Abstract
Ca(OTf)2/Bu4NPF6 catalytic system has been illustrated for the synthesis of Diels–Alder adduct for the first time. This procedure tolerates substrate diversity and delivers high yield. Use of environmentally benign catalyst, high yields and substrate diversity are the highlight of the existing method.
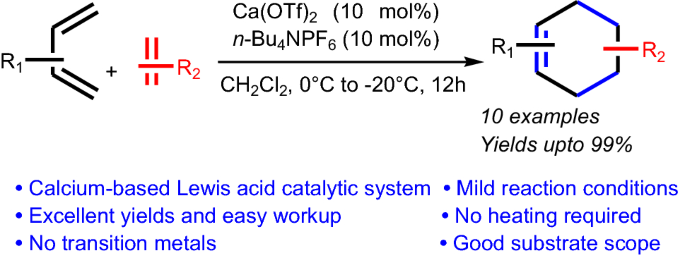
Graphic Abstract
Ca(OTf)2, along with n-Bu4NPF6 as the co-catalyst, is shown to be an effective Lewis acidic catalyst system for the Diels–Alder reaction. Apart from being sustainable and efficient, this procedure is robust and products are obtained in near-quantitative yields.

References
Diels O and Alder K 1928 Synthesen in der hydroaromatischen Reihe Justus Liebigs Ann. Chem. 460 98
Dewar M J S, Olivella S and Stewart J J P 1986 Mechanism of the Diels–Alder reaction: reactions of butadiene with ethylene and cyanoethylenes J. Am. Chem. Soc. 108 5771
Yates P and Eaton P 1960 Acceleration of the Diels–Alder reaction by aluminum chloride J. Am. Chem. Soc. 82 4436
Fray GI and Robinson R 1961 Catalysis of the Diels–Alder reaction J. Am. Chem. Soc. 83 249
Houk K N and Strozier R W 1973 Lewis acid catalysis of Diels–Alder reactions J. Am. Chem. Soc. 95 4094
Rideout DC and Breslow R 1980 Hydrophobic acceleration of Diels–Alder reactions J. Am. Chem. Soc. 102 7816
Ruiz-Lopez M F, Assfeld X, Garcia J I, Mayoral J A and Salvatella L 1993 Solvent effects on the mechanism and selectivities of asymmetric Diels–Alder reactions J. Am. Chem. Soc. 115 8780
Breslow R and Maitra U 1984 On the origin of product selectivity in aqueous Diels–Alder reactions Tetrahedron Lett. 25 1239
Blokzij W, Blandamer M J and Engberts J B F N 1991 Diels–Alder reactions in aqueous solutions. Enforced hydrophobic interactions between diene and dienophile J. Am. Chem. Soc. 113 4241
Kumar A 1994 Can internal pressure describes the effect of salt in aqueous Diels–Alder reactions? A possible explanation J. Org. Chem. 59 230
Jørgensen K A 2000 Catalytic Asymmetric Hetero‐Diels–Alder Reactions of Carbonyl Compounds and Imines Angew. Chem. Int. Ed. 39 3558
Schaus S E, Brånalt J and Jacobsen E N 1998 Asymmetric Hetero-Diels–Alder Reactions Catalyzed by Chiral (Salen)Chromium(III) Complexes J. Org. Chem. 63 403
Deguin B and Vogel P 1992 Hetero-Diels–Alder addition of sulfur dioxide to 1,3-dienes. Suprafaciality, regioselectivity, and stereoselectivity J. Am. Chem. Soc. 114 9210
Streith J and Defoin A 1994 Hetero Diels–Alder Reactions with Nitroso Dienophiles: Application to the Synthesis of Natural Product Derivatives Synth. 11 1107
Waldmann H 1994 Asymmetric Hetero Diels–Alder Reactions Synth. 6 535
Houk K N, Lin Y T and Brown F K 1986 Evidence for the concerted mechanism of the Diels–Alder reaction of butadiene with ethylene J. Am. Chem. Soc. 108 554
Kagan H B and Riant O 1992 Catalytic asymmetric Diels–Alder reactions Chem. Rev. 92 1007
Nicolaou K C, Snyder S A, Montagnon T and Vassilikogiannakis G 2002 The Diels–Alder reaction in total synthesis Angew. Chem. Int. Ed Engl. 41 1668
Du H, Long J, Hu J, Li X and Ding K 2002 3,3‘-Br2-BINOL-Zn Complex: A Highly Efficient Catalyst for the Enantioselective Hetero-Diels−Alder Reaction Org. Lett. 42 4349
Hara K, Akiyama R and Sawamura M 2005 Strong Counteranion Effects on the Catalytic Activity of Cationic Silicon Lewis Acids in Mukaiyama Aldol and Diels−Alder Reactions Org. Lett. 25 5621
Fang X, Chen X and Chi YC 2011 Enantioselective Diels–Alder Reactions of Enals and Alkylidene Diketones Catalyzed by N-Heterocyclic Carbenes Org. Lett. 13 4708
Funel J A and Abele S 2013 Industrial Applications of the Diels–Alder Reaction Angew. Chem. Int. Ed. 52 3822
Goussé C and Gandini A 1999 Diels–Alder polymerization of difurans with bismaleimides Polym. Int. 48 723
Gacal B, Durmaz H, Tasdelen MA, Hizal G, Tunca U, Yagci Y and Demirel A L 2006 Anthracene−Maleimide-Based Diels−Alder “Click Chemistry” as a Novel Route to Graft Copolymers Macromolecules 39 5330
Plass T, Milles S, Koehler C, Szymański J, Mueller R, Wiessler M, Schultz C and Lemke EA 2012 Amino acids for Diels–Alder reactions in living cells Angew. Chem. Int. Ed. Engl. 51 4166
Liu Y-L and Chuoa T-W 2013 Self-healing polymers based on thermally reversible Diels–Alder chemistry Polym. Chem. 4 2194
Shi Z, Hau S, Luo J, Kim T D, Tucker N M, Ka J W and Jen A K Y 2007 Highly Efficient Diels–Alder Crosslinkable Electro‐Optic Dendrimers for Electric‐Field Sensors Adv. Func. Mat. 17 2557
Sarkar S, Bekyarova E and Haddon R C 2012 Chemistry at the Dirac Point: Diels–Alder Reactivity of Graphene Acc. Chem. Res. 45 673
Gates M and Tschudi G 1952 The synthesis of morphine J. Am. Chem. Soc. 74 1109
Woodward R B, Sondheimer F, Taub D, Heusler K and McLamore W M 1952 The Total Synthesis of Steroids J. Am. Chem. Soc. 74 4223
Corey E J, Guzman-Perez A and Loh T-P 1994 Demonstration of the Synthetic Power of Oxazaborolidine-Catalyzed Enantioselective Diels–Alder Reactions by Very Efficient Routes to Cassiol and Gibberellic Acid J. Am. Chem. Soc. 116 3611
Begouin J-M and Niggemann M 2013 Based Lewis Acid Catalysts Chem. Eur. J. 19 8030
Acknowledgements
This work is generously supported by the Department of Science and Technology (DST- SERB/ ECR/2015/000363) India to SM.
Author information
Authors and Affiliations
Contributions
S.M. and R.K. carried out the synthesis, purification, and characterization of the synthesized molecules. S.M. directed the synthetic work and wrote the paper based upon drafts written by R.K.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kalepu, R., Mishra, S. Lewis acid catalyst system for Diels–Alder reaction. J Chem Sci 132, 48 (2020). https://doi.org/10.1007/s12039-020-1749-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-020-1749-8

