Abstract
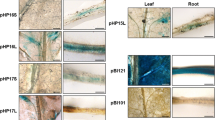
Chloroplastic pyruvate phosphate dikinase (PPDK) (chlPPDK) is a key enzyme in the photosynthesis of C4 plants. PPDK is expressed in high abundance in C4 plants but only in trace amounts in C3 plants. The existing research reveals a higher expression of MechlPPDK in cultivated cassava varieties than that in wild cassava W14. However, knowledge about the transcriptional regulation of the MechlPPDK gene in cassava (Manihot esculenta Crantz) is insufficient. Therefore, we aim to identify the transcription profile of MechlPPDK and the core promoter region of MechlPPDK. We cloned the MechlPPDK coding sequence and its 5′-flanking sequence from the cassava variety Ku50. A series of deletion constructions fused to a uidA reporter gene were performed in the 5′-flanking sequence and stably transformed into tobacco. We found that P1 (from −590 bp to +114 bp) had the highest promoter activity among P1 to P3. The 5′-flanking sequence of MechlPPDK responded differently to varied abiotic stresses. Our results will further the understanding of the regulation of MechlPPDK expression.





Similar content being viewed by others
Abbreviations
- CDS:
-
Coding sequence
- EDTA:
-
Ethylene diamine tetraacetic acid
- PEPC:
-
Phosphoenolpyruvate carboxylase
- PPDK:
-
Pyruvate phosphate dikinase
- RACE:
-
Rapid-amplification of cDNA ends
- TSS:
-
Transcriptional start site
- GUS:
-
β-glucuronidase
References
Allen GC, Floresvergara MA, Krasynanski S, Kumar S, Thompson WF (2006) A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc 1:2320–2325. https://doi.org/10.1038/nprot.2006.384
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Ceballos H, Okogbenin E, Pérez JC, Debouck DG (2010) Cassava. Handbook Plant Breed 7(1):53–96
Doubnerova HV, Miedzinska L, Dobra J, Vankova R, Ryslava H (2014) Phosphoenolpyruvate carboxylase, NADP-malic enzyme, and pyruvate, phosphate dikinase are involved in the acclimation of Nicotiana tabacum L. to drought stress. J Plant Physiol 171(5):19–25. https://doi.org/10.1016/j.jplph.2013.10.017
Eastmond PJ, Astley HM, ParsleyK AS, Williams BP, Menard GN (2015) Arabidopsis uses two gluconeogenic gateways for organic acids to fuel seedling establishment. Nat Commun 6:6659. https://doi.org/10.1038/ncomms7659
El-Sharkawy MA (2004) Cassava biology and physiology. Plant Mol Biol 56(4):481–501. https://doi.org/10.1007/s11103-005-2270-7
El-Sharkawy MA (2006) International research on cassava photosynthesis, productivity, eco-physiology, and responses to environmental stresses in the tropics. Photosynthetica 44(4):481–512. https://doi.org/10.1007/s11099-006-0063-0
Feng XM, Cao LJ, Adam RD, Zhang XC, Lu SQ (2008) The catalyzing role of ppdk in giardia lamblia. Biochem Biophys Res Commun 367(2):394–398. https://doi.org/10.1016/j.bbrc.2007.12.139
Fukayama H, Tsuchida H, Agarie S, Nomura M, Onodera H, Ono K, Lee BH, Hirose S, Toki S, Ku MSB, Makino A, Matsuoka M, Miyao M (2001) Significant accumulation of c4-specific pyruvate, orthophosphate dikinase in a C3 plant, Rice. Plant Physiol 127(3):1136–1146. https://doi.org/10.1104/pp.127.3.1136
Huang S, Greenway H, Colmer TA (2005) Protein synthesis by rice coleoptiles during prolonged anoxia: implications for glycolysis, growth and energy utilization. Ann Bot 96(4):703–715. https://doi.org/10.1093/aob/mci222
IBM Corporation (2011) SPSS statistics software. Release 19.0 IBM Corp., Armonk, NY
Imaizumi N, Ku MSB, Ishihara K, Samejima M, Kaneko S, Matsuoka M (1997) Characterization of the gene for pyruvate,orthophosphate dikinase from rice, a c3 plant, and a comparison of structure and expression between c3 and c4 genes for this protein. Plant Mol Biol 34(5):701–716. https://doi.org/10.1023/A:1005884515840 Characterization of the gene for pyruvate,orthophosphate dikinase from rice, a C3 plant, and a comparison of structure and expression between C3 and C4 genes for this protein
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907. https://doi.org/10.1089/dna.1987.6.583
Jefferson RA (1988) Plant reporter genes: the GUS gene fusion system. Setlow JK (ed) Gen Eng 10:247–263. https://doi.org/10.1007/978-1-4615-7081-3_13
Kurai T, Wakayama M, Abiko T, Yanagisawa S, Aoki N, Ohsugi R (2011) Introduction of the ZmDof1 gene into rice enhances carbon and nitrogen assimilation under low-nitrogen conditions. Plant Biotechnol J 9(8):826–837. https://doi.org/10.1111/j.1467-7652.2011.00592.x
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 −ΔΔ C T method. Methods 25:402–408. https://doi.org/10.1006/meth.2001
Matsuoka M, Numazawa T (1991) Cis-acting elements in the pyruvate, orthophosphate dikinase gene from maize. Mol Gen Genet 228(1–2):143–152. https://doi.org/10.1007/bf00282459
Matsuoka M, Tada Y, Fujimura T, Kano-Murakami Y (1993) Tissue-specific light-regulated expression directed by the promoter of a C4 gene, maize pyruvate,orthophosphate dikinase, in a C3 plant, rice. Proc Natl Acad Sci USA 90(20):9586–9590. https://doi.org/10.1073/pnas.90.20.9586
Moons A, Valcke R, Van MM (1998) Low-oxygen stress and water deficit induce cytosolic pyruvate orthophosphate dikinase (PPDK) expression in roots of rice, a C3 plant. Plant J 15(1):89–98. https://doi.org/10.1046/j.1365-313X.1998.00185.x
Naidu SL (2003) Cold tolerance of C4 photosynthesis in Miscanthus x giganteus: adaptation in amounts and sequence of C4 photosynthetic enzymes. Plant Physiol 132(3):1688–1697. https://doi.org/10.1104/pp.103.021790
Omoto E, Taniguchi M, Miyake H (2012) Adaptation responses in C4 photosynthesis of maize under salinity. J Plant Physiol 169(5):469–477. https://doi.org/10.1016/j.jplph.2011.11.009
Parsley K, Hibberd JM (2006) The Arabidopsis ppdk gene is transcribed from two promoters to produce differentially expressed transcripts responsible for cytosolic and plastidic proteins. Plant Mol Biol 62(3):339–349
Riechmann JL, Ratcliffe OJ (2000) A genomic perspective on plant transcription factors. Curr Opin Plant Biol 3(5):423–434. https://doi.org/10.1016/S1369-5266(00)00107-2
Rombauts S, Déhais P, Van MM, RouzéP (1999) PlantCARE, a plant cis-acting regulatory element database. Nucleic Acids Res 27: 295–296 doi: https://doi.org/10.1093/nar/27.1.295
Rosche E, Westhoff P (1995) Genomic structure and expression of the pyruvate, orthophosphate dikinase gene of the dicotyledonous C4 plant Flaveria trinervia (Asteraceae). Plant Mol Biol 29(4):663–678. https://doi.org/10.1007/bf00041157
Taylor L, Nunesnesi A, Parsley K, Leiss A, Leach G, Coates S, Wingler A, Ernie A, Ibberd JM (2010) Cytosolic pyruvate,orthophosphate dikinase functions in nitrogen remobilization during leaf senescence and limits individual seed growth and nitrogen content. Plant J 62(4):641–652. https://doi.org/10.1111/j.1365-313X.2010.04179.x
Wang H, Liu C, Ma PA, Lu C, Li K, Wang W (2018) Functional characterization of cytosolic pyruvate phosphate dikinase gene, and promoter, of cassava in response to abiotic stress in transgenic tobacco. Crop Sci 58:2002–2009. https://doi.org/10.2135/cropsci2018.03.0204
Wang W, Feng B, Xiao J, Xia Z, Zhou X, Li P, Zhang W, Wang Y (2014) Cassava genome from a wild ancestor to cultivated varieties. Nat Commun 5(5):5110. https://doi.org/10.1038/ncomms6110
Yanagisawa S (1998) Involvement of maize Dof zinc finger proteins in tissue-specific and light-regulated gene expression. Plant Cell 10:75–89. https://doi.org/10.1105/tpc.10.1.75
Yu H, Wang T (2016) Proteomic dissection of endosperm starch granule associated proteins reveals a network coordinating starch biosynthesis and amino acid metabolism and glycolysis in Rice endosperms. Front Plant Sci 7:707. https://doi.org/10.3389/fpls.2016.00707
Zhang Y, Chen X, Lu C, Wang WQ (2012) Cloning and expression analysis of phosphoenolpyruvate carboxylase gene pepc from cassava (Manihot esculenta Crantz). J Trop Subtrop Botany:369–375
Zhang Y, Verhoeff NI, Chen Z, Chen S, Wang M, Zhu Z, Ouwerkerk PB (2015) Functions of OsDof25 in regulation of OsC4PPDK. Plant Mol Biol 89:229–242. https://doi.org/10.1007/s11103-015-0357-3
Acknowledgements
This study was funded by the National Natural Science Foundation of China (Grant No. 31701509) and the China Agriculture Research System (CARS-12).
Author information
Authors and Affiliations
Contributions
Kaimian Li and Wenquan Wang directed and designed the experiments. Haiyan Wang performed most of the research and drafted the manuscript. Xu Shen planted the transformed tobacco plants, and Cheng Lu performed some field experiments. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Communicated by: Elliosha Hajari
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 58 kb)
ESM 2
Fig. A1. Structure of MechlPPDK gene. Location of exons and introns are indicated by box and line between boxes. The numbers above the box indicate length of exons. Red box indicates 5’untranslated region and white box indicates 3’untranslated region. ATG and TGA indicate translation initiation codon and termination codon (PNG 897 kb)
ESM 3
Fig.A2. The map of pCAMBIA1381Z:pMechPPDK (PNG 870 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Shen, X., Lu, C. et al. Molecular Functional Characterisation of MechlPPDK Promoter in Transgenic Tobacco. Tropical Plant Biol. 13, 287–295 (2020). https://doi.org/10.1007/s12042-020-09257-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12042-020-09257-0