem engineering, niche construction and diversity. Trends Ecol Evol 23:304–310
Erwin DH (2015) Early metazoan life: divergence, environment and ecology. Philos Trans R Soc B. https://doi.org/10.1098/rstb.2015.0036
Erwin DH (2016) Wonderful life revisited: chance and contingency in the Ediacaran-Cambrian radiation. In: Ramsay G, Pence CH (eds) Chance in evolution. University of Chicago Press, Chicago, pp 277–298
Erwin DH, Davidson EH (2009) The evolution of hierarchical gene regulatory networks. Nat Rev Genet 10:141–148. https://doi.org/10.1038/nrg2499
Erwin DH, Tweedt SM (2011) Ecosystem engineering and the Ediacaran-Ordovician diversification of Metazoa. Evol Ecol 26:417–433
Feinerman O, Korman A (2017) Individual versus collective cognition in social insects. J Exp Biol 220:73–82. https://doi.org/10.1242/jeb.143891
Flack JC (2012) Multiple time-scales and the developmental dynamics of social systems. Philos Trans R Soc B 367:1802–1810. https://doi.org/10.1098/rstb.2011.0214
Flack JC (2017a) Life’s information hierarchy. In: Walker SI, Davies PCW, Ellis GFR (eds) From matter to life. Cambridge University Press, Cambridge, pp 283–302
Flack JC (2017b) Coarse-graining as downward causation. Philos Trans R Soc A 375(2109):20160338
Gompel N, Prud’homme B, Wittkopp PJ, Kassner VA, Carroll SB (2005) Chance caught on the wing: cis-regulatory evolution and the origin of pigment patterns in Drosophila. Nature 433:481–487
Grotzinger JP, Knoll AH (1999) Stromatolites in Precambrian carbonates: evolutionary mileposts or environmental dipsticks? Ann Rev Earth Planet Sci 27:313–358
Hallgrimsson B et al (2019) The developmental-genetics of canalization. Sem Cell Dev Biol 88:69–79. https://doi.org/10.1016/j.semcdb.2018.05.019
Hein AM, Rosenthal SB, Hagstrom GI, Berdahl A, Torney CJ, Couzin ID (2015) The evolution of distributed sensing and collective computation in animal populations. eLife. https://doi.org/10.7554/eLife.10955
Hopfield JJ, Tank DW (1986) Computing with neural circuits: a model. Science 233:625–633. https://doi.org/10.1126/science.3755256
Jekely G, Keijzer F, Godfrey-Smith P (2015) An option space for early neural evolution. Philos Trans R Soc Lond B. https://doi.org/10.1098/rstb.2015.0181
Jensen S, Droser ML, Gehling JG (2005) Trace fossil preservation and the early evolution of animals. Palaeogeogr Palaeoclimatol Palaeoecol 220:19–29
Jones CG, Lawton JH, Shachak M (1997) Positive and negative effects of organisms as physical ecosystem engineers. Ecology 78:1946–1957
Kiessling W, Simpson C, Foote M (2010) Reefs as cradles of evolution and sources of biodiversity in the Phanerozoic. Science 327:196–198. https://doi.org/10.1126/Science.1182241
Knoll AH (2011) The multiple origins of complex multicellularity. Ann Rev Earth Planet Sci 39:217–239
Lapierrel P, Gogarten JP (2009) Estimating the size of the bacterial pan-genome. Trends Genet 25:107–110. https://doi.org/10.1016/j.tig.2008.12.004
Lopez-Garcia P, Moreira D (2015) Open questions on the origin of eukaryotes. Trends Ecol Evol 30:697–708. https://doi.org/10.1016/j.tree.2015.09.005
Maynard Smith J, Szathmary E (1995) The major transitions in evolution. W. H. Freeman, New York
McGhee GR Jr (2011) Convergent evolution. MIT Press, Cambridge
McGhee GR Jr (2016) Can evolution be directional without being teleological? Stud Hist Philos Biol Biomed Sci 58:93–99. https://doi.org/10.1016/j.shpsc.2015.12.006
McInerney J, Cummins C, Haggerty L (2011) Goods-thinking vs. tree-thinking: finding a place for mobile genetic elements. Mob Genet Elem 1:304–308. https://doi.org/10.4161/mge.19153
McInerney JO, O’Connell MJ, Pisani D (2014) The hybrid nature of the Eukaryota and a consilient view of life on Earth. Nat Rev Microbiol 12:449–455. https://doi.org/10.1038/nrmicro3271
McInerney JO, McNally A, O’Connell MJ (2017) Why prokaryotes have pangenomes. Nat Microbiol. https://doi.org/10.1038/nmicrobiol.2017.40
McShea DW (1991) Complexity and evolution: what everybody knows. Biol Philos 6:303–324
Melo D, Porto A, Cheverud JM, Marroig G (2016) Modularity: genes, development, and evolution. Ann Rev Ecol Evol Syst 47:463–486. https://doi.org/10.1146/annurev-ecolsys-121415-032409
Michod RE (1999) Darwinian dynamics. Princeton University Press, Princeton
Morris KV, Mattick JS (2014) The rise of regulatory RNA. Nat Rev Genet 15:423–437. https://doi.org/10.1038/nrg3722
Nandell CD, Bucci V, Drescher K, Levin SL, Bassler BL, Xavier JB (2014) Cutting through the complexity of cell collectives. Proc R Soc Lond B. https://doi.org/10.1098/rspb.2012.2770
Nordbotten JM, Levin SA, Szathmary E, Stenseth NC (2018) Ecological and evolutionary dynamics of interconnectedness and modularity. Proc Natl Acad Sci USA 115:750–755. https://doi.org/10.1073/pnas.1716078115
Odling-Smee FJ, Laland KN, Feldman MW (2003) Niche construction: the neglected process in evolution. Princeton University Press, Princeton
Peak D, West JD, Messinger SM, Mott KA (2004) Evidence for complex, collective dynamics and emergent, distributed computation in plants. Proc Natl Acad Sci USA 101:918–922. https://doi.org/10.1073/pnas.0307811100
Peter IS, Davidson EH (2011) A gene regulatory network controlling the embryonic specification of endoderm. Nature 474:635–639
Peter IS, Davidson EH (2015) Genomic control processes. Development and evolution. Academic Press, London
Peter IS, Davidson EH (2017) Assessing regulatory information in developmental gene regulatory networks. Proc Natl Acad Sci USA 114:5862–5869. https://doi.org/10.1073/pnas.1610616114
Peter IS, Faure E, Davidson EH (2012) Predictive computation of genomic logic processing functions in embryonic development. Proc Natl Acad Sci USA 109:16434–16442. https://doi.org/10.1073/pnas.1207852109
Pigliucci M (2008) Is evolvability evolvable? Nat Rev Genet 9:75–82
Plotnick RE (2003) Ecological and L-system based simulations of trace fossils. Palaeogeogr Palaeoclimatol Palaeoecol 192:45–58. https://doi.org/10.1016/S0031-0182(02)00678-8
Queller DC (1997) Cooperators since life began. Quart Rev Biol 72:184–188
Rebeiz M, Patel NH, Hinman VF (2015) Unraveling the tangled skein: the evolution of transcriptional regulatory networks in development. Ann Rev Genom Human Genet 16:103–131. https://doi.org/10.1146/annurev-genom-091212-153423
Reid P, Dupraz CD, Visscher PT, Sumner DY (2003) Microbial processes forming marine stromatolites. In: Krumbein WE, Paterson DM, Zavarzin GA (eds) Fossil and recent biofilms. Springer, Dordrecht, pp 103–118
Schmitz JF, Zimmer F, Bornberg-Bauer E (2016) Mechanisms of transcription factor evolution in Metazoa. Nucl Acids Res 44:6287–6297. https://doi.org/10.1093/nar/gkw492
Sebe-Pedros A, de Mendoza A (2016) Transcription factors and the origin of animal multicellularity. In: Ruiz-Trillo I, Nedelcu AM (eds) Evolutionary transitions to multicellular life, vol 2. Springer, Dordrecht, pp 379–394
Sebe-Pedros A, Ruiz-Trillo I, de Mendoza A, Lang BF, Degnan BM (2011) Unexpected repertoire of metazoan transcription factors in the unicellular holozoan Capsaspora owczarzaki. Mol Biol Evol 28:1241–1254. https://doi.org/10.1093/molbev/msq309
Sebe-Pedros A et al (2016) The dynamic regulatory genome of Capsaspora and the origin of animal multicellularity. Cell 165:1224–1237. https://doi.org/10.1016/j.cell.2016.03.034
Seilacher A (1986) Evolution of behavior as expressed in marine trace fossils. In: Nitecki MH, Kitchel JA (eds) Evolution of animal behavior. Oxford University Press, Oxford, pp 62–87
Shapiro JA (1988) Thinking about bacterial populations as multicellular organisms. Ann Rev Microbiol 52:81–104
Smaldino PE, Richerson PJ (2013) Human cumulative cultural evolution as a form of distributed computation. In: Mechelucci P (ed) Handbook of human computation. Springer, New York, pp 979–992
Szabo A, Mayor R (2016) Modelling collective cell migration of neural crest. Curr Opin Cell Biol 42:22–28. https://doi.org/10.1016/j.ceb.2016.03.023
Takahashi KH (2018) Multiple modes of canalization: links between genetic, environmental canalizations and developmental stability, and their trait-specificity. Sem Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2018.05.018
Tosches MA (2017) Developmental and genetic mechanisms of neural circuit evolution. Dev Biol 431:16–25. https://doi.org/10.1016/j.ydbio.2017.06.016
Tweedt SM, Erwin DH (2015) Origin of metazoan developmental toolkits and their expression in the fossil record. In: Ruiz-Trillo I, Nedelcu AM (eds) Evolution of multicellularity. Academic Press, London, pp 47–77
Valiant L (2013) Probably approximately correct. Basic Books, New York
Van Belleghem SM et al (2017) Complex modular architecture around a simple toolkit of wing pattern genes. Nat Ecol Evol 1:52. https://doi.org/10.1038/s41559-016-0052
Wagner GP, Altenberg L (1996) Complex adaptations and the evolution of evolvability. Evolution 50:967–976
Wagner PJ, Kosnik MA, Lidgard S (2006) Abundance distributions imply elevated complexity of post-Paleozoic marine ecosystems. Science 314:1289–1292
Wagner GP, Pavlicev M, Cheverud JM (2007) The road to modularity. Nat Rev Genet 8:921–931
Waters CM, Bassler BL (2005) Quorum sensing: cell-to-cell communication in bacteria. Ann Rev Cell Dev Biol 21:319–346. https://doi.org/10.1146/annurev.cellbio.21.012704.131001
Watson RA, Szathmary E (2016) How can evolution learn? Trends Ecol Evol 31:147–157. https://doi.org/10.1016/j.tree.2015.11.009
Watson RA et al (2016) Evolutionary connectionism: algorithmic principles underlying the evolution of biological organisation in evo-devo, evo-eco and evolutionary transitions. Evol Biol 43:553–581. https://doi.org/10.1007/s11692-015-9358-z
Webster M (2018) Morphological homeostasis in the fossil record. Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2018.05.016
Wood R (1999) Reef evolution. Oxford University Press, Oxford
Acknowledgements
I appreciate the opportunity extended by the organizers to participate in the Santa Fe Institute working group meeting on collective computation (B. Daniels, J. Flack and M. Laubichler) and to contribute to this special issue. The discussions among the participants in the meeting also greatly influenced the resulting paper. I was supported by NASA through National Astrobiology Institute (Grant No. NNA13AA90A) to the MIT node.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Special Issue on Quantifying Collectivity.
Rights and permissions
About this article
Cite this article
Erwin, D.H. Tempos and modes of collectivity in the history of life. Theory Biosci. 140, 343–351 (2021). https://doi.org/10.1007/s12064-019-00303-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12064-019-00303-4