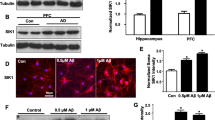
Abstract
Activation of glial cells (including microglia and astrocytes) appears central to the initiation and progression of neuroinflammation in Alzheimer’s disease (AD). The low-density lipoprotein receptor-related protein 1 (LRP1) is a major receptor for amyloid-β (Aβ), which plays a critical role in AD pathogenesis. LRP1 regulates inflammatory response by modulating the release of pro-inflammatory cytokines and phagocytosis. However, the effects of LRP1 on microglia- and astrocytic cell-mediated neuroinflammation and their underlying mechanisms in AD remain unclear. Therefore, using APP/PS1 transgenic mice, we found that LRP1 is downregulated during disease progression. Silencing of brain LRP1 markedly exacerbated AD-related neuropathology including Aβ deposition, neuroinflammation, and synaptic and neuronal loss, which was accompanied by a decline in spatial cognitive ability. Further mechanistic study revealed that silencing of LRP1 initiated neuroinflammation by increasing microgliosis and astrogliosis, enhancing pro-inflammatory cytokine production, and regulating toll-like receptor 4 (TLR4)-mediated activation of nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathways. Taken together, these findings indicated that LRP1 suppresses microglia and astrocytic cell activation by modulating TLR4/NF-κB/MAPK signaling pathways. Our results further provide insights into the role of LRP1 in AD pathogenesis and highlight LRP1 as a potential therapeutic target for the treatment of AD.










Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid-β
- BBB:
-
Blood-brain barrier
- GFAP:
-
Glial fibrillary acidic protein
- Iba1:
-
Ionized calcium-binding adapter molecule 1
- LRP1:
-
Low-density lipoprotein receptor-related protein 1
- LPS:
-
Lipopolysaccharide
- IL-1β:
-
Interleukin-1β
- IL-6:
-
Interleukin-6
- MAPKs:
-
Mitogen-activated protein kinases
- MWM:
-
Morris water maze
- MyD88:
-
Myeloid differentiation primary response protein 88
- NFTs:
-
Neurofibrillary tangles
- NF-κB:
-
Nuclear factor-kappa B
- TLR4:
-
Toll-like receptor 4
- TNF-α:
-
Tumor necrosis factor-α
- TRAF6:
-
Tumor necrosis factor receptor-associated factor 6
References
Ono K (2018) Alzheimer’s disease as oligomeropathy. Neurochem Int 119:57–70
Salinaro AT, Pennisi M, Di Paola R, Scuto M, Crupi R, Cambria MT, Ontario ML, Tomasello M et al (2018) Neuroinflammation and neurohormesis in the pathogenesis of Alzheimer’s disease and Alzheimer-linked pathologies: modulation by nutritional mushrooms. Immun Ageing 15:8
Morales I, Guzman-Martinez L, Cerda-Troncoso C, Farias GA, Maccioni RB (2014) Neuroinflammation in the pathogenesis of Alzheimer’s disease A rational framework for the search of novel therapeutic approaches. Front Cell Neurosci 8:112
Woo JH, Lee JH, Kim H, Park SJ, Joe EH, Jou I (2015) Control of inflammatory responses: a new paradigm for the treatment of chronic neuronal diseases. Exp Neurobiol 24:95–102
Ransohoff RM (2016) How neuroinflammation contributes to neurodegeneration. Science 353:777–783
Wang S, Zhang X, Zhai L, Sheng X, Zheng W, Chu H, Zhang G (2018) Atorvastatin attenuates cognitive deficits and neuroinflammation induced by A beta(1-42) involving modulation of TLR4/TRAF6/NF-kappa B pathway. J Mol Neurosci 64:363–373
Muhammad T, Ikram M, Ullah R, Rehman SU, Kim MO (2019) Hesperetin, a citrus flavonoid, attenuates LPS-induced neuroinflammation, apoptosis, and memory impairments by modulating TLR4/NF-kappaB signaling. Nutrients 11:648
Dilshara MG, Lee KT, Jayasooriya RGPT, Kang CH, Park SR, Choi YH, Choi IW, Hyun JW et al (2014) Downregulation of NO and PGE(2) in LPS-stimulated BV2 microglial cells by trans-isoferulic acid via suppression of PI3K/Akt-dependent NF-kappa B and activation of Nrf2-mediated HO-1. Int Immunopharmacol 18:203–211
Park HY, Kim TH, Kim CG, Kim GY, Kim CM, Kim ND, Kim BW, Hwang HJ et al (2013) Purpurogallin exerts anti-inflammatory effects in lipopolysaccharide-stimulated BV2 microglial cells through the inactivation of the NF-kappa B and MAPK signaling pathways. Int J Mol Med 32:1171–1178
De Vita T, Albani C, Realin N, Migliore M, Basit A, Ottonello G, Cavalli A (2019) Inhibition of serine palmitoyltransferase by a small organic molecule promotes neuronal survival after astrocyte amyloid beta 1-42 injury. ACS Chem Neurosci 10:1627–1635
Seok SM, Park TY, Park HS, Baik EJ, Lee SH (2015) Fructose-1,6-bisphosphate suppresses lipopolysaccharide-induced expression of ICAM-1 through modulation of toll-like receptor-4 signaling in brain endothelial cells. Int Immunopharmacol 26:203–211
Xia W, Luo P, Hua P, Ding P, Li C, Xu J, Zhou H, Gu Q (2019) Discovery of a new pterocarpan-type antineuroinflammatory compound from Sophora tonkinensis through suppression of the TLR4/NF kappa B/MAPK signaling pathway with PUI as a potential target. ACS Chem Neurosci 10:295–303
Yepes M, Sandkvist M, Moore EG, Bugge TH, Strickland DK, Lawrence DA (2003) Tissue-type plasminogen activator induces opening of the blood-brain barrier via the LDL receptor-related protein. J Clin Invest 112:1533–1540
May P, Rohlmann A, Bock HH, Zurhove K, Marth JD, Schomburg ED, Noebels JL, Beffert U et al (2004) Neuronal LRP1 functionally associates with postsynaptic proteins and is required for normal motor function in mice. Mol Cell Biol 24:8872–8883
Chuang TY, Guo Y, Seki SM, Rosen AM, Johanson DM, Mandell JW, Lucchinetti CF, Gaultier A (2016) LRP1 expression in microglia is protective during CNS autoimmunity. Acta Neuropathol Commun 4:UNSP 68
Wyss-Coray T, Loike JD, Brionne TC, Lu E, Anankov R, Yan FR, Silverstein SC, Husemann J (2003) Adult mouse astrocytes degrade amyloid-beta in vitro and in situ. Nat Med 9:453–457
Liu Q, Zerbinatti CV, Zhang J, Hoe HS, Wang B, Cole SL, Herz J, Muglia L et al (2007) Amyloid precursor protein regulates brain apolipoprotein e and cholesterol metabolism through lipoprotein receptor LRP1. Neuron 56:66–78
Lillis AP, Van Duyn LB, Murphy-Ullrich JE, Strickland DK (2008) LDL receptor-related protein 1: unique tissue-specific functions revealed by selective gene knockout studies. Physiol Rev 88:887–918
Liu CC, Hu J, Zhao N, Wang J, Wang N, Cirrito JR, Kanekiyo T, Holtzman DM et al (2017) Astrocytic LRP1 mediates brain a beta clearance and impacts amyloid deposition. J Neurosci 37:4023–4031
Van Acker ZP, Bretou M, Annaert W (2019) Endo-lysosomal dysregulations and late-onset Alzheimer’s disease: impact of genetic risk factors. Mol Neurodegener 14:20
Wujak L, Schnieder J, Schaefer L, Wygrecka M (2018) LRP1: a chameleon receptor of lung inflammation and repair. Matrix Biol 68–69:366–381
Liu Q, Trotter J, Zhang J, Peters MM, Cheng H, Bao J, Han X, Weeber EJ et al (2010) Neuronal LRP1 knockout in adult mice leads to impaired brain lipid metabolism and progressive, age-dependent synapse loss and neurodegeneration. J Neurosci 30:17068–17078
Mantuano E, Brifault C, Lam MS, Azmoon P, Gilder AS, Gonias SL (2016) LDL receptor-related protein-1 regulates NF kappa B and microRNA-155 in macrophages to control the inflammatory response. Proc Natl Acad Sci U S A 113:1369–1374
May P, Bock HH, Nofer JR (2013) Low density receptor-related protein 1 (LRP1) promotes anti-inflammatory phenotype in murine macrophages. Cell Tissue Res 354:887–889
Schubert K, Collins LE, Green P, Nagase H, Troeberg L (2019) LRP1 controls TNF release via the TIMP-3/ADAM17 axis in endotoxin-activated macrophages. J Immunol 202:1501–1509
Gaultier A, Arandjelovic S, Li X, Janes J, Dragojlovic N, Zhou GP, Dolkas J, Myers RR et al (2008) A shed form of LDL receptor-related protein-1 regulates peripheral nerve injury and neuropathic pain in rodents. Eur J Clin Investig 118:161–172
Yang L, Liu CC, Zheng H, Kanekiyo T, Atagi Y, Jia L, Wang D, N'Songo A et al (2016) LRP1 modulates the microglial immune response via regulation of JNK and NF-kappa B signaling pathways. J Neuroinflamm 13:304
Roy DS, Arons A, Mitchell TI, Pignatelli M, Ryan TJ, Tonegawa S (2016) Memory retrieval by activating engram cells in mouse models of early Alzheimer’s disease. Nature 531:508–512
Jiang T, Tan L, Zhu XC, Zhang QQ, Cao L, Tan MS, Gus LZ, Wang HF et al (2014) Upregulation of TREM2 ameliorates neuropathology and rescues spatial cognitive impairment in a transgenic mouse model of Alzheimer’s disease. Neuropsychopharmacology 39:2949–2962
Jiang T, Tan L, Zhu XC, Zhou JS, Cao L, Tan MS, Wang HF, Chen Q et al (2015) Silencing of TREM2 exacerbates tau pathology, neurodegenerative changes, and spatial learning deficits in P301S tau transgenic mice. Neurobiol Aging 36:3176–3186
Dodart JC, Marr RA, Koistinaho M, Gregersen BM, Malkani S, Verma IM, Paul SM (2005) Gene delivery of human apolipoprotein E alters brain beta burden in a mouse model of Alzheimers disease. Proc Natl Acad Sci U S A 102:1211–1216
Zhao WX, Zhang JH, Cao JB, Wang W, Wang DX, Zhang XY, Yu J, Zhang YY et al (2017) Acetaminophen attenuates lipopolysaccharide-induced cognitive impairment through antioxidant activity. J Neuroinflamm 14:17
Liu C, Wu YX, Zha S, Liu MP, Wang Y, Yang GD, Ma KG, Fei YL et al (2016) Treatment effects of tanshinone IIA against ntracerebroventricular streptozotocin-induced memory deficits in mice. Brain Res 1631:137–146
Shi Y, Huang W, Wang Y, Zhang R, Hou L, Xu J, Qiu Z, Xie Q et al (2018) (9)-(−)-Meptazinol, a novel dual-binding AChE inhibitor, rescues cognitive deficits and pathological changes in APP/PS1 transgenic mice. Transl Neurodegener 7:21
Yang WN, Zhang JS, Shi LL, Ji SF, Yang XH, Zhai WY, Zong HF, Qian YH (2019) Protective effects of tanshinone IIA on SH-SY5Y cells against oA beta(1-42)-induced apoptosis due to prevention of endoplasmic reticulum stress. Int J Biochem Cell Biol 107:82–91
Ou Z, Kong X, Sun X, He X, Zhang L, Gong Z, Huang J, Xu B et al (2018) Metformin treatment prevents amyloid plaque deposition and memory impairment in APP/PS1 mice. Brain Behav Immun 69:351–363
Fu AKY, Hung KW, Yuen MYF, Zhou X, Mak DSY, Chan ICW, Cheung TH, Zhang B et al (2016) IL-33 ameliorates Alzheimer’s disease-like pathology and cognitive decline. Proc Natl Acad Sci U S A 113:E2705–E2713
Newcombe EA, Camats-Perna J, Silva ML, Valmas N, Huat TJ, Medeiros R (2018) Inflammation: the link between comorbidities, genetics, and Alzheimer’s disease. J Neuroinflamm 15:276
Liu Y, Zhang Y, Zheng X, Fang T, Yang X, Luo X, Guo A, Newell KA et al (2018) Galantamine improves cognition, hippocampal inflammation, and synaptic plasticity impairments induced by lipopolysaccharide in mice. J Neuroinflamm 15:112
Ruzicka J, Urdzikova LM, Svobodova B, Amin AG, Karova K, Dubisova J, Zaviskova K, Kubinova S et al (2018) Does combined therapy of curcumin and epigallocatechin gallate have a synergistic neuroprotective effect against spinal cord injury? Neural Regen Res 13:119–127
Silverberg GD, Messier AA, Miller MC, Machan JT, Majmudar SS, Stopa EG, Donahue JE, Johanson CE (2010) Amyloid efflux transporter expression at the blood-brain barrier declines in normal aging. J Neuropathol Exp Neurol 69:1034–1043
Shinohara M, Fujioka S, Murray ME, Wojtas A, Baker M, Rovelet-Lecrux A, Rademakers R, Das P et al (2014) Regional distribution of synaptic markers and APP correlate with distinct clinicopathological features in sporadic and familial Alzheimer’s disease. Brain 137:1533–1549
Kang DE, Pietrzik CU, Baum L, Chevallier N, Merriam DE, Kounnas MZ, Wagner SL, Troncoso JC et al (2000) Modulation of amyloid beta-protein clearance and Alzheimer’s disease susceptibility by the LDL receptor-related protein pathway. J Clin Invest 106:1159–1166
Bell RD, Deane R, Chow N, Long X, Sagare A, Singh I, Streb JW, Guo H et al (2009) SRF and myocardin regulate LRP-mediated amyloid-beta clearance in brain vascular cells. Nat Cell Biol 11:143–U83
Hsieh YH, Deng JS, Chang YS, Huang GJ (2018) Ginsenoside Rh2 ameliorates lipopolysaccharide-induced acute lung injury by regulating the TLR4/PI3K/Akt/mTOR, Raf-1/MEK/ERK, and Keap1/Nrf2/HO-1 signaling pathways in mice. Nutrients 10:E1208
Deane R, Wu ZH, Sagare A, Davis J, Yan SD, Hamm K, Xu F, Parisi M et al (2004) LRP/amyloid beta-peptide interaction mediates differential brain efflux of A beta isoforms. Neuron 43:333–344
Walter S, Letiembre M, Liu Y, Heine H, Penke B, Hao W, Bode B, Manietta N et al (2007) Role of the toll-like receptor 4 in neuro-inflammation in Alzheimer’s disease. Cell Physiol Biochem 20:947–956
Jeong J, Pandey S, Li Y, Badger JD, Lu W, Roche KW (2019) PSD-95 binding dynamically regulates NLGN1 trafficking and function. Proc Natl Acad Sci U S A 116:12035–12044
Guarnieri FC, Pozzi D, Raimondi A, Fesce R, Valente MM, Delvecchio VS, Van Esch H, Matteoli M et al (2017) A novel SYN1 missense mutation in non-syndromic X-linked intellectual disability affects synaptic vesicle life cycle, clustering and mobility. Hum Mol Genet 26:4699–4714
Head E, Corrada MM, Kahle-Wrobleski K, Kim RC, Sarsoza F, Goodus M, Kawas CH (2009) Synaptic proteins, neuropathology and cognitive status in the oldest-old. Neurobiol Aging 30:1125–1134
Whitfield DR, Vallortigara J, Alghamdi A, Howlett D, Hortobagyi T, Johnson M, Attems J, Newhouse S et al (2014) Assessment of ZnT3 and PSD95 protein levels in Lewy body dementias and Alzheimer’s disease: association with cognitive impairment. Neurobiol Aging 35:2836–2844
Giannakopoulos P, Herrmann FR, Bussiere T, Bouras C, Kovari E, Perl DP, Morrison JH, Gold G et al (2003) Tangle and neuron numbers, but not amyloid load, predict cognitive status in Alzheimer’s disease. Neurology 60:1495–1500
Arendt T (2009) Synaptic degeneration in Alzheimer’s disease. Acta Neuropathol 218:167–179
Funding
This work was supported by the Natural Science Basic Research Plan in Shaanxi Province of China (2018JM7056); China Postdoctoral Science Foundation (2017T100758, 2016M590955); Postdoctoral Science Foundation of Shaanxi Province (2016BSHYDZZ04); Undergraduates Innovating Experiment Project of Nation (GJ201910698164); Undergraduates Innovating Experiment Project of Shaanxi Province (SJ201910698089); and Natural Science Foundation of China (81500928, 81571251).
Author information
Authors and Affiliations
Contributions
WNY, YYH, JBR, and XQP designed the research, performed the majority of the experiments, and wrote the paper. BD, CHL, XYW, SFJ, and YBM performed the behavior experiments and interpreted the results. QZZ and HJ interpreted the results and gave technical support. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, Y., Ruganzu, J.B., Zheng, Q. et al. Silencing of LRP1 Exacerbates Inflammatory Response Via TLR4/NF-κB/MAPKs Signaling Pathways in APP/PS1 Transgenic Mice. Mol Neurobiol 57, 3727–3743 (2020). https://doi.org/10.1007/s12035-020-01982-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-01982-7